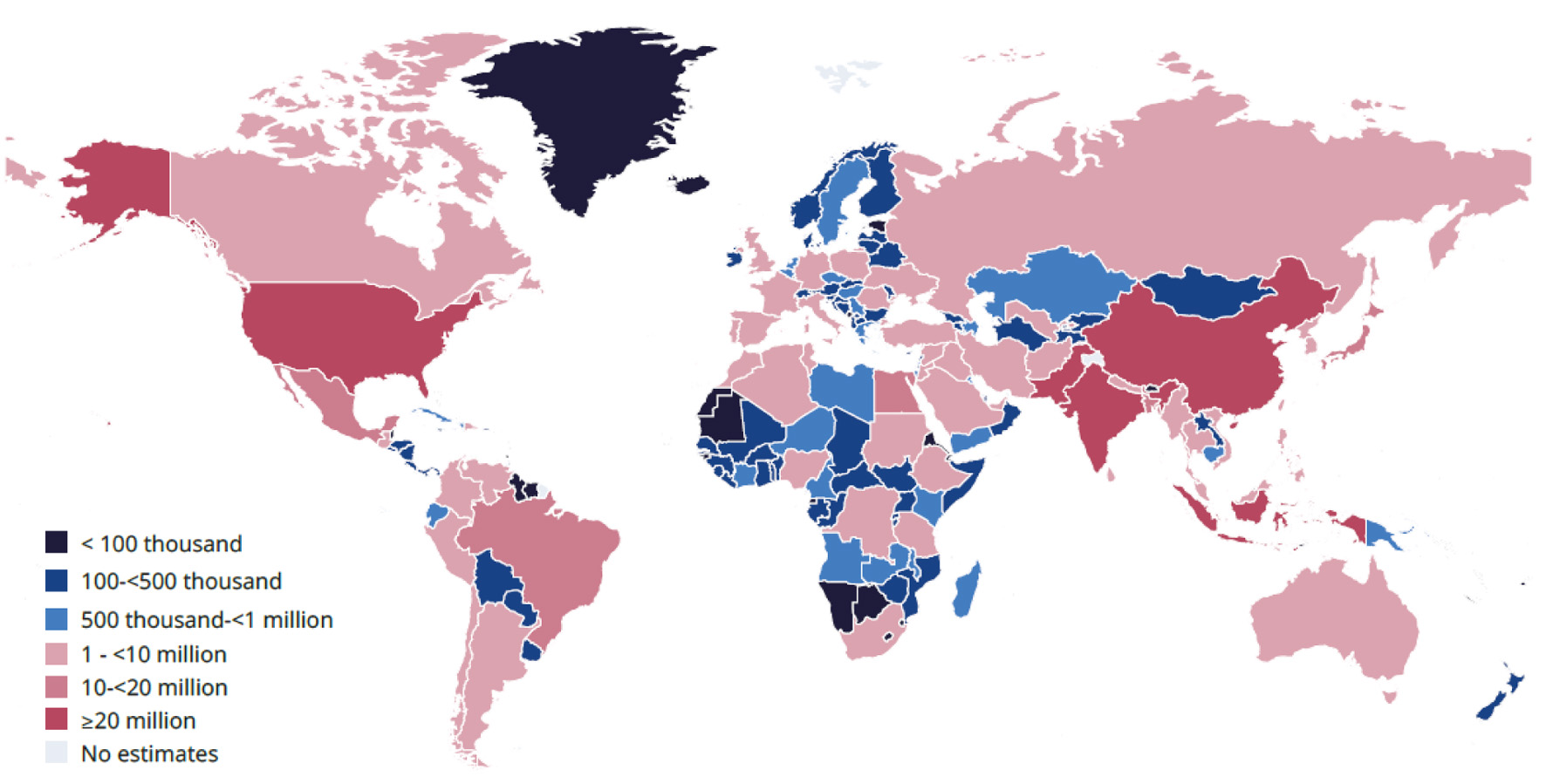
↓ Figure 1. Estimated number of adults
(20–79 years old) with diabetes by country in 2024. The International Diabetes Federation (IDF)
has granted permission for copyright for the requested purpose.
| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Review
Volume 16, Number 1, February 2026, pages 1-14
Imeglimin: Discovery, Pharmacology, and Trials
Figures

Tables
| Characteristic | Details/data |
|---|---|
| AUC: area under the curve; Cmax: maximum plasma concentration; DPP-4: dipeptidyl-peptidase-4; GIP: glucose-dependent insulinotropic polypeptide; GLP-1: glucagon-like peptide-1; HbA1c: glycated hemoglobin; MATE1: multidrug and toxin extrusion transporter 1; NAD+: nicotinamide adenine dinucleotide; OGTT: oral glucose-tolerance test; QT/QTc: QT interval/heart-rate-corrected QT interval; ROS: reactive oxygen species; t½: terminal elimination half-life; Tmax: time to reach Cmax. | |
| Alternative names | PXL-008 (development code), EMD 387008 (development code) |
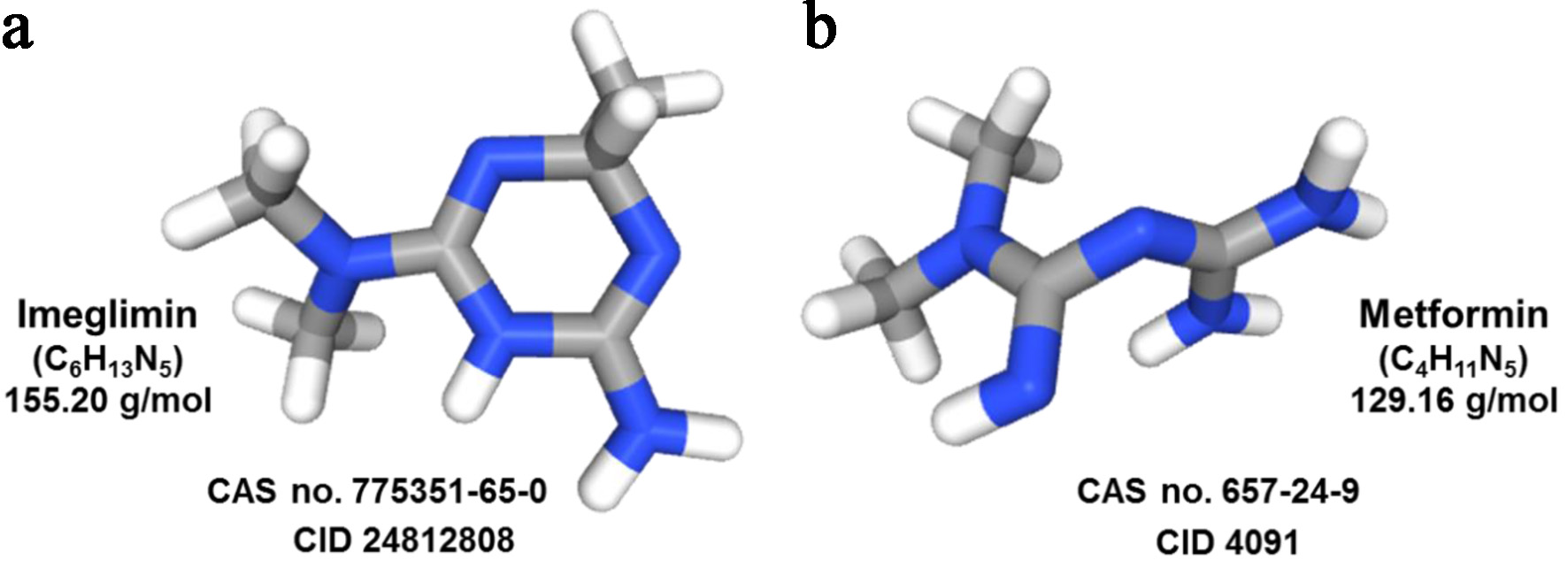
| Chemical class and scaffold | First-in-class oral antidiabetic; a small-molecule containing a tetrahydro-triazin ring; structurally inspired by metformin |
| Molecular formula/molecular weight | C6H13N5 (free base); molecular weight approximately 155.20 g/mol (191.66 g/mol for the marketed hydrochloride salt, C6H14ClN5) |
| Aqueous solubility | Highly soluble in water (> 100 mg/mL at 25 °C), enabling oral dosage-formulation |
| Optimized dose | A dosage of 1,000 mg twice daily balances efficacy and tolerability |
| Administration route | Available in oral tablets; investigated as monotherapy and add-on to metformin, DPP-4 inhibitors, insulin, and other oral hypoglycemic agents |
| Absorption and bioavailability | Rapid absorption (Tmax approximately 1–2 h); absolute oral bioavailability reported to be near 100%. Food has limited impact |
| Elimination half-life | t½ approximately 9–12 h; primarily renal excretion of unchanged drug |
| Drug–drug interactions | No clinically relevant changes in exposure when co-administered with metformin or sitagliptin; cimetidine (MATE1 inhibitor) raises Cmax and AUC by approximately 30% without clinical impact |
| Mitochondrial modulation | Partial complex I inhibition, correction of complex III deficiency, reduction of ROS, increase in NAD+ synthesis and ATP generation, preserving β-cell function |
| Incretin effects | Stimulates endogenous GLP-1 and GIP release, augmenting insulinotropic action |
| β-cell function | A +112% increase in insulin secretory response after 7 days; a 36% increase in β-cell glucose sensitivity |
| Insulin sensitivity | Improves hepatic and skeletal muscle insulin signaling; decreases hepatic glucose output; OGTT-Stumvoll index increases indicating improved insulin sensitivity |
| Monotherapy | Reduces HbA1c by approximately −0.8% to −0.9% compared to placebo at 24 weeks |
| Add-on to metformin | Additional HbA1c drop −0.44% over metformin alone at 12 weeks |
| Add-on to insulin | HbA1c reduction of −0.60% at 16 weeks; sustained through 52 weeks with good tolerance |
| General tolerability | Comparable adverse event rates to placebo; most events are mild gastrointestinal issues, notably diarrhea; minimal risk of hypoglycemia |
| Cardiovascular profile | No QT/QTc prolongation at therapeutic (2,250 mg) or supratherapeutic (6,000 mg) single doses |
| Demographics and disease factors | Better response with older age, therapy-naive status, and lower baseline HbA1c; data-driven clusters reveal heterogeneity |
| 12-month follow-up | Reduction in HbA1c by 0.7% to 0.9% (at 24 weeks) modest improvements in weight and lipids; efficacy independent of age, sex, body mass index, and renal function |
| Author, year [Ref.] | Method | Findings | Conclusion |
|---|---|---|---|
| AE: adverse events; AUC: area under the curve; bid: twice daily; CGM: continuous glucose monitoring; CI: confidence interval; Cmax: maximum plasma concentration; ΔΔQTcF: time-matched, placebo-subtracted, baseline-adjusted QTc interval (Fridericia-corrected); DPP4-I: dipeptidyl Peptidase-4 inhibitor; ECG: electrocardiogram; FPG: fasting plasma glucose; GI: gastrointestinal; GIP: gastric inhibitory peptide; GLP1-RA: glucagon-like peptide-1 receptor agonist; GSIS: glucose-stimulated insulin secretion; HbA1c: glycated hemoglobin; HOMA-β: homeostatic model assessment of β-cell function; HR: heart rate; ISR: insulin secretion rate; LSM: least squares mean; MAGE: mean amplitude of glycemic excursions; MATE: multidrug and toxic extrusion transporter; NNT: number needed to treat; OCT: organic cation transporter; OD: once daily; OGTT: oral glucose tolerance test; OHA: oral hypoglycemic agent; OR: odds ratio; PG: plasma glucose; RCT: randomized, placebo-controlled trial; TIR: total insulin response (incremental area under the curve 0–45 min); TEAE: treatment emergent adverse event; T2DM: type 2 diabetes mellitus. | |||
| Pirags et al, 2012 [17] | In a 4-week, phase IIa, three-arm parallel-group study, participants were randomly assigned to receive either imeglimin 2,000 mg OD, imeglimin 1,000 mg bid, or metformin 850 mg bid. Responses to the OGTT were subsequently evaluated. In an 8-week phase IIa, four-arm controlled multi-center study, patients were randomized to receive imeglimin 500 mg bid, imeglimin 1,500 mg bid, metformin 850 mg bid or placebo. | Imeglimin was as effective as metformin in reducing the AUC (PG) and AUC (0–6 h), FPG, and HbA1c, and exhibited a more favorable tolerability profile compared to metformin. | Compared to metformin, imeglimin displays a superior benefit-to-risk profile among individuals living with T2DM, making this drug suitable for combination therapies with other classes of glucose-lowering agents and for administration to large patient populations. |
| Fouqueray et al, 2013 [10] | A total of 156 participants were randomized in a 1:1 ratio to receive either imeglimin (1,500 mg bid) or placebo, both administered in addition to a fixed dose of metformin (1,500–2,000 mg/day). The primary efficacy endpoint was the change in HbA1c from baseline, while secondary endpoints included FPG and the proinsulin/insulin ratio. | After 12 weeks, the reduction in HbA1c with metformin-imeglimin, compared to placebo, was −0.44% (P < 0.001). Metformin-imeglimin also led to significant improvements in FPG and the proinsulin/insulin ratio from baseline (−0.91 mg/dL and −7.5, respectively) relative to metformin-placebo (0.36 mg/dL and 11.81). The safety profile of metformin-imeglimin therapy was generally comparable to that of metformin-placebo. | The addition of imeglimin to metformin improved glycemic control showing potential as a novel therapeutic option for T2DM. |
| Pacini et al, 2015 [18] | A double-blind, RCT was conducted in 33 individuals living with T2DM, who were either drug-naive or withdrawn from their previous metformin monotherapy for 2 weeks and received imeglimin 1,500 mg bid or placebo for 1 week. GSIS was assessed with hyperglycemic clamp. The primary endpoint was insulin secretion defined by TIR total insulin response (incremental area under the curve (iAUC) 0–45 min) and ISR calculated from C-peptide deconvolution. Moreover, β-cell glucose sensitivity at steady state, hepatic insulin extraction and insulin clearance were also calculated. | A 7-day imeglimin treatment increased the insulin secretory response to glucose by +112%, first-phase ISR by +110% and second-phase ISR by +29%. Imeglimin improved β-cell glucose sensitivity by +36% and tended to decrease hepatic insulin extraction without affecting glucagon secretion. | Among individuals with T2DM, imeglimin improved β-cell function. |
| Fouqueray et al, 2020 [19] | Healthy Caucasian men received either metformin 850 mg bid with placebo (n = 16) or sitagliptin 100 mg OD with placebo (n = 16) on days 1–6, followed by metformin 850 mg bid with imeglimin 1,500 mg bid or sitagliptin 100 mg OD once daily with imeglimin 1,500 mg bid on days 7–12. | Co-administration with imeglimin reduced metformin’s systemic exposure (AUC0-τ) and maximum concentration by 14% and 10%, respectively, and decreased urinary excretion of unchanged metformin from 40% to 34%. The 90% CI showed no significant effect on metformin exposure. Sitagliptin’s AUC0-τ and maximum concentration were unchanged by imeglimin, with median times to maximum concentration of 0.7–1.0 h and mean elimination half-lives of 8.2–8.7 h for both regimens. | Co-administration of imeglimin with either metformin or sitagliptin did not lead to clinically significant alterations in the systemic exposure to metformin or sitagliptin. However, slight decreases in exposure (as measured by AUC0-τ and maximum concentration) and renal elimination were observed when metformin was administered alongside imeglimin compared to placebo. |
| Dubourg et al, 2020 [13] | Healthy participants received single doses of imeglimin 2,250 mg, imeglimin 6,000 mg, moxifloxacin 400 mg, and placebo. Twelve-lead Holter ECGs were continuously recorded from 1 h before dosing through at least 24 h post-dose. | The upper bound of the two-sided 90% CI for ΔΔQTcF did not exceed the regulatory threshold of 10 ms in any of the imeglimin dose groups. There were no QTcF values > 500 ms nor changes from pre-dose in QTcF above 60 ms in the imeglimin groups. Imeglimin did not have any relevant effect on HR and PR or QRS intervals. Assay sensitivity was demonstrated by the effect of moxifloxacin 400 mg, with a lower bound two-sided 90% CI for ΔΔQTcF of 10.6 ms. | Both therapeutic and supratherapeutic doses of imeglimin were not associated with QT/QTc interval prolongation. |
| Chevalier et al, 2020 [20] | A phase 1 study was conducted in 16 individuals who received a single dose of 1,500 mg imeglimin alone on day 1 followed by a 6-day treatment with cimetidine 400 mg bid. On day 8, a single dose of imeglimin was co-administered with cimetidine. | The Cmax and AUC of imeglimin increased in a clinically non-significant manner when combined with cimetidine, mainly due to reduced renal elimination from MATE1 inhibition. | No clinically meaningful drug-drug interactions have been identified between imeglimin and cimetidine, a well-established inhibitor of the MATE1, MATE2-K, OCT1, and OCT2 transporters. |
| Chevalier et al, 2021 [21] | A single-dose, open-label, parallel-group study was conducted in seven participants with normal hepatic function and seven participants with moderate hepatic impairment, each of whom received a single 1,000 mg dose of imeglimin. | Subjects with moderate hepatic impairment showed a 1.3-fold higher Cmax and 1.5-fold higher AUC for imeglimin than those with normal liver function, but this was not clinically significant. The increased plasma levels and renal excretion, with unchanged elimination rate, suggest greater oral absorption and/or reduced hepatic uptake in these individuals. | Imeglimin demonstrated a favorable safety profile and was well tolerated by all participants. |
| Dubourg et al, 2021 [22] | Treatment-naive adults or individuals who had previously received a single oral antidiabetic agent were randomized to receive imeglimin orally at doses of 500 mg, 1,000 mg, or 1,500 mg, or matched placebo, administered bid for 24 weeks. The primary endpoint was the change in HbA1c from baseline to week 24, adjusted for placebo. | At week 24, imeglimin significantly decreased HbA1c. Treatment-emergent AEs were registered for 68.0%, 62.2%, 73.3%, and 68.0% of patients receiving imeglimin 500, 1,000 or 1,500 mg and placebo, respectively. A small increase in GI adverse effects occurred with the 1,500 mg dose while hypoglycemia was balanced among groups. | In Japanese patients with T2DM, imeglimin monotherapy demonstrated favorable tolerability and produced significant improvements in glycemic control without increasing the incidence of hypoglycemic events compared to placebo. |
| Dubourg et al, 2021 [23] | This phase 3, double-blind, RCT was conducted at 30 sites in Japan with adults living with T2DM stable on diet and exercise. Participants were randomized to receive either imeglimin (1,000 mg bid n = 106) or placebo (n = 107) for 24 weeks. The study’s primary endpoint was mean HbA1c change from baseline to week 24; the key secondary endpoint was responder rates at week 24. | Compared to placebo, the adjusted mean difference in change from baseline HbA1c at week 24 was −0.87% (95% CI −1.04 to −0.69 (−9.5 mmol/mol; 95% CI −11.4 to −7.5); P < 0.0001). Forty-seven (44.3%) individuals reported ≥ 1 AE in the imeglimin group versus 48 adverse events (44.9%) in the placebo group. | Imeglimin demonstrated a significant improvement in HbA1c levels among Japanese patients with T2DM compared to placebo, while exhibiting a safety profile comparable to that of placebo. |
| Dubourg et al, 2022 [24] | TIMES 2 was a phase 3, open-label trial of T2DM patients inadequately controlled by diet/exercise or single-agent antidiabetic therapy. A total of 714 participants received imeglimin 1,000 mg bid for 52 weeks, either alone (n = 134), or with other therapies α-glucosidase inhibitor (n = 64), biguanide (n = 64), dipeptidyl peptidase-4 inhibitor (DPP4-I; n = 63), glinide (n = 64), glucagon-like peptide-1 receptor agonist (GLP1-RA; n = 70), sodium-glucose co-transporter-2 inhibitor (n = 63), sulphonylurea (n = 127), or thiazolidinedione (n = 65). The primary endpoint was safety; secondary endpoints included changes in HbA1c and FPG at week 52. | Of the patients, 75.5% experienced at least one TEAE, mostly mild or moderate; serious TEAEs (unrelated to the drug) occurred in 5.6%. No notable changes were seen in ECG, vital signs, physical exams, or labs. After 52 weeks, HbA1c dropped by 0.46% with imeglimin alone, 0.56–0.92% with oral combinations, and 0.12% with injectable GLP1-RA. The largest reduction (0.92%) was seen in those taking imeglimin plus DPP4-I. | Imeglimin demonstrates sustained safety and efficacy, with good tolerability, when used as monotherapy or in combination with oral agents in Japanese patients with T2DM. |
| Reilhac et al, 2022 [25] | This double-blind, randomized phase 3 trial enrolled adults living with T2DM poorly controlled by insulin. Participants received either 1,000 mg bid imeglimin (n = 108) or placebo (n = 107) alongside insulin for 16 weeks, followed by a 36-week open-label extension where all received imeglimin. The primary outcome was the change in mean HbA1c from baseline to week 16. | At week 16, imeglimin reduced HbA1c by an adjusted mean of −0.60% (95% CI: −0.80 to −0.40; P < 0.0001) compared to placebo, a decrease maintained at 52 weeks (−0.64%, 95% CI: −0.82 to −0.46). Rates of AEs, serious adverse events, and hypoglycemia were similar across groups, with all hypoglycemic episodes in the imeglimin group being mild and requiring no assistance. | Imeglimin demonstrated significant improvement in HbA1c levels among Japanese patients with T2DM who were inadequately controlled by insulin therapy, while exhibiting a safety profile comparable to placebo. The efficacy of imeglimin as an adjunct to insulin was sustained over a 52-week period. |
| Theurey et al, 2022 [26] | This trial involved 59 T2DM subjects previously on stable metformin, who were washed out for 4 weeks and then randomized to 1,500 mg bid imeglimin (n = 30) or placebo (n = 29). The primary endpoint was change in AUC during OGTT from baseline to week 18. Secondary endpoints included glycemic control and insulin secretion and sensitivity indices. | The study achieved its primary endpoint, with imeglimin showing a significant reduction in AUC glucose compared to placebo (−429.6 mmol/L/min, P = 0.001). FPG dropped by −1.22 mmol/L (P = 0.022) and HbA1c by −0.62% (P = 0.013). Imeglimin was well tolerated, with fewer adverse events (26.7%) than placebo (58.6%). | The findings align with a mechanism of action that encompasses both enhanced insulin secretion and improved insulin sensitivity, providing additional evidence for the potential of imeglimin to advance healthcare outcomes in patients with T2DM. |
| Hagi et al, 2023 [27] | Data were integrated from two 24-week RCTs conducted in adults with T2DM. Outcomes—including LSM change in HbA1c from baseline to week 24 as well as safety assessments—were evaluated across subgroups defined by demographics, clinical characteristics, and comorbidities. | Imeglimin showed a statistically significant reduction in HbA1c compared to placebo across all patient subgroups (P < 0.05), including various demographic, clinical, and comorbidity factors. This difference was evident from week 4 and persisted through week 24. No new safety issues were detected in any subgroup. | The effectiveness and safety profile of imeglimin were established across diverse patient populations, regardless of initial demographic or clinical parameters. |
| Takahashi et al, 2024 [28] | In this prospective observational study, 16 adults hospitalized for T2DM and treated with metformin (500–1,000 mg/day) wore a CGM sensor for up to 14 days. After initial monitoring on their usual therapy (period 1), participants either switched to imeglimin (2,000 mg/day; I/M group) or had their metformin dose increased (M/I group) and were monitored again (period 2). Following a treatment switch-returning to metformin or adding imeglimin after a washout period-blood glucose was tracked for at least two additional days (period 3). | Three participants withdrew from the CGM analysis (one due to gastrointestinal symptoms during metformin escalation), leaving 13 for the final review. Only metformin and imeglimin doses were adjusted. Each evaluation lasted 48 h. Most subjects doubled their metformin to an average of 1,153.8 ± 315.2 mg/day. Mean blood glucose dropped similarly with both imeglimin add-on and metformin escalation, but imeglimin more effectively reduced MAGE and other glycemic variability measures, with unchanged time in-range. Imeglimin also better controlled post-meal glucose spikes after breakfast and dinner. No severe hypoglycemia occurred. | Imeglimin effectively corrects glycemic variability thereby contributing to improving T2DM pathophysiology. |
| Hagi et al, 2024 [29] | Data from two 24-week RCTs of imeglimin monotherapy in Japanese adults with T2DM were analyzed. The study assessed proportions of responders and sustained responders at each visit for the 1,000 mg bid group, used multivariate logistic regression to identify factors linked to response, and conducted subgroup analyses comparing early vs. non-early responders based on HbA1c change at week 4. | A total of 38.0% of imeglimin-treated patients and 7.2% of placebo-treated patients were responders (P < 0.001, NNT = 4). The proportion of sustained responders at weeks 4, 8, 12, 16 and 20 was 10.6%, 19.0%, 24.0%, 25.7%, and 29.1%, respectively (> 70% of responders at each visit). Improvements in HbA1c and FPG were significantly greater in early responders versus early non-responders from week 4; between-group differences remained significant to week 24. Older age (OR 1.09, 95% CI 1.04–1.14; P < 0.001); treatment-naive status vs. previous treatment (OR 3.70, 95% CI 1.55–8.82; P = 0.003), and lower baseline HbA1c (OR 0.06, 95% CI 0.02–0.16; P < 0.001) predicted response. | A significantly higher percentage of patients receiving imeglimin 1,000 mg twice daily as monotherapy achieved responder status compared to those administered placebo. Over 70% of participants demonstrated a sustained response, suggesting reliable treatment outcomes. Advanced age, no previous therapy, and early response independently predicted imeglimin’s effectiveness. |
| Katsuyama et al, 2024 [30] | A retrospective chart review of 68 patients collected metabolic data at imeglimin initiation and at 3, 6, and 12 months. | HbA1c levels decreased by 0.7% at 3 months, 1.1% at 6 months, and 1.0% at 12 months following the initiation of imeglimin therapy irrespective of age, sex, BMI, T2DM duration, renal function, or concurrent use of other hypoglycemic agents. Additionally, significant decreases in body weight, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and non-HDL-C were noted during imeglimin treatment. | This real-world setting study demonstrates the long-term effects of imeglimin confirming its glucose-lowering efficacy and suggesting favorable effects of imeglimin on body weight and serum lipid profile. |
| Hagi et al, 2024 [31] | A data-driven cluster analysis using non-hierarchical k-means methodology was conducted on randomized, double-blind trials of imeglimin monotherapy and adjunctive therapy with insulin. The analysis was based on four baseline variables: 1) disease duration; 2) BMI; 3) HbA1c; and either 4a) HOMA-β for monotherapy trials or 4b) total daily insulin dose for adjunctive trials. | Four clusters with distinct clinical profiles were found in both monotherapy (1–4) and adjunctive therapy (I–IV) trials. Clusters 1/I had lower index values, 2/II had longer diabetes duration, 3/III showed higher baseline BMI and insulin markers, and 4/IV had higher baseline HbA1c. Changes in HbA1c and effect sizes at week 24 differed by cluster, with cluster 4 showing the greatest reduction. In imeglimin adjunctive therapy, all clusters except III experienced significant HbA1c improvements versus placebo at week 16. | Variations in imeglimin response were identified across clusters of patients with T2DM. Stratifying patients may facilitate the identification of individuals most likely to benefit from imeglimin therapy. |
| Usui et al, 2025 [32] | A single-center, open-label randomized trial enrolled patients with T2DM who were drug-naive or using one OHA, which was discontinued for 8 weeks before randomization. Participants received either imeglimin (2,000 mg/day) or metformin (1,000 mg/day), and OGTTs were conducted before treatment and at 12 and 24 weeks. | At 24 weeks, HbA1c reduction was similar in imeglimin and metformin groups. Both showed decreased post-challenge glucose, but insulin rose only in imeglimin. GLP-1 levels increased in both groups; GIP levels rose only in imeglimin. In imeglimin, the insulin increase correlated with higher GLP-1 at 12 weeks and with higher GIP at 24 weeks. | In contrast to metformin, imeglimin not only enhances GLP-1 secretion but also increases GIP secretion, alongside its direct insulinotropic effects on glucose regulation. |
| Ito et al, 2025 [33] | Data were obtained from the 52-week, open-label, phase 3 TIMES-2 study conducted in Japanese patients with T2DM. Participants receiving both imeglimin and metformin were stratified into two subgroups according to the presence of gastrointestinal symptoms and diarrhea and were evaluated for efficacy and safety outcomes. | Among 64 patients, 40.6% had GI symptoms and 17.2% experienced diarrhea, mostly within the first 4 months of treatment. Metformin dose and age were not linked to these events, which were usually mild and half resolved within a week. Diarrhea was significantly associated with diabetes duration under 5 years (OR 5.979; P = 0.039). Significant hypoglycemic effects were seen regardless of GI symptoms or diarrhea, though HbA1c improvement was greater in those with these symptoms. | It is important to maintain heightened awareness of the potential for gastrointestinal symptoms, such as diarrhea, during combination therapy with imeglimin and metformin. |
| Takahashi et al, 2025 [34] | In this multicenter, open-labelled, prospective, randomized, parallel-group comparison study, the addition of imeglimin (2,000 mg/day) or metformin escalation was applied for 24 weeks in 73 eligible subjects of whom 65 participants comprised the full analysis set. The primary endpoint was the mean change in HbA1c over 24 weeks. As the secondary endpoints, the occurrence of adverse events, changes in metabolic parameters, biomarkers and factors associated with HbA1c improvement were analyzed. | After 24 weeks, imeglimin (n = 33) showed a greater reduction in HbA1c than metformin dose escalation (n = 32), with a change difference of −0.21% (95% CI: −0.41%, −0.01%) (P = 0.038). Seven participants discontinued imeglimin due to serious gastrointestinal adverse events. Imeglimin also significantly reduced body weight and liver enzyme levels, and improvements in HbA1c were correlated with fatty liver disease indicators. | The combination of imeglimin with a DPP-4i and low-dose metformin demonstrated greater improvement in HbA1c levels compared to escalating the dose of metformin alone. |
| Kaku et al, 2025 [35] | FAMILIAR is a multicenter, randomized, double-blind trial assessing the efficacy and safety of imeglimin versus placebo in 117 Japanese adults with T2DM not adequately controlled by DPP-4i monotherapy, alongside diet and exercise. Participants received oral imeglimin 1,000 mg or placebo twice daily for 24 weeks, then continued imeglimin for 80 weeks. The main outcome was change in HbA1c at week 24, with ongoing safety monitoring. | A total of 117 patients were randomized to imeglimin (n = 58), placebo (n = 54) or were excluded (n = 5). The LSM (standard error) changes in HbA1c level (baseline to Week 24) for the imeglimin and placebo groups, respectively, were −0.65% (0.11%) and 0.38% (0.11%) in the overall population (group-difference −1.02% (95% CI −1.33%, −0.72%); P < 0.001); −0.47% (0.17%) and 0.32% (0.18%) in patients aged < 65 years (−0.79% (−1.29%, −0.29%); P = 0.003); and −0.80% (0.14%) and 0.42% (0.14%) among subjects aged ≥ 65 years (−1.22% (−1.61%, −0.82%); P < 0.001). One patient in the imeglimin group had mild hypoglycemia; the safety profile was favorable. | Imeglimin represents a potential new treatment option for patients with T2DM and inadequate glycemic control with DPP-4i including those aged 65 years or older. |
| Author, year [Ref.] | Method | Findings | Conclusions |
|---|---|---|---|
| AE: adverse event; bid: twice daily; BMI: body mass index; CKD: chronic kidney disease; CI: confidence interval; FPG: fasting plasma glucose; HbA1c: glycated hemoglobin; HDL-C: high density lipoprotein cholesterol; HOMA-IR: homeostasis model of insulin resistance; LDL-C: low density lipoprotein cholesterol; RCTs: randomized controlled trials; SMD: standardized mean difference; T2DM: type 2 diabetes mellitus. | |||
| Abdelhaleem et al, 2021 [36] | Eight studies comprising 1,555 T2DM patients were included. | The imeglimin group demonstrated statistically significant superiority over the control group in terms of HbA1c and FPG (P < 0.00001). However, no significant effects were observed on HOMA-IR or lipid parameters, including triglycerides, LDL-C, and HDL-C (all P > 0.05). Regarding safety, imeglimin was well tolerated and did not result in any treatment-emergent or serious AEs. | Imeglimin improved glycemic control by lowering HbA1c and FPG, but did not significantly affect HOMA-IR or lipid levels. |
| Singh et al, 2023 [37] | Among the seven phase 2 studies and three phase 3 studies available at the time of this meta-analysis, only three published double-blind RCTs evaluated the efficacy and safety of imeglimin 1,000 mg bid compared to placebo. | Employing the random-effects model of two monotherapy studies (n=360) this metanalysis found that imeglimin 1000 mg twice daily significantly reduced HbA1c levels (Δ −0.9%, 95% CI: −1.1 to −0.74%; P < 0.0001) compared to placebo, with no observed heterogeneity (I2 = 0%). Furthermore, a pooled meta-analysis of all three RCTs (n = 574) indicated a significant reduction in HbA1c with imeglimin 1000 mg twice daily (Δ −0.79%; 95% CI: −1.00 to −0.59%; P < 0.0001) relative to placebo, although high heterogeneity was present. | This meta-analysis showed that imeglimin leads to a statistically significant reduction in HbA1c levels among individuals with T2DM, while maintaining an acceptable tolerability profile. |
| Hagi et al, 2023 [16] | Nine RCTs, with a total of 1,655 subjects, were included. | Dose-specific analysis of data showed that imeglimin monotherapy significantly reduced HbA1c at doses > 1,000 mg bid compared to placebo. As adjunct therapy, imeglimin also improved HbA1c at 1,000 and 1,500 mg. Subgroup analyses found efficacy regardless of CKD status, with greater effects seen in Japanese patients and those with lower BMI. Imeglimin did not differ from placebo in all-cause discontinuation rates or AE incidence. | Imeglimin 1,000 mg administered bid may offer optimal therapeutic benefits for glycemic control while maintaining a favorable safety profile. |
| Teawri et al, 2025 [38] | Thirteen RCTs and nine observational studies were included in the quantitative and qualitative analyses, respectively. | Imeglimin reduced HbA1c and FPG in a dose-dependent and combination-dependent manner, with higher doses and combination therapy showing greater benefits. It improved β-cell function without decreasing insulin resistance, and no major adverse events occurred. | Imeglimin has shown efficacy and safety in managing T2DM, particularly in enhancing glycemic control and β-cell function. Small sample sizes and varied results indicate that larger, more rigorous clinical trials are necessary. |
| Song et al, 2025 [5] | A total of 12 RCTs assessing the impact of imeglimin on metabolic parameters were included. | Imeglimin significantly lowered FPG (SMD: −0.51) and HbA1c (SMD: −0.45), and improved HOMA-β (SMD: 0.59). It had no significant effect on insulin, HOMA-IR, or C-peptide levels (P > 0.05). However, imeglimin was associated with an increase in LDL (SMD: 0.32). | Imeglimin has shown efficacy and a favorable safety profile in managing T2DM, especially in terms of glycemic control. |