Figures
↓ Figure 1. The diagram depicts the balance between the classical ACE/Ang II/AT1R axis and the protective ACE2/Ang 1-7/MAS axis of the renin-angiotensin system (RAS). Ang II promotes oxidative stress, inflammation, impaired insulin signaling, and β-cell dysfunction, thereby driving progression to T2DM, whereas ACE2/Ang 1-7/MAS counteract these effects and preserve metabolic homeostasis.
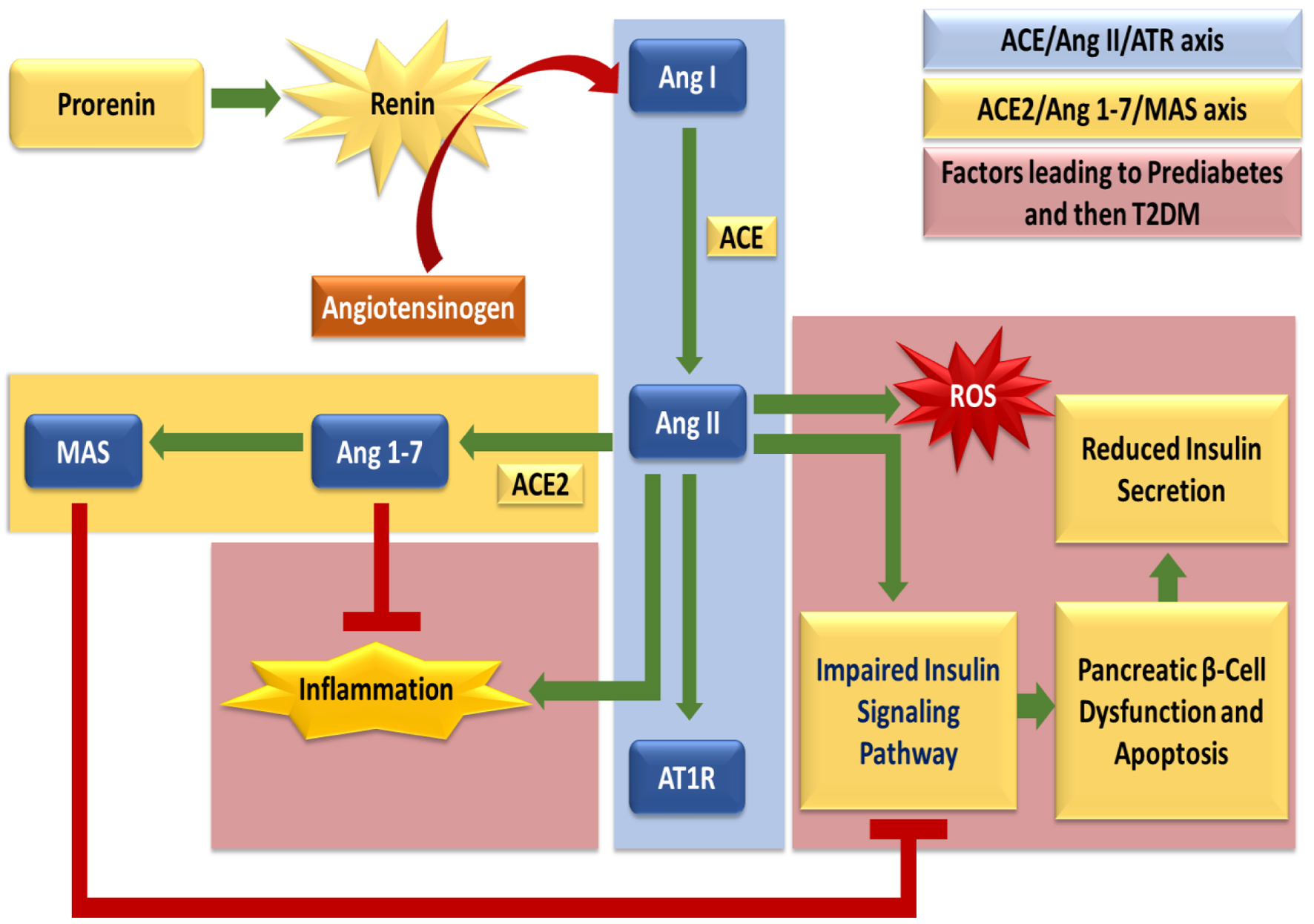
↓ Figure 2. The diagram depicts the role of the classical RAS in the progression from prediabetes to T2DM. Upregulation of renin, ACE, Ang II, and AT1R enhances oxidative stress via NOX activation and promotes inflammation and insulin resistance. Reduced ACE2 activity weakens protective Ang 1-7 signaling, worsening glucose dysregulation. Persistent metabolic imbalance increases the risk of T2DM and complications, including nephropathy, cardiomyopathy, retinopathy, and cardiac remodeling, driven by dominance of the ACE/Ang II/AT1R axis.
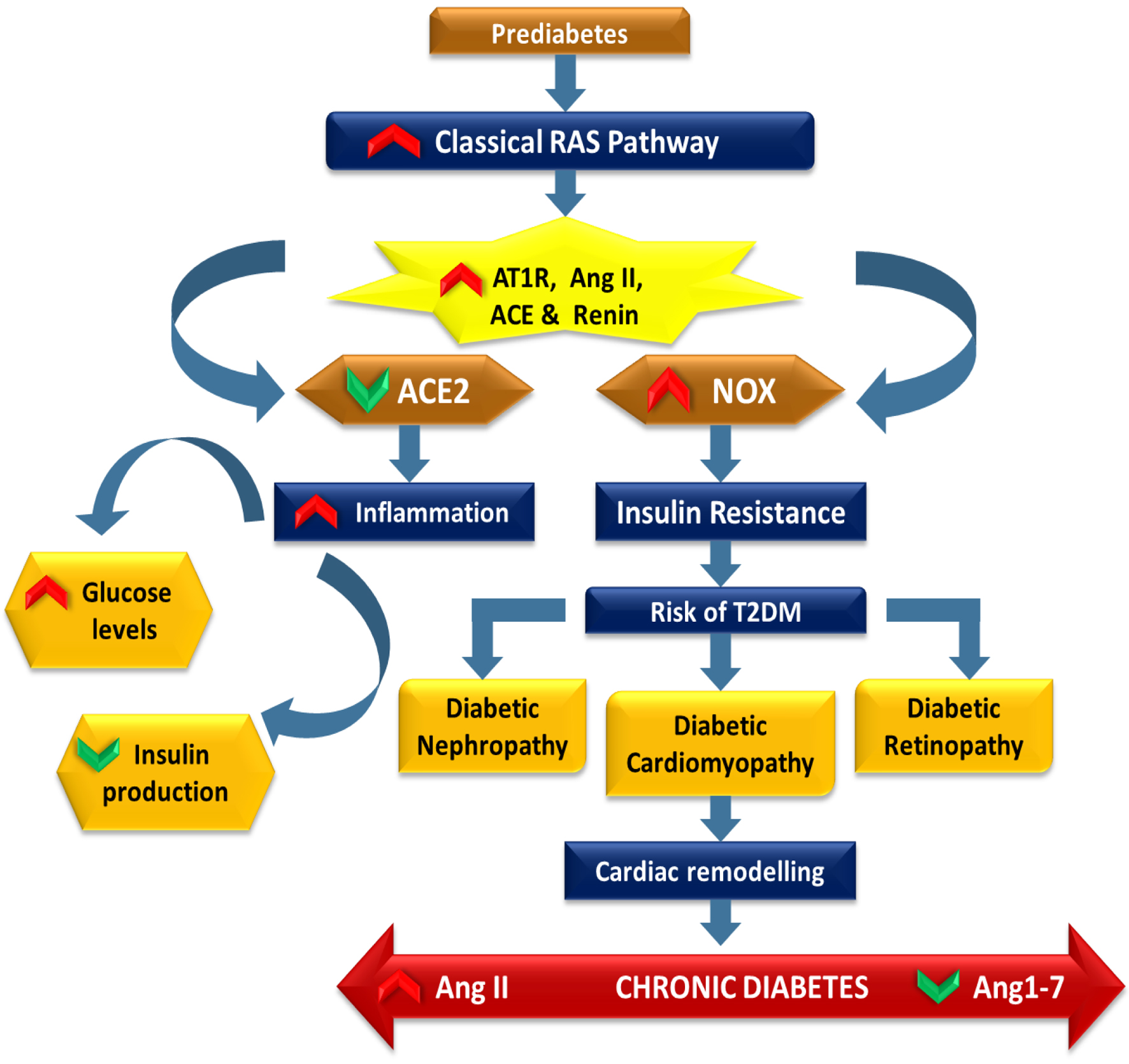
↓ Figure 3. This schematic depicts progressive antioxidant pathway dysregulation from prediabetes to T2DM. In prediabetes, reduced dietary antioxidants and hyperglycemia impair SOD, GPx, and CAT activity, promoting β-cell dysfunction, reduced insulin secretion, and apoptosis. In diabetes, persistent hyperglycemia disrupts NOX, SODs, CAT, GSTs, and eNOS, increasing oxidative stress and free radical generation. This activates AGEs and the polyol pathway, contributing to diabetic retinopathy, diabetic nephropathy, and cardiovascular complications.
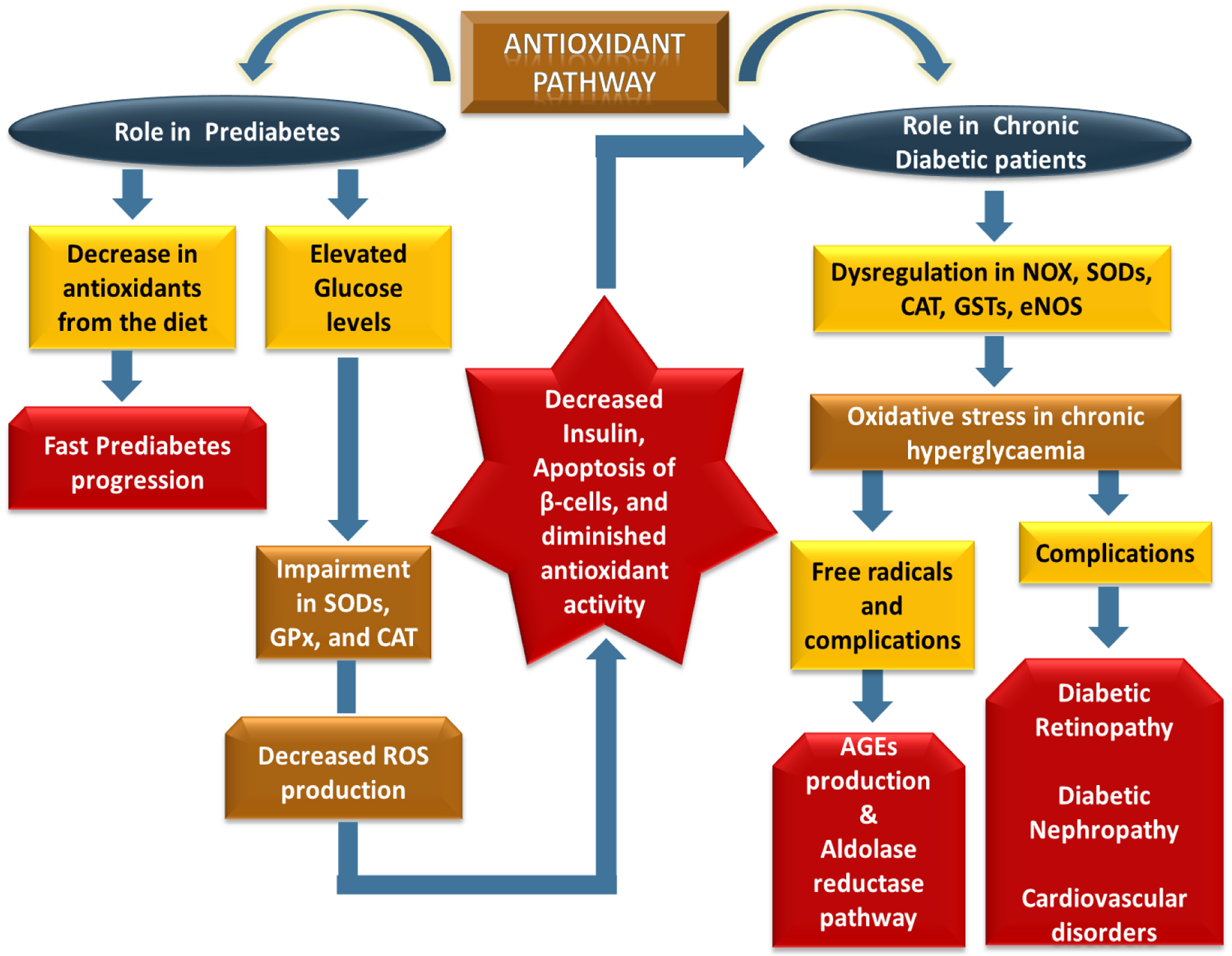
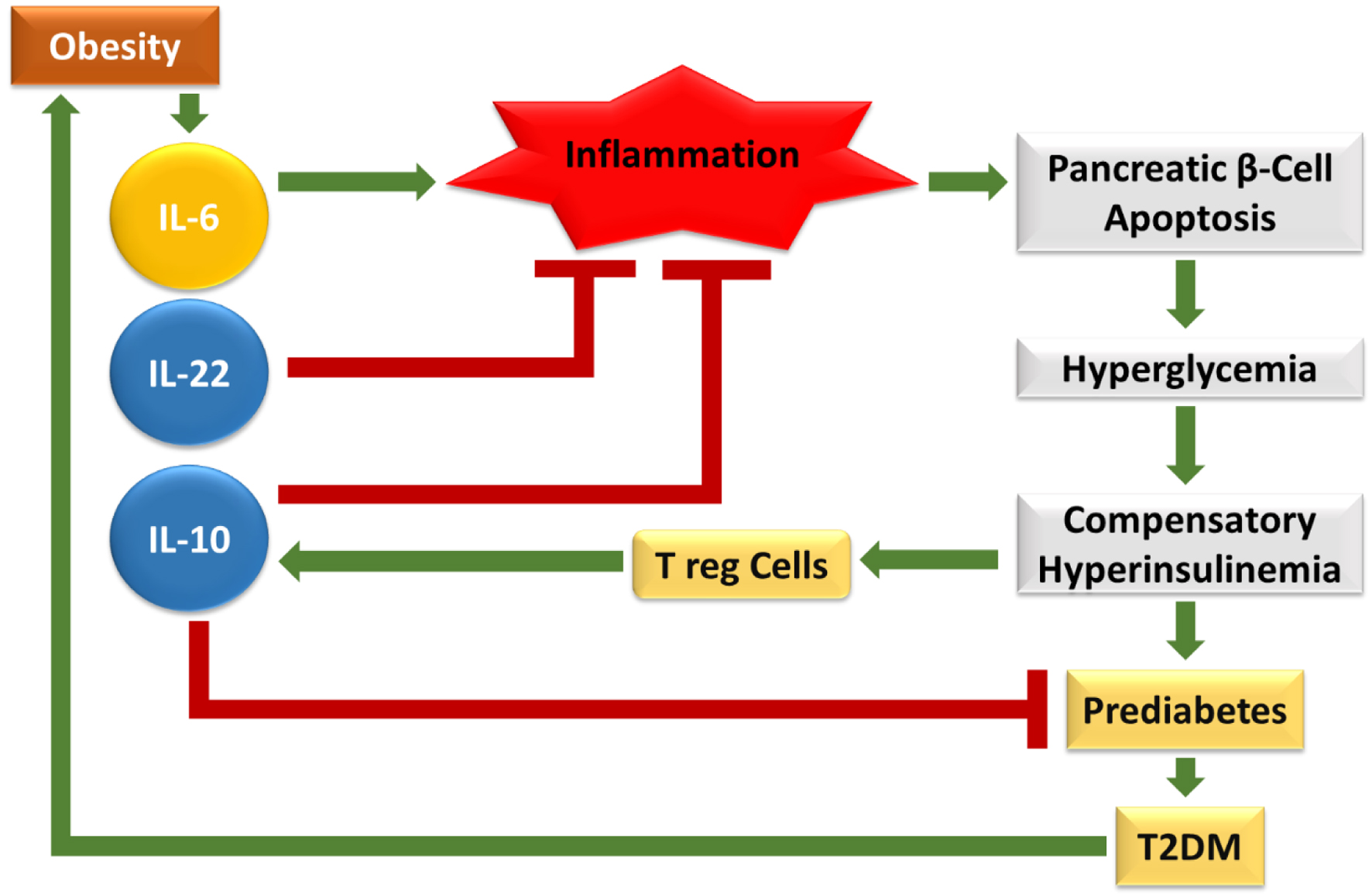
↓ Figure 4. The given figure depicts how obesity-induced cytokine imbalance drives T2DM progression. Elevated IL-6 promotes chronic inflammation, leading to pancreatic β-cell apoptosis, hyperglycemia, compensatory hyperinsulinemia, prediabetes, and ultimately T2DM. Anti-inflammatory cytokines (IL-10, IL-22) and Treg cells counteract inflammation and protect β-cells. However, in obesity, pro-inflammatory dominance sustains immune dysregulation, accelerating β-cell dysfunction and metabolic deterioration.
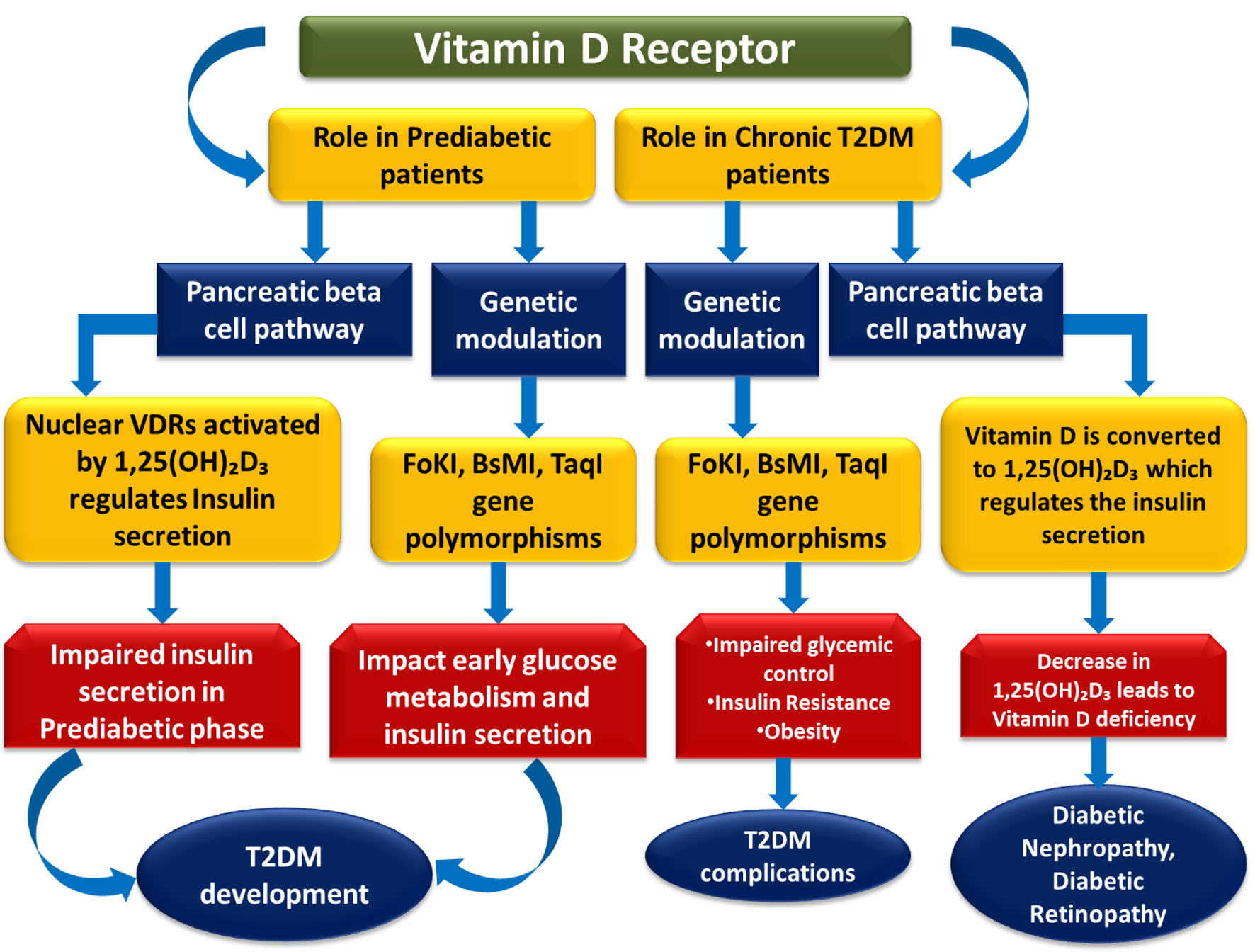
↓ Figure 5. This figure depicts the role of VDR signaling in the progression from prediabetes to T2DM. Activation of nuclear VDRs by 1,25(OH)2D3 regulates pancreatic β-cell function and insulin secretion, while VDR polymorphisms (FokI, BsmI, TaqI) influence glucose metabolism. Impaired signaling contributes to insulin deficiency, insulin resistance, poor glycemic control, and vitamin D deficiency, ultimately leading to T2DM development and complications such as nephropathy and retinopathy.
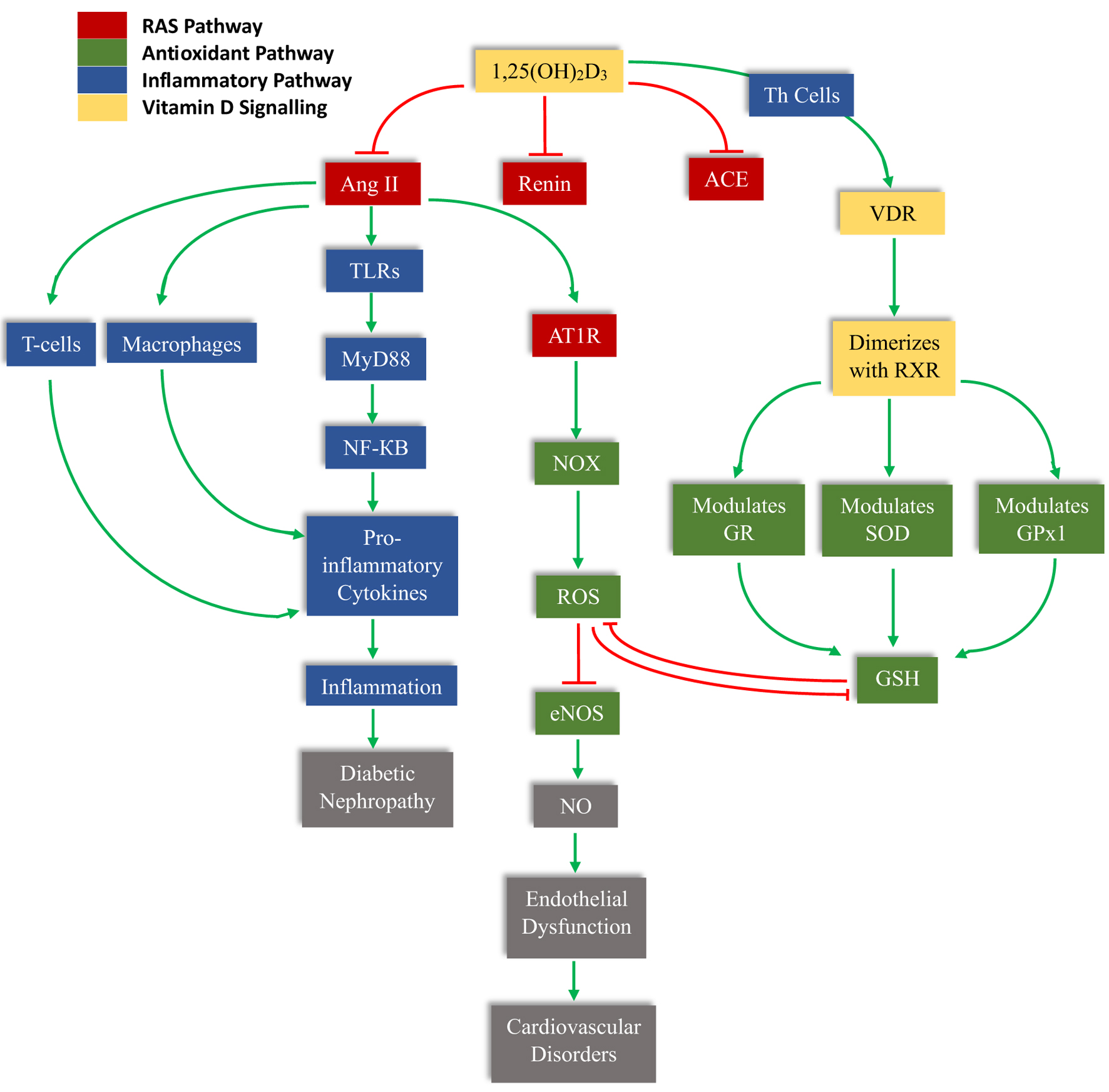
↓ Figure 6. The schematic illustrates the central role of 1,25(OH)2D3 (calcitriol) in modulating RAS activation, immune signaling, oxidative stress, and antioxidant defense. Calcitriol suppresses renin and ACE expression, thereby reducing Ang II formation. Ang II activates AT1R, stimulating NADPH oxidase (NOX)–mediated ROS generation. Excess ROS decreases eNOS activity and nitric oxide (NO) bioavailability, leading to endothelial dysfunction and subsequent cardiovascular disorders. Ang II also activates TLRs/MyD88/NF-κB signaling in T cells and macrophages, enhancing pro-inflammatory cytokine production, inflammation, and progression to diabetic nephropathy. Concurrently, calcitriol binds to the VDR, which dimerizes with RXR and transcriptionally regulates antioxidant enzymes, including GR, SOD, and GPx1, thereby restoring glutathione (GSH) levels and attenuating ROS. Green arrows indicate activation or stimulation, while red blunt arrows represent inhibition.
Table
↓ Table 1. Summary of Key Pathways, Genes, and Pathophysiological Implications in T2DM
| Pathway | Key genes/molecules | Pathophysiological impact | Therapeutic potential |
|---|
| RAS | ACE, AGT, AGTR1 | Increases insulin resistance; promotes fibrosis and vasoconstriction. | ACE inhibitors and ARBs for renal and cardiac protection. |
| Oxidative stress | NOX, SOD, GPx, Nrf2 | Causes DNA damage and β-cell apoptosis; impairs mitochondrial function. | Antioxidant supplementation; Nrf2 activators. |
| Inflammatory | TNF-α, IL-6, IL-1β, NF-κB | Triggers systemic insulin resistance; promotes “cytokine storm” in complications. | Anti-inflammatory agents; IL-1-β antagonists. |
| Vitamin D (VDR) | VDR, CYP27B1, RXR | Regulates insulin secretion; inhibits RAS and inflammation. | Vitamin D supplementation; VDR agonists. |





