| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 16, Number 1, February 2026, pages 22-28
Relationship Between Subclinical Hypothyroidism and Cardio-Ankle Vascular Index: A Cross-Sectional Study
Qiang Weia, Tian Tian Caib, Xiao Ya Zhengb, c
aPrevention of Disease Department, Jiulongpo Hospital, Chongqing University of Chinese
Medicine, Chongqing, China
bDepartment of Endocrinology, the First Affiliated
Hospital of Chongqing Medical University, Chongqing, China
cCorresponding
Author: Xiao Ya Zheng, Department of Endocrinology, the First Affiliated Hospital of Chongqing
Medical University, Chongqing 400016, China
Manuscript submitted September 28, 2025, accepted January 3, 2026, published online February 25,
2026
Short title: Relationship Between SCH and CAVI
doi:
https://doi.org/10.14740/jem1570
| Abstract | ▴Top |
Background: The established detrimental effects of subclinical hypothyroidism (SCH) on the cardiovascular system have been well-documented; however, the association between SCH and cardio-ankle vascular index (CAVI) remains inconclusive.
Methods: A retrospective, cross-sectional study was conducted to assess the association between SCH and CAVI. CAVI measurements were obtained using the VaSera VS-1000 vascular screening system. Multivariate logistic regression analysis was conducted to identify independent predictors of elevated CAVI.
Results: The study included 247 subjects diagnosed with SCH and 1,749 age- and gender-matched control group. Individuals with SCH demonstrated lower basal metabolic rates, higher body weight and fat levels, as well as increased blood lipids and high-sensitivity C-reactive protein (hsCRP) levels. Additionally, individuals with SCH showed elevated CAVI values. CAVI showed a positive correlation with higher levels of thyroid-stimulating hormone (TSH) levels and a negative correlation with free triiodothyronine (FT3) levels, with no statistically significant correlation to free thyroxine (FT4) levels. Logistic regression analysis identified advanced age and hypertension as major independent risk factors associated with elevated CAVI. SCH and overweight were identified as independent risk factors for elevated CAVI. Furthermore, hyperlipidemia characterized by high total cholesterol (TC), triglycerides (TG), low-density lipoprotein (LDL), and apolipoprotein B (apoB) is also correlated with an elevated risk of CAVI.
Conclusions: This study offers epidemiological evidence supporting a correlation between elevated TSH and an increased risk of high CAVI in individuals with SCH. Individuals with SCH and elevated TSH levels appear to be at increased risk of arterial stiffness, suggesting a potential link between thyroid function and early vascular alterations.
Keywords: Subclinical hypothyroidism; Cardio-ankle vascular index; Thyroid-stimulating hormone; Cardiovascular disease
| Introduction | ▴Top |
Subclinical hypothyroidism (SCH) is defined by elevated serum thyroid-stimulating hormone (TSH) concentrations in the presence of normal free thyroxine (FT4) levels [1]. With a reported prevalence ranging from 3% to 15% in studied populations [2–4], this condition is particularly prevalent among women and older adults [2]. The primary etiology is autoimmune thyroiditis. As SCH is typically asymptomatic, its identification depends entirely on biochemical laboratory testing [5].
The cardiovascular implications of SCH have become a topic of interest due to the known influence of thyroid hormones on cardiac function and vasculature [6–8]. Substantial evidence suggests a correlation between SCH and elevated cardiac risks, such as alterations in blood pressure and cholesterol levels [9–12]. However, the precise relationship between SCH and the incidence of cardiovascular diseases remains uncertain [13, 14].
Arterial stiffness is linked to cardiovascular disease progression [15, 16]. A novel arterial stiffness parameter known as the cardio-ankle vascular index (CAVI), which is independent of blood pressure, has been developed and implemented globally [17–19]. CAVI can be calculated with application of Bramwell-Hill’s equation [20]. The current instrumentation employed for the evaluation of CAVI, ankle-brachial index (ABI), and brachial-ankle pulse wave velocity (baPWV) is capable of obtaining all three measurements using a single device [18, 21]. CAVI has been extensively utilized in numerous clinical studies, specifically those focused on evaluating arterial stiffness in patients diagnosed with cardiovascular diseases and individuals at elevated risk for developing such conditions [22]. CAVI is considered better than baPWV and ABI because it correlates strongly with left ventricular function and serum lipid profile [23].
The objective of the current research is to compare the prevalence of elevated CAVI in patients with and without SCH in China.
| Materials and Methods | ▴Top |
Subjects
We enrolled adults over 18 years of age who received a health check-up at our hospital during the period spanning September 2019 to September 2021. The analysis focused on participants with complete anthropometric, biochemical, and thyroid function data, as well as CAVI test results. Individuals with a history of thyroid disease (including hyperthyroidism, clinical hypothyroidism, or thyroid hormone therapy) were excluded. Following this exclusion and ethical approval from Chongqing Medical University, the study yielded a final cohort of 1,996 participants (846 men and 1,140 women) for analysis. Due to the retrospective design of the study, informed consent was waived. This study was reviewed and approved by the Institutional Review Board (IRB) of the First Affiliated Hospital of Chongqing Medical University. This study was conducted in compliance with the ethical standards of the First Affiliated Hospital of Chongqing Medical University on human subjects, as well as with the principles of the Declaration of Helsinki.
Anthropometric and vascular sclerosis indicators
Height, weight, waist circumference, and blood pressure were measured as part of the anthropometric assessment, with specific methods and body mass index (BMI) calculation detailed in prior work [24]. A BoCA X1 body composition analyzer employing bioelectrical impedance was used to quantify fat percentage and fat weight. Vascular function was evaluated using a VaSera VS-1000 system (Fukuda Denshi, Tokyo, Japan) to record CAVI, ABI, and baPWV, following standardized protocols [24].
Laboratory test
Blood biochemical analyses included total cholesterol (TC), triglycerides (TG), low-density lipoprotein (LDL), high-density lipoprotein (HDL), apolipoprotein A (apoA), apolipoprotein B (apoB), fasting plasma glucose (FPG), and high-sensitivity C-reactive protein (hsCRP). These indicators were measured by an immunochemical-automated analyzer (Type 7600, Hitachi Ltd., Japan).
The serum levels of free triiodothyronine (FT3), free thyroxine (FT4), and ultrasensitive thyroid-stimulating hormone (uTSH) were detected by an immunochemical-automated analyzer (Beckman Coulter UniCelDxI 800, USA) as previously described [25]. The reference ranges used for diagnosing normal thyroid function were: uTSH (0.56–5.91 µIU/mL), FT3 (2.01–4.82 pg/mL), and FT4 (0.59–1.25 ng/dL).
Definitions
The following operational definitions and clinical thresholds were used in this study, consistent with cited references [18, 21–23, 26–28]: SCH: TSH > 5.91 µIU/mL with normal FT3 and FT4. Elevated vascular indices: CAVI ≥ 9 m/s, ABI ≥ 1.3, baPWV ≥ 1,800 cm/s. Older age: age ≥ 65 years. Overweight: body mass index (BMI) ≥ 25 kg/m2. Hypertension: systolic blood pressure (SBP) ≥ 140 mm Hg or diastolic blood pressure (DBP) ≥ 90 mm Hg. Dyslipidemia parameters: high TC: ≥ 5.6 mmol/L; high LDL: ≥ 2.6 mmol/L; high TG: ≥ 1.7 mmol/L; low HDL: < 0.90 mmol/L (men) or < 1.00 mmol/L (women); high apoB: ≥ 1.2 g/L.
Statistical analyses
Statistical analysis was conducted using GraphPad Prism 5.0. Normally distributed continuous variables are presented as mean ± standard deviation (mean ± SD) and were analyzed by Student’s t-test. Categorical variables are described as percentages (%), and tested by Chi-square tests or Fisher’s exact tests. Pearson correlation analysis was employed to investigate the relationship between CAVI and uTSH. Logistic regression was used to obtain odds ratios (ORs) of categorical associated variables. In the context of this logistic regression analysis, an “associated factor” refers to any variable entered into the multivariate model. A “significant associated factor” is specifically defined as a variable with a P-value < 0.05 in the final multivariable logistic regression model. Statistical significance was defined as a two-sided P-value < 0.05.
| Results | ▴Top |
This study analyzed 247 subjects with SCH and 1,749 subjects with normal thyroid function, matched for age and gender. The prevalence of SCH in the studied population was 12.4%. As shown in Table 1, compared with control group, individuals with SCH had significantly increased body weight, waist circumference, BMI, and body fat percentage, but decreased heart rate (HR) and basal metabolic rate (BMR). In patients with SCH, TC, TG, LDL, apoB, and apoB/apoA ratios were higher compared to the control group. However, HDL, apoA, and fasting blood glucose levels were similar between the SCH and control groups (P-values were 0.311, 0.582, and 0.095, respectively). hsCRP levels were slightly higher in the SCH group compared to the control group (P = 0.051), and detail data are shown in Table 2.
 Click to view |
Table 1. Anthropometric Indicators of the
Studied Population |
 Click to view |
Table 2. Blood Biochemical Indicators of the
Studied Population |
It is noteworthy that individuals diagnosed with SCH demonstrate significantly higher levels of CAVI (7.93 ± 1.38 vs. 7.04 ± 1.31, P < 0.001), ABI (1.22 ± 0.08 vs. 1.11 ± 0.08, < 0.001), and baPWV (1,418 ± 302.5 vs. 1,346 ± 279.8, P = 0.003) in comparison to the control cohort. The CAVI is regarded as indicative of atherosclerosis when it reaches or exceeds a value of 9 [18]. Therefore, high CAVI is operationally defined as CAVI ≥ 9, and a comparison was conducted on the prevalence of high CAVI among both subjects with SCH and control group. We found that subjects with SCH had a significantly higher percentage of high CAVI than control group (14.98% vs. 5.60%, P < 0.001). When high ABI and baPWV were defined as ABI ≥ 1.3 and baPWV ≥ 1,800 cm/s, respectively, individuals with SCH exhibited a significantly higher prevalence of high ABI and baPWV compared to control group (2.02% vs. 0.69%, 10.53% vs. 5.77%, with P-values of 0.049 and 0.008, respectively), details in Table 3. These findings suggest that individuals with SCH may have an increased susceptibility to atherosclerosis.
 Click to view |
Table 3. Comparison of the Arteriosclerosis
Indicators in Subjects With Euthyroidism and Subclinical
Hypothyroidism |
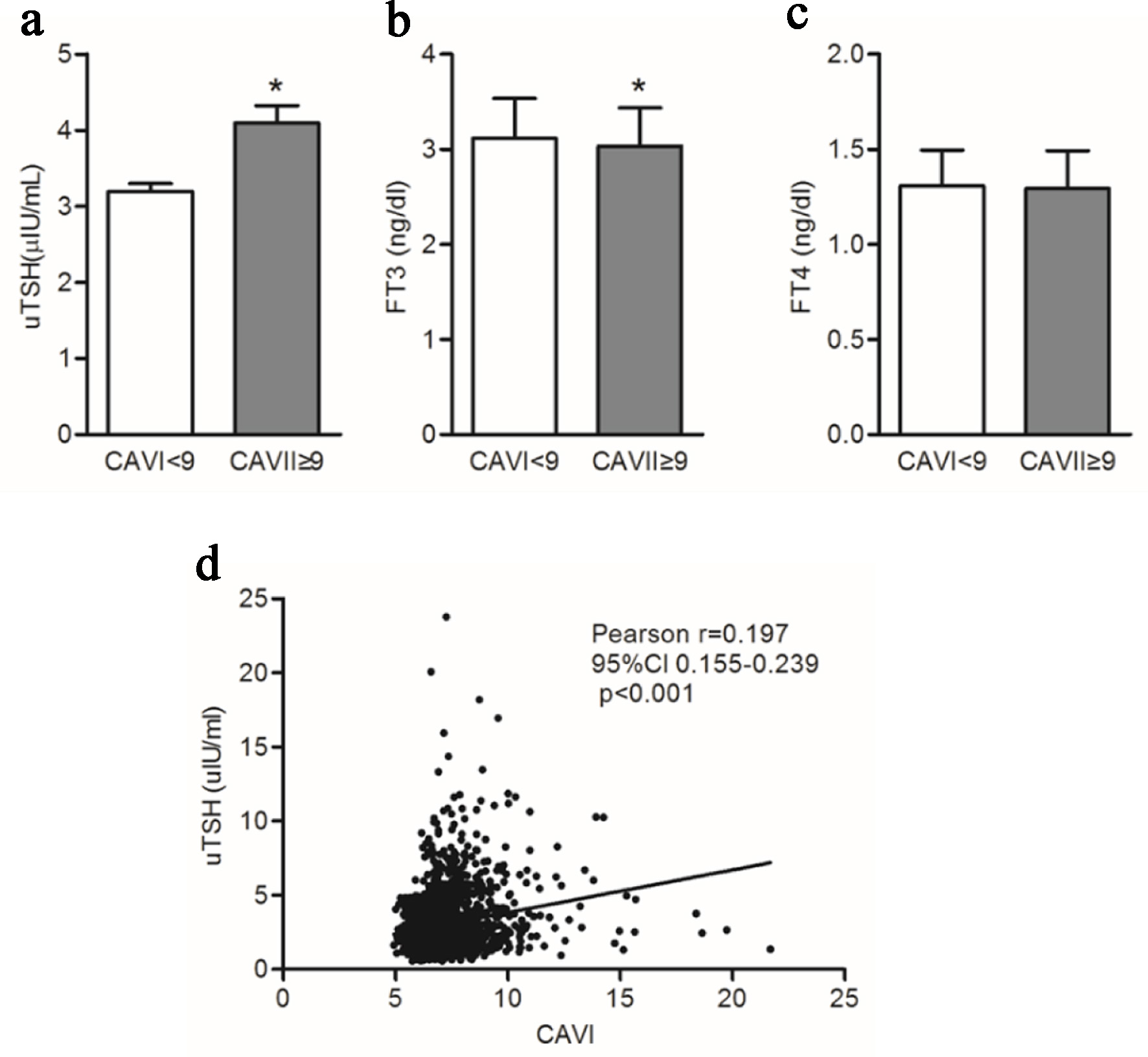
When participants were stratified into two groups according to CAVI values (< 9 and ≥ 9), the group with CAVI ≥ 9 demonstrated a statistically significant increase in uTSH levels (4.10 ± 2.65 vs. 3.19 ± 2.52, P < 0.001) and a slight decrease in FT3 levels (3.03 ± 0.40 vs. 3.12 ± 0.42, P < 0.001), whereas FT4 levels did not differ significantly between groups. Moreover, CAVI is significantly positively correlated with the level of uTSH (Pearson r = 0.20, 95% confidence interval (95% CI), 0.16–0.24, P < 0.001). Details are shown in Figure 1.
 Click for large image |
Figure 1. The relationship between CAVI and thyroid function. (a–c) Comparison of uTSH, FT3, and FT4 in subjects CAVI < 9 and ≥ 9. (d) Correlation analysis between uTSH and CAVI. *A statistically significant difference was observed between the groups (CAVI < 9 vs. ≥ 9), with a P value < 0.001. CAVI: cardio-ankle vascular index; FT3: free triiodothyronine; FT4: free thyroxine; uTSH: ultrasensitive thyroid-stimulating hormone. |
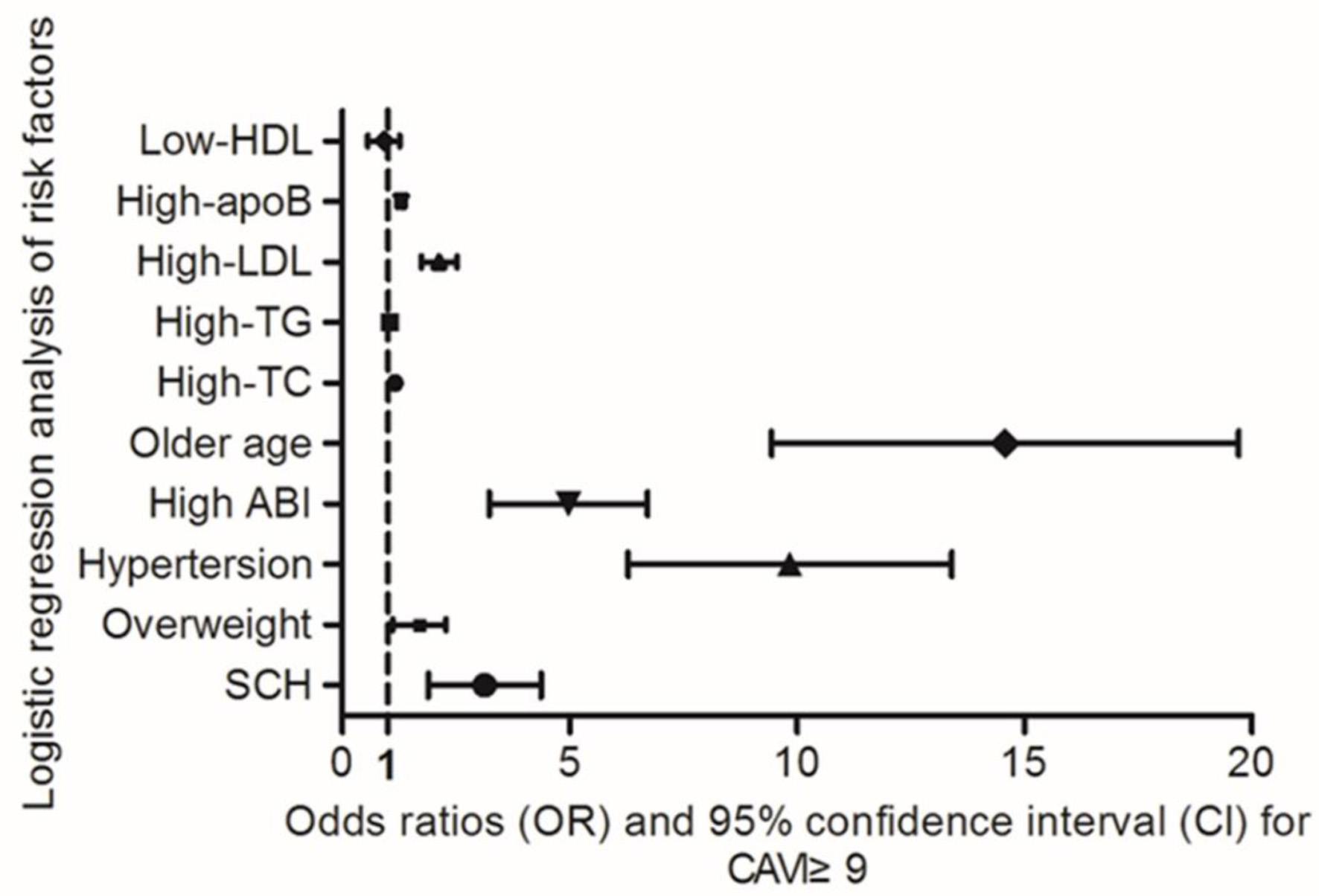
High CAVI (yes or no) was introduced as a dependent variable in the multivariate logistic regression analysis models, and older age, HBP, overweight, high TC, high LDL, high TG, low HDL, high apoB, and SCH (both classified as yes or no) as independent variables. In the present model, advanced age and hypertension was identified as significant associated factors for elevated CAVI, with ORs of 12.89 (95% CI, 10.49–20.35) and 9.41 (95% CI, 6.52–13.60), respectively. Additionally, high ABI, SCH, and overweight were determined to be independent associated factors for elevated CAVI, with ORs of 4.79 (95% CI, 3.32–6.80), 2.97 (95% CI, 1.98–4.45) and 1.63 (95% CI, 1.14–2.31), respectively. Hyperlipidemia is associated with an increased risk of elevated CAVI, particularly in relation to high LDL (OR 2.1, 95% CI, 1.75–2.54), high apoB (OR 1.29, 95% CI, 1.21–1.38), high TC (OR 1.16, 95% CI, 1.11–1.22), and high TG (OR 1.04, 95% CI, 1.01–1.22). Details are shown in Figure 2.
 Click for large image |
Figure 2. Logistic regression analysis of factors associated with elevated CAVI (≥ 9). CAVI was dichotomized into normal (< 9, coded 0) and abnormal (≥ 9, coded 1). Similarly, older age, hypertension, overweight, high LDL, high TG, low HDL, high ABI, and high apoB were defined as binary variables (presence = 1, absence = 0). ABI: ankle-brachial index; apoB: apolipoprotein B; CAVI: cardio-ankle vascular index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; TG: triglycerides. |
| Discussion | ▴Top |
Recent meta-analyses of population-based studies with long-term follow-up periods have provided further insight into the increased risk of cardiovascular disorders in patients with SCH. Specifically, SCH has been shown to elevate the risk of cardiovascular morbidity and mortality, particularly when TSH levels exceed 10 mU/L [9]. Increased arterial stiffness is closely associated with the progression of cardiovascular disease. A newer, blood pressure–independent measure of arterial stiffness, the CAVI, has been developed and is now widely adopted in clinical and research [18, 23, 26]. However, whether an association exists between SCH and CAVI, particularly in the Chinese population, has not been established. Therefore, this study was conducted in a Chinese population to investigate this association.
In this study, 247 subjects with SCH and 1,749 age- and gender-matched control group were included. The prevalence of SCH in this studied population was 12.4%. The observed differences in body weight, fat percentage, and dyslipidemia, which are themselves established risk factors for cardiovascular disease, may be part of the link between SCH and cardiovascular risk [29, 30]. However, the cross-sectional design precludes determining whether these differences are a cause or a consequence of SCH, or are influenced by shared confounders. The levels of hsCRP were marginally elevated in patients with SCH as compared to the control group, indicating a potential association between SCH and chronic low-grade metabolic inflammation. A previous study [31] investigated the presence of atherosclerosis, endothelial dysfunction, and inflammation in SCH. The findings revealed the presence of endothelial dysfunction and low-grade chronic inflammation in individuals with SCH. Many factors, including alterations in lipid profile, hyperhomocysteinemia, and low-grade chronic inflammation, were identified as potential contributors to endothelial dysfunction in individuals with SCH.
In the present study, patients with SCH exhibited higher CAVI, ABI, and baPWV values compared with euthyroid controls. Upon establishing specific cutoff values for defining elevated levels of CAVI, ABI, and baPWV, it becomes evident that a greater proportion of patients with SCH surpass these thresholds compared to the control group. This discrepancy suggests an increased susceptibility to atherosclerosis among individuals with SCH. Alternatively, upon stratifying the research participants into low and high CAVI groups, defined by CAVI < 9 and ≥ 9, respectively, our analysis revealed a significant elevation in serum uTSH levels in the high CAVI group. This increase in uTSH was found to be positively associated with CAVI, while levels of FT3 showed a slight decrease and FT4 levels exhibited no statistically significant difference between the two groups. A previous study [32] established a dose-response relationship between serum TSH levels and hypertension risk among euthyroid individuals, indicating that even within the normal range, a higher TSH level correlates with a greater risk of developing hypertension. Further supporting evidence comes from an investigation [33], which identified a positive association between normal-range serum TSH levels and obesity. The relationship between TSH levels and various diseases in individuals is becoming more prominent in research. Previous studies have demonstrated a connection between TSH levels and conditions such as stroke [34], cardiovascular diseases [9, 12], metabolic syndrome [35], and dyslipidemia [36]. Our research indicates a clear association between serum TSH levels and CAVI, providing new epidemiological evidence for the detrimental effects of elevated TSH levels on increased arterial stiffness and the risk of cardiovascular diseases in humans.
Using logistic regression, we found that advanced age and hypertension were significantly and independently associated with high CAVI. High ABI, SCH, and overweight were determined to be independent associated factors for elevated CAVI. Additionally, hyperlipidemia (high TC, TG, LDL, and apoB) is also associated with an increased risk of elevated CAVI. How SCH exactly increases the CAVI remains unclear. Current understanding offers several possible explanations for this correlation. First, elevated TSH in SCH is associated with impaired endothelial function, a potential driver of atherosclerotic changes [37]. Second, high TSH levels in SCH individuals are also linked to dyslipidemia, which may promote arterial stiffness through mechanisms involving lipotoxicity and persistent, low-grade metabolic inflammation [36]. Beyond the classical pathways involving lipid metabolism and systemic inflammation, elevated TSH may contribute to arterial stiffness through more direct effects on vascular function [38–40]. Potential mechanisms include dysregulation of vascular smooth muscle tone, possibly mediated by altered nitric oxide bioavailability and endothelial function, as well as increased oxidative stress leading to accelerated vascular aging and reduced elasticity [41]. Furthermore, subclinical hypothyroidism has been linked to a pro-oxidative state and metabolic dysregulation, which can synergistically promote vascular remodeling and stiffening [42, 43]. While our current study did not directly measure these parameters, they represent important mechanistic avenues for future research to elucidate the direct vascular effects of TSH.
There are several potential limitations of our study as in the cross-sectional observational study we previously published [24, 25]. The main limitation is the lack of information on lifestyle, smoking, and diet, which may be helpful to understand the relationship between CAVI and SCH. Further studies including detailed personal information were required. Second, because this was a cross-sectional study, causality cannot be established. Longitudinal studies are required to confirm whether elevated TSH directly contributes to arterial stiffness progression. Third, despite adjusting for a range of demographic and clinical variables, our study lacked data on several important potential confounders. These include detailed medication history (e.g., statins and antihypertensive agents), thyroid antibody status (thyroid peroxidase antibody (TPOAb), thyroglobulin antibody (TgAb)), and objective measures of physical activity. The absence of this information may limit the ability to fully account for all confounding pathways, particularly those related to pharmacological interventions, autoimmune thyroiditis, and energy expenditure. Future prospective studies incorporating these parameters would provide a more robust assessment of the independent relationship between TSH and arterial stiffness.
Conclusions
In summary, this study provides epidemiological evidence for the association between serum TSH levels and the association of high CAVI in patients with SCH. Elevated serum TSH levels are an important associated factor for increased CAVI, and SCH patients with higher TSH levels have a greater association of developing increased arterial stiffness and cardiovascular diseases. The specific molecular mechanisms underlying the association between SCH and CAVI need to be further elucidated in future studies.
Acknowledgments
We would like to thank the Health Check-up Center of our hospital for its assistance in data collection.
Financial Disclosure
This project was supported by the Natural Science Foundation of Chongqing (No. CSTB2024NSCQ-MSX0035).
Conflict of Interest
All authors declare no conflict of interest.
Informed Consent
This study used Health Check-up data that were provided by Health Check-up Center of our hospital and did not involve direct interaction with human subjects. Thus, informed consent was deemed unnecessary and was waived by the Human Research Ethics Committee of the First Affiliated Hospital of Chongqing Medical University.
Author Contributions
Xiao Ya Zheng designed and wrote manuscript. Qiang Wei collected data. Tian Tian Cai contributed to manuscript drafting and English language revision and polishing.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
ABI: ankle-brachial index; apoA: apolipoprotein A; apoB: apolipoprotein B; baPWV: brachial-ankle pulse wave velocity; BMI: body mass index; BMR: basal metabolic rate; CAVI: cardio-ankle vascular index; DBP: diastolic blood pressure; FPG: fasting plasma glucose; FT3: free triiodothyronine; FT4: free thyroxine; HDL: high-density lipoprotein; hsCRP: hypersensitivity C-reactive protein; LDL: low-density lipoprotein; SBP: systolic blood pressure; SCH: subclinical hypothyroidism; SD: standard deviation; uTSH: ultrasensitive thyroid-stimulating hormone
| References | ▴Top |
- Biondi B, Cappola AR, Cooper DS. Subclinical hypothyroidism: a
review. JAMA. 2019;322(2):153-160.
doi pubmed - Biondi B, Cooper DS. The clinical significance of subclinical thyroid
dysfunction. Endocr Rev. 2008;29(1):76-131.
doi pubmed - Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, Dayan
CM, Okosieme OE. Global epidemiology of hyperthyroidism and hypothyroidism. Nat Rev Endocrinol.
2018;14(5):301-316.
doi pubmed - Aoki Y, Belin RM, Clickner R, Jeffries R, Phillips L, Mahaffey KR.
Serum TSH and total T4 in the United States population and their association with participant
characteristics: National Health and Nutrition Examination Survey (NHANES 1999-2002). Thyroid.
2007;17(12):1211-1223.
doi pubmed - Peeters RP. Subclinical hypothyroidism.
N Engl J Med. 2017;377(14):1404.
doi pubmed - Owen PJ, Sabit R, Lazarus JH. Thyroid disease and vascular function.
Thyroid. 2007;17(6):519-524.
doi pubmed - Klein I, Danzi S. Thyroid disease and the heart. Circulation.
2007;116(15):1725-1735.
doi pubmed - Iqbal A, Schirmer H, Lunde P, Figenschau Y, Rasmussen K, Jorde R.
Thyroid stimulating hormone and left ventricular function. J Clin Endocrinol Metab.
2007;92(9):3504-3510.
doi pubmed - Rodondi N, Bauer DC, Cappola AR, Cornuz J, Robbins J, Fried LP,
Ladenson PW, et al. Subclinical thyroid dysfunction, cardiac function, and the risk of heart
failure. The Cardiovascular Health study. J Am Coll Cardiol.
2008;52(14):1152-1159.
doi pubmed - Liu Y, Ma M, Li L, Liu F, Li Z, Yu L, Yang T, et al. Association
between sensitivity to thyroid hormones and dyslipidemia in patients with coronary heart
disease. Endocrine. 2023;79(3):459-468.
doi pubmed - Gencer B, Collet TH, Virgini V, Bauer DC, Gussekloo J, Cappola AR,
Nanchen D, et al. Subclinical thyroid dysfunction and the risk of heart failure events: an
individual participant data analysis from 6 prospective cohorts. Circulation.
2012;126(9):1040-1049.
doi pubmed - Rodondi N, den Elzen WP, Bauer DC, Cappola AR, Razvi S, Walsh JP,
Asvold BO, et al. Subclinical hypothyroidism and the risk of coronary heart disease and
mortality. JAMA. 2010;304(12):1365-1374.
doi pubmed - Moon S, Kim MJ, Yu JM, Yoo HJ, Park YJ. Subclinical hypothyroidism
and the risk of cardiovascular disease and all-cause mortality: a meta-analysis of prospective
cohort studies. Thyroid. 2018;28(9):1101-1110.
doi pubmed - Cooper DS, Biondi B. Subclinical thyroid disease. Lancet.
2012;379(9821):1142-1154.
doi pubmed - van Popele NM, Grobbee DE, Bots ML, Asmar R, Topouchian J, Reneman
RS, Hoeks AP, et al. Association between arterial stiffness and atherosclerosis: the Rotterdam
Study. Stroke. 2001;32(2):454-460.
doi pubmed - Cecelja M, Chowienczyk P. Role of arterial stiffness in
cardiovascular disease. JRSM Cardiovasc Dis. 2012;1(4).
doi pubmed - Ibata J, Sasaki H, Kakimoto T, Matsuno S, Nakatani M, Kobayashi M,
Tatsumi K, et al. Cardio-ankle vascular index measures arterial wall stiffness independent of
blood pressure. Diabetes Res Clin Pract. 2008;80(2):265-270.
doi pubmed - Shirai K, Utino J, Otsuka K, Takata M. A novel blood
pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI).
J Atheroscler Thromb. 2006;13(2):101-107.
doi pubmed - Shirai K. Analysis of vascular function using the cardio-ankle
vascular index (CAVI). Hypertens Res. 2011;34(6):684-685.
doi pubmed - Shirai K, Hiruta N, Song M, Kurosu T, Suzuki J, Tomaru T, Miyashita
Y, et al. Cardio-ankle vascular index (CAVI) as a novel indicator of arterial stiffness: theory,
evidence and perspectives. J Atheroscler Thromb. 2011;18(11):924-938.
doi pubmed - Sun CK. Cardio-ankle vascular index (CAVI) as an indicator of
arterial stiffness. Integr Blood Press Control. 2013;6:27-38.
doi pubmed - Nakamura K, Tomaru T, Yamamura S, Miyashita Y, Shirai K, Noike H.
Cardio-ankle vascular index is a candidate predictor of coronary atherosclerosis. Circ J.
2008;72(4):598-604.
doi pubmed - Takaki A, Ogawa H, Wakeyama T, Iwami T, Kimura M, Hadano Y, Matsuda
S, et al. Cardio-ankle vascular index is superior to brachial-ankle pulse wave velocity as an
index of arterial stiffness. Hypertens Res. 2008;31(7):1347-1355.
doi pubmed - Zheng X, Wei Q, Long J, Gong L, Chen H, Luo R, Ren W, et al.
Gender-specific association of serum uric acid levels and cardio-ankle vascular index in Chinese
adults. Lipids Health Dis. 2018;17(1):80.
doi pubmed - Zheng X, Yu S, Long J, Wei Q, Liu L, Liu C, Ren W. Comparison of the
clinical characteristics of primary thyroid lymphoma and diffuse sclerosing variant of papillary
thyroid carcinoma. Endocr Connect. 2022;11(1).
doi pubmed - Saiki A, Sato Y, Watanabe R, Watanabe Y, Imamura H, Yamaguchi T, Ban
N, et al. The role of a novel arterial stiffness parameter, Cardio-Ankle Vascular Index (CAVI),
as a surrogate marker for cardiovascular diseases. J Atheroscler Thromb.
2016;23(2):155-168.
doi pubmed - Saiki A, Ohira M, Yamaguchi T, Nagayama D, Shimizu N, Shirai K,
Tatsuno I. New horizons of arterial stiffness developed using Cardio-Ankle Vascular Index
(CAVI). J Atheroscler Thromb. 2020;27(8):732-748.
doi pubmed - Ohkuma T, Ninomiya T, Tomiyama H, Kario K, Hoshide S, Kita Y,
Inoguchi T, et al. Brachial-ankle pulse wave velocity and the risk prediction of cardiovascular
disease: an individual participant data meta-analysis. Hypertension.
2017;69(6):1045-1052.
doi pubmed - Svare A, Nilsen TI, Bjoro T, Asvold BO, Langhammer A. Serum TSH
related to measures of body mass: longitudinal data from the HUNT Study, Norway. Clin Endocrinol
(Oxf). 2011;74(6):769-775.
doi pubmed - Treister-Goltzman Y, Yarza S, Peleg R. Lipid profile in mild
subclinical hypothyroidism: systematic review and meta-analysis. Minerva Endocrinol (Torino).
2021;46(4):428-440.
doi pubmed - Turemen EE, Cetinarslan B, Sahin T, Canturk Z, Tarkun I. Endothelial
dysfunction and low grade chronic inflammation in subclinical hypothyroidism due to autoimmune
thyroiditis. Endocr J. 2011;58(5):349-354.
doi pubmed - He W, Li S, Wang B, Mu K, Shao X, Yao Q, Li L, et al. Dose-response
relationship between thyroid stimulating hormone and hypertension risk in euthyroid individuals.
J Hypertens. 2019;37(1):144-153.
doi pubmed - Zhang X, Li Y, Zhou X, Han X, Gao Y, Ji L. Association between serum
thyrotropin within the euthyroid range and obesity. Endocr J. 2019;66(5):451-457.
doi pubmed - Chaker L, Baumgartner C, den Elzen WP, Collet TH, Ikram MA, Blum MR,
Dehghan A, et al. Thyroid function within the reference range and the risk of stroke: an
individual participant data analysis. J Clin Endocrinol Metab.
2016;101(11):4270-4282.
doi pubmed - Ruhla S, Weickert MO, Arafat AM, Osterhoff M, Isken F, Spranger J,
Schofl C, et al. A high normal TSH is associated with the metabolic syndrome. Clin Endocrinol
(Oxf). 2010;72(5):696-701.
doi pubmed - Wang F, Tan Y, Wang C, Zhang X, Zhao Y, Song X, Zhang B, et al.
Thyroid-stimulating hormone levels within the reference range are associated with serum lipid
profiles independent of thyroid hormones. J Clin Endocrinol Metab.
2012;97(8):2724-2731.
doi pubmed - Volzke H, Robinson DM, Spielhagen T, Nauck M, Obst A, Ewert R, Wolff
B, et al. Are serum thyrotropin levels within the reference range associated with endothelial
function? Eur Heart J. 2009;30(2):217-224.
doi pubmed - Saric MS, Jurasic MJ, Budincevic H, Milosevic M, Kranjcec B, Kovacic
S, Leskovar J, et al. The role of thyroid hormones in carotid arterial wall remodeling in women.
Rom J Intern Med. 2022;60(1):24-33.
doi pubmed - Kwon BJ, Roh JW, Lee SH, Lim SM, Park CS, Kim DB, Jang SW, et al. A
high normal thyroid-stimulating hormone is associated with arterial stiffness, central systolic
blood pressure, and 24-hour systolic blood pressure in males with treatment-naive hypertension
and euthyroid. Int J Cardiol. 2014;177(3):949-956.
doi pubmed - Isaila OM, Stoian VE, Fulga I, Piraianu AI, Hostiuc S. The
relationship between subclinical hypothyroidism and carotid intima-media thickness as a
potential marker of cardiovascular risk: a systematic review and a meta-analysis. J Cardiovasc
Dev Dis. 2024;11(4).
doi pubmed - Razvi S, Jabbar A, Pingitore A, Danzi S, Biondi B, Klein I, Peeters
R, et al. Thyroid hormones and cardiovascular function and diseases. J Am Coll Cardiol.
2018;71(16):1781-1796.
doi pubmed - Pingitore A, Gaggini M, Mastorci F, Sabatino L, Cordiviola L,
Vassalle C. Metabolic syndrome, thyroid dysfunction, and cardiovascular risk: the triptych of
evil. Int J Mol Sci. 2024;25(19).
doi pubmed - Wang P, Zhang W, Liu H. Research status of subclinical hypothyroidism
promoting the development and progression of cardiovascular diseases. Front Cardiovasc Med.
2025;12:1527271.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution 4.0 International
License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any
medium, including commercial use, provided the original work is properly
cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.