| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 16, Number 2, April 2026, pages 118-127
Neuroprotective and Neurodegenerative Markers in the Early Detection of Peripheral Neuropathy in Type 2 Diabetes Patients
Iman Naeem Tahera, b , Narjis Hadi Al-Saadia
aDepartment of Chemistry, College of Science, University of Karbala, Karbala, Iraq
bCorresponding Author: Iman Naeem Taher, Department of Chemistry, College of Science, University of Karbala, Karbala, Iraq
Manuscript submitted November 5, 2025, accepted February 2, 2026, published online April 8, 2026
Short title: Biomarkers in Diabetic Neuropathy
doi: https://doi.org/10.14740/jem1595
| Abstract | ▴Top |
Background: Diabetic peripheral neuropathy (DPN) is a prevalent, severe diabetic consequence. Exploring relationship between biomarker levels and DPN may help improve understanding disease mechanisms and support future biomarker-oriented research. Meanwhile, fibroblast growth factor 1 (FGF1) and heat shock protein 27 (HSP27) indicated nerve protection; whereas, semaphorin3A (Sema3A) and nerve filament light chain (NFL) indicated damage to a nerve. This study was designed to assess the association between biomarker levels and occurrence of DPN.
Methods: A case-control study comprised 45 DPN patients diagnosed by nerve conduction studies (NCS), 48 diabetic without neuropathy (DWN) patients, and 45 healthy controls. The participants’ ages ranged from 35 to 60 years. Serum FGF1, HSP27, Sema3A, and NFL concentrations were measured using sandwich and competitive enzyme linked immunosorbent assay (ELISA) techniques.
Results: DPN group showed elevation in HSP27 and Sema3A levels relative to DWN group (P = 0.016 and P = 0.027, respectively). The receiver operating characteristic (ROC) curve showed the area under the curve (AUC) values between DPN and DWN groups, which were 0.469 for FGF1, 0.746 for HSP27, 0.783 for Sema3A, and 0.625 for NFL. Besides, the results indicate that there is an association between FGF1, Sem3A and neuropathy (β = −0.010, P < 0.001 for FGF1; β = 3.007, P ≤ 0.001 for Sema3A), and a significant elevation was found in age and duration of injury with disease (β = 0.129, P < 0.001 for age; β = 0.589, P < 0.001 for duration).
Conclusion: The results indicate that HSP27 and Sema3A are linked to DPN and show a capacity to differentiate between DPN and DWN patients. A notable correlation is observed between age, duration, and disease progression. Further studies are needed.
Keywords: Type 2 diabetes; Peripheral neuropathy; Neuroprotective markers; Neurodegenerative
| Introduction | ▴Top |
A prevalent and debilitating condition of diabetes mellitus is diabetic peripheral neuropathy (DPN). Health care providers often misdiagnose it and treat it insufficiently [1]. Peripheral neuropathy affects both large and small nerve fibers. Nerve conduction studies (NCS) can accurately show damage to large fibers, which are responsible for touch, pressure, and vibration. In contrast, small fibers convey pain and temperature sensations and are difficult to diagnose using routine clinical examinations. Notably, neuropathic pain resulting from small-fiber damage may manifest only at a later stage, even though the degenerative process has begun earlier [2]. DPN is a common chronic complication of type 2 diabetes mellitus, affecting up to 50% of patients after prolonged disease duration. The predominant symptomatic features are numbness, pain, and tingling in both lower extremities. In more advanced stages, foot ulcers can occur, increasing the risk of serious infections happening and possibly leading to the amputation of the lower limb [3, 4]. Chronic hyperglycemia is considered the fundamental cause of DPN, as persistently elevated blood glucose levels damage both the microvasculature and peripheral nerves. Strict glycemic control significantly reduces the risk of DPN in patients with type 1 diabetes; however, it does not provide the same benefit in those with type 2 diabetes because it may be related to other metabolic risk factors. Moreover, components of the metabolic syndrome, including central obesity and hypertriglyceridemia, are recognized as important additional risk factors for the development of DPN [5]. Several pieces of research revealed that older adults and low-income and undereducated populations have an elevated incidence of DPN. Several factors may help to explain this, including longer duration of diabetes often seen in older individuals, as well as limited access to adequate healthcare among people with lower socioeconomic status [6, 7]. Overall, clinical management aims to ease symptoms, improve glycemic control, and reduce cardiovascular risk factors [8]. Pain is one of the most distressing symptoms for patients with painful PN and adversely affects both their psychological and physical well-being. Neuropathic pain medications such as gabapentinoids, serotonin-norepinephrine reuptake inhibitors, tricyclic antidepressants, alpha-lipoic acid, sodium channel blockers, and topical capsaicin treat the pain. Pregabalin, duloxetine, tapentadol, and the 8% capsaicin patch have been approved by the US Food and Drug Administration (FDA) to treat painful DPN. Electrical stimulation of the spinal cord was recently approved by the FDA [9–11]. Through its interaction with specific receptors, the fibroblast growth factor (FGF) family—a group of signaling proteins—plays a vital role in several biological functions, including angiogenesis, tissue repair, metabolic regulation, and early developmental processes. As scientific understanding of FGFs has grown recently, interest has steadily increased in their therapeutic potential. Such curiosity has prompted a growing number of researchers to investigate the localized application of FGF variants—such as FGF-1, FGF-2, FGF-4, FGF-7, FGF-21, and FGF-23—in the treatment of diabetic foot ulcers (DFUs), which is in line with the results of a promising clinical study [12]. FGF1 promotes cellular proliferation in addition to influencing various cell types within the body, including those in liver, blood vessels, and skin [13]. In a study including patients with type 2 diabetes mellitus, serum FGF1 levels were quantified utilizing the enzyme linked immunosorbent assay (ELISA) method. The authors indicated that FGF1 levels were significantly correlated with type 2 diabetes, exhibiting elevated serum concentrations in diabetic patients [14]. The other protective agent is heat shock protein 27 (HSP27). It is a small chaperone protein. It, sometimes known as HSPB1, is a member of the sHSP family. HSP27 helps maintain the proper shape of proteins and allows damaged proteins to be repaired, which helps maintain cellular balance and protects the cell from various types of stress [15]. It is a vital chaperone significantly contributing to numerous fundamental and diverse physiological functions. These encompass thermotolerance, apoptosis, cytoskeletal dynamics, cellular differentiation, protein folding, and others [16]. HSP27 may be of value as a biomarker in DPN due to its role in inhibiting mitochondrial apoptosis in neuronal cells. In addition, HSP27 exerts neuroprotective effects in DPN through its involvement in modulating immune responses that are strongly linked to initiation and progression of the disease, as indicated by this study [17]. A broad class of proteins known as semaphorin3A (Sema3A) plays a critical role in various physiological and pathological processes. These proteins have been identified in viruses, insects, and mammalian species and are widely expressed across a range of tissues. The semaphorin family was classified into eight distinct groups according to structural features and distribution across various biological phyla, encompassing approximately 30 unique proteins. Interestingly, classes 3 through 7 are exclusively present in vertebrates, whereas class 1 semaphorins are restricted to invertebrates [18]. Sema3A plays a role in immune regulation as well as in various pathological and biological processes. Recent studies have demonstrated their association with metabolic disorders such as excess adiposity, fat tissue inflammation processes, as well as diabetes-related clinical sequelae, such as ocular and renal impairments, nerve damage, and delayed wound healing [19]. One possible biomarker for distal symmetric polyneuropathy (DSPN) is the neurofilament light chain (NFL), a blood biomarker of neuroaxonal damage in several neurodegenerative diseases [20]. Researchers have mostly used NFLs to assess axonal damage in the neuronal system [21].
| Materials and Methods | ▴Top |
Study design
The present work is a case-control study including 163 male diabetic patients. Patients were clinically diagnosed with DPN and included by specialist physicians at the diabetes center according to standard diagnostic criteria. Still, only 93 of them were approved, as a number of individuals were excluded due to their failure to undergo the mandatory NCS, and then divided into two groups. The first group, 45 patients with DPN, after they were diagnosed using NCS, were performed and interpreted by experienced neurologists at the Imam Al-Hassan Hospital. DPN was diagnosed based on clinical evaluation and NCS. All patients underwent a detailed clinical neurological assessment performed by a neurologist. Based on the clinical findings, patients were subsequently referred to the NCS unit for evaluation. The NCS assessment included measurement of nerve conduction velocity, sensory amplitudes, and distal latencies. Abnormal findings in at least two peripheral nerves, particularly in a symmetrical and length-dependent pattern, were considered indicative of DPN. The evaluation primarily focused on distal sensory nerves of the lower limbs. The second group comprised 48 patients diagnosed with diabetes without neuropathy (DWN) who visited the Al-Hassan Center for Endocrinology and Diabetes in the period from September 2024 to January 2025, Karbala, Iraq. The third group, 45 apparently healthy participants as controls, came to the central blood bank in Karbala to donate blood. The participants’ ages ranged from 35 to 60 years. Exclusion criteria consisted of cancer patients, patients with type 1 diabetes, underage patients, those with nerve compression-related neuropathy, neuropathy disease, and hypertension sufferers. The study was designed and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies in epidemiology [22].
The specimens were collected from patients and healthy individuals after they gave their consent. This paper followed the University of Kerbala, College of Science’s ethical guidelines under reference number 007CSE.
Specimen collection
Venous blood samples of 5 mL were collected from both diabetic patients and healthy subjects. Specimens were placed in gel tubes at room temperature to allow for clotting, followed by centrifugation at 3,000 × g for 10 min. The serum was isolated and stored in an Eppendorf tube at −80 °C until required for use.
Evaluation of biochemicals
The serum FGF1, HSP27, Sema3A, and NFL concentrations were measured using a competitive and sandwich ELISA kit (BT-LB Company, China).
Statistical analysis
Statistical Package for the Social Sciences (SPSS) was utilized to analyze the data. Moreover, many statistical tools were used to perform the comparison and normalize the data. Finally, a P-value of 0.05 or less was deemed statistically significant in all conducted statistical.
ELISA assay procedure
All serum biomarker levels were determined using commercially available human ELISA kits (Cat. No. E3828Hu for FGF1, Cat. No. E1786Hu for HSP27, Cat. No. E2078Hu for Sema3A, and Cat. No. E4467Hu for NFL; Bioassay Technology Laboratory, BT LAB, China).
Blinding
The measurement of vital indicators was conducted using sampling serums without access to clinical information for participants.
| Results | ▴Top |
Characteristic feature
The questionnaire included demographic questions covering name, age, duration of diabetes, weight, height, HbA1c, blood pressure measurement, smoking status, and symptoms of neuropathy, which include muscle weakness, paresthesia, numbness, a burning sensation, and pain, as well as a family history of the illness (Table 1).
 Click to view | Table 1. Clinical Characteristic Feature |
Distribution of samples
The results exhibited a non-normal distribution (P < 0.05) depending on distribution tests (Kolmogorov-Smirnov and Shapiro-Wilk tests) (Fig. 1).
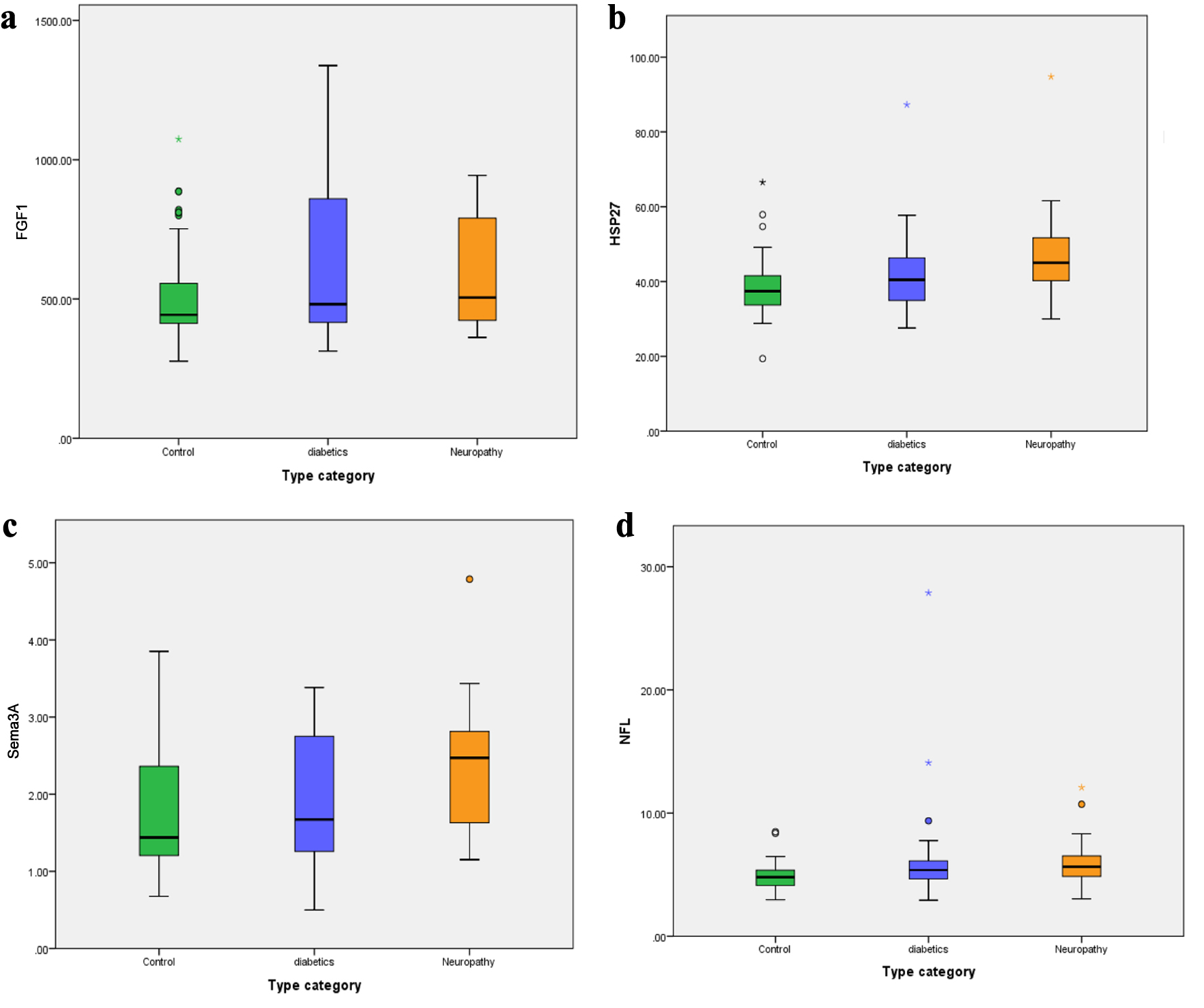 Click for large image | Figure 1. Box-whisker plots of serum (a) fibroblast growth factor 1 (FGF1), (b) heat shock protein 27 (HSP27), (c) semaphorin3A (Sema3A), and (d) nerve filament light chain (NFL) levels in diabetic neuropathy, diabetic patients, and control groups. |
FGF1, HSP27, Sema3A, and NFL concentrations were measured in the serum of DPN patients and control groups. The data indicated a significant increase in HSP27 (P = 0.003), Sema3A (P = 0.034), and NFL (P = 0.029) levels in DPN patients compared with the control, but no significant value in FGF1 levels was observed (P = 0.370) (Table 2).
 Click to view | Table 2. Concentration of FGF1, HSP27, Sema3, and NFL in DPN and Control Groups |
Furthermore, when comparing DPN patients with the DWN group, there was a statistically significant increase in serum HSP27 (P = 0.016) and Sema3A levels (P = 0.027). In contrast, the differences in FGF1 and NFL levels between these groups were not statistically significant (P = 0.609 and P = 0.360, respectively) (Table 3).
 Click to view | Table 3. Concentration of FGF1, HSP27, Sema3A, NFL in the DPN and DWN Groups |
Patients with the DPN were subclassified into mild and severe according to severity. The degree of parameters acquired notable value for FGF1 (P = 0.008) and HSP27 (P = 0.029), but Sema3A and NFL disappeared (P = 0.198 for Sema3A and P = 0.152 for NFL) as shown in Table 4.
 Click to view | Table 4. Concentration of Parameters Under Study With Respect to Severity in Patients With DPN |
Multiple logistic regression was used to find the association between the parameters under study and the incidence of diabetic neuropathy in Table 5.
 Click to view | Table 5. Associations Between the Parameters Under Study and Diabetic Neuropathy |
Binary logistic regression was used to find association between variables (age, duration, BMI) and disease in Table 6.
 Click to view | Table 6. Association Between Variables (Age, Duration, BMI) and Diabetic Neuropathy |
The calculated area under the curve (AUC) for NGF1, HSP27, Sema3A, and NFL between the neuropathy and diabetic groups is shown in Figure 2 and Table 7.
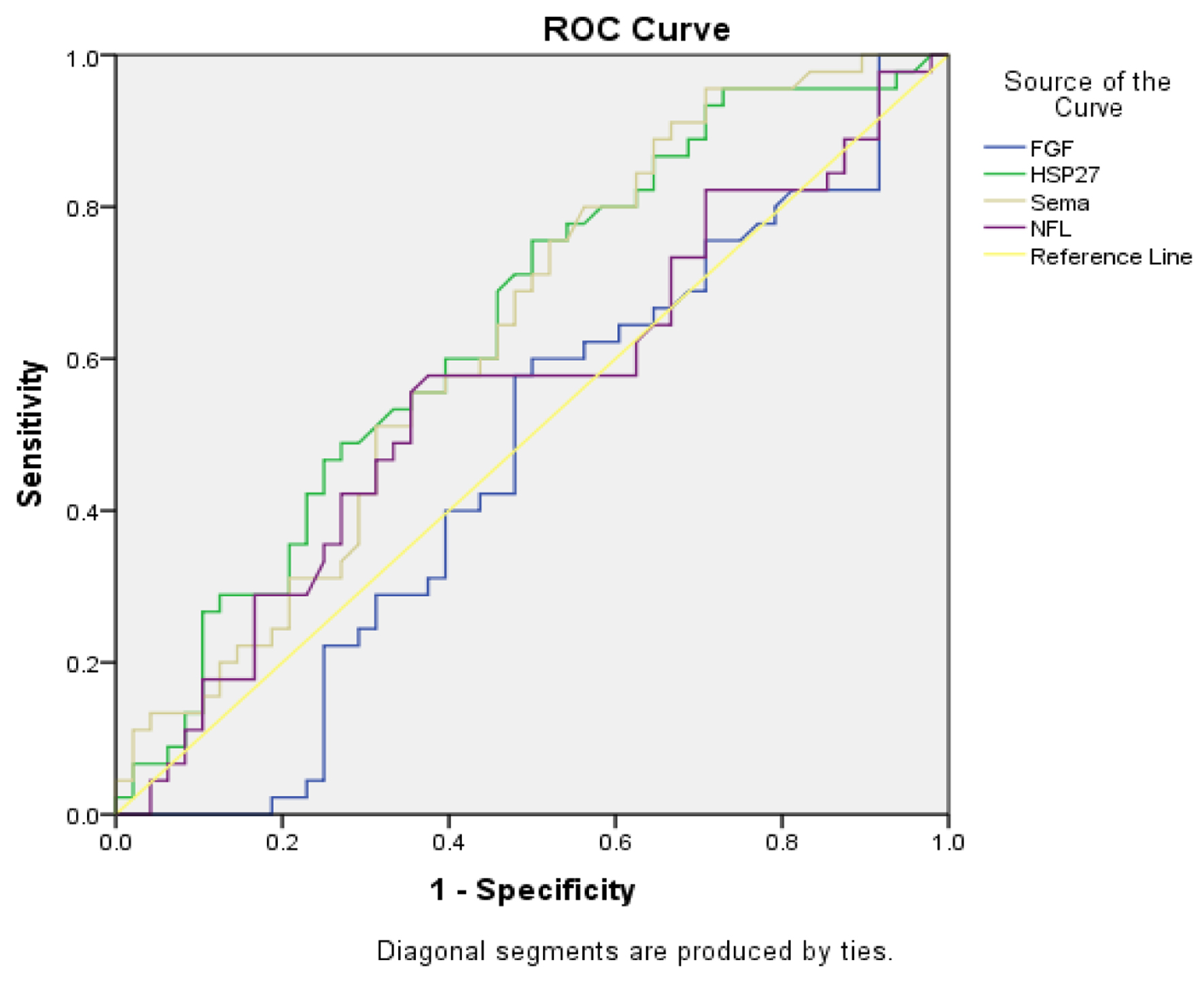 Click for large image | Figure 2. Receiver operating characteristic (ROC) analyses for fibroblast growth factor 1 (FGF1), heat shock protein 27 (HSP27), semaphorin3A (Sema3A), and nerve filament light chain (NFL). |
 Click to view | Table 7. Coordinates of ROC Curve for Serum FGF1, HSP27, Sema3A, and NFL |
| Discussion | ▴Top |
DPN develops in nearly half of individuals with type 2 diabetes. Significantly, autonomic dysfunction, foot ulcers, and neuropathic pain will greatly raise morbidity and death rate, and reduce the quality of life [3]. To support early diagnosis and explore the potential use of certain biomarkers as therapeutic targets, this study evaluated the nerve damage and nerve protection biomarkers. The results showed a significant association between higher levels of HSP27 and the presence of DPN, when comparing the neuropathy group to each of diabetic and control groups. This increase might be seen as the body’s way of coping with ongoing nerve stress and injury, trying to fix the nerve damage, or due to the buildup of incorrectly folded proteins [23]. Another study highlighted the diverse roles of HSP, demonstrating its dual functions. On the one hand, it acts to reduce oxidative stress and inhibit programmed cell death (apoptosis), while on the other hand, it may also exert negative effects under certain pathological conditions [24]. Sensory and motor neuron survival after peripheral nerve injury depends on transcriptional and posttranslational control of HSP27. HSP27 exerts a neuroprotective effect on downstream of cytochrome c release from mitochondria and upstream of caspase-3 activation [25]. Supporting our finding regarding the body’s attempt to repair nerve damage was a study on rats following surgical transection of the sciatic nerve that found that upregulation of HSP27 following adult peripheral nerve injury could both support damaged neuron survival and help to change the cytoskeleton linked with axonal development [26]. Another study conducted in diabetic rats demonstrated that diabetes itself leads to an increased expression of HSP27 even in the absence of nerve injury. This expression further rises following nerve damage, particularly in areas where axons and Schwann cells interact, highlighting a potential role for HSP27 in peripheral nerve regeneration [27]. It reflects the dual role of HSP27, as its elevation may indicate either a protective response or, conversely, an exacerbation of tissue damage or chronic inflammation. This function was demonstrated in a study conducted on COVID-19 patients; this study suggested HSP27 as a sign of advanced pathogenesis of the disease, causing dysregulated systemic inflammation and poorer clinical outcomes in COVID-19 acute respiratory distress syndrome (ARDS) [28]. In the neuronal system, Sema3A is well-known as an axon guidance factor. It has several reported biological roles; it is connected to several human diseases, including autoimmune diseases, angiocardiography, reduced bone density, and tumor development [29]. A statistically significant increase in Sema3A levels was detected in DPN patient group relative to diabetic and control groups; as elevated Sema3A levels have been associated with reduced intraepidermal nerve fiber density (IENFD) as a response to neural stress and chronic injury, and this result agrees with the study that showed increased Sema3A expression and mTOR signaling overactivation were linked to lower IENFD in the skin of diabetic patients relative to control individuals [30]. The present study agrees with the results of a mouse study, suggesting that higher Sema3A expression generated by keratinocytes could be linked to the lower count of tiny fibers seen in DPN [31]. Sema3A exerts a modulatory function in both immunological and inflammatory responses, which is involved in several stages of the immune response, including initiation, antigen presentation, inflammation, and effector T-cell activity, and this has been confirmed by a previous study [32]. A study showed that Sema3A and its receptors are found together in macrophages that come from human monocytes and are being changed by macrophage-colony stimulating factor (M-CSF) [33]. This study found a significant increase in the level of NFL in patients with DPN compared with the control group; the elevated levels of NFL reflect peripheral nerve damage or the gradual loss of nerve fiber. This result agrees with studies showing that common DSPN and nerve dysfunction in newly diagnosed diabetes are linked to higher serum NFL levels [20]. Another study agrees with this result, which showed a higher NFL level with neuropathy patients than healthy controls [34]. Participants with diabetic neuropathy exhibited higher levels of NFL protein in another study on humans [35, 36]. Although NFL levels showed a statistically significant difference when patients with DPN were compared with healthy controls, this difference was not statistically significant when compared with patients with DWN, which could be due to a limited sample size; it could diminish statistical ability to detect differences and increase susceptibility of the results to the influence of extreme values, or perhaps differences in neural response by patients, or differences in degree or type of neural damage. NFLs are becoming more important than other markers as possible signs of nerve damage because their levels go up unusually when there is damage to the nerve fibers in diseases that affect the nervous system, inflammation, and blood vessel problems. They contain naturally unstructured regions, with a key characteristic of these domains being a high content of lysine 10 and 11 residues. Lysine and serine are the most prevalent amino acids within the neurofilament tail domain [37]. In this study, FGF1 remains higher in patients with neuropathy compared to control and diabetes groups; the increase may be a compensatory attempt to repair damaged nerves. It plays a role in nerve regeneration and tissue repair [38]. Elevated FGF1 does not necessarily indicate effective nerve regeneration; another factor might be preventing effective nerve repair, such as chronic inflammation, oxidative stress, or impaired blood supply to the nerve [39]. In healthy adult individuals, brain and kidney tissues show high FGF1 expression. Studies have shown that multiple tissue-specific promoters direct its expression, producing transcripts of varying lengths. The FGF1 gene is widely expressed during development in the neural tube, heart, and lung [40]. Previous research suggests that FGF1 may be linked to better insulin sensitivity. However, the exact molecular mechanisms through which FGF1 influences insulin resistance are not yet unclear [41]. FGF1 plays a regulatory role in the development of post-mitotic neuronal cells, a process that is mediated through coordinated gene expression. This regulation involves a cascade of signals that begin at the cell surface and are transmitted to the nucleus, where FGF1 may remain in the interchromosomal region or associate with distinct nuclear substructures following its nuclear import [42]. In this study, patients with DPN showed significantly higher values of age and duration of diabetes compared to patients with DWN. These findings confirm that advanced age is an important risk factor for the onset and progression of DPN in individuals with diabetes. Elderly patients experience a decline in nerve regeneration efficiency and a reduction in neuronal plasticity. In addition, a longer duration of diabetes leads to continuous and cumulative nerve injury over time, accompanied by a decline in the capacity for nerves to repair [6, 43–45]. While BMI was not statistically significant in the logistic regression analysis model, and the lack of statistical significance may be related to the binary nature of the result, in addition to the stronger effects of age and duration of injury, the apparent effect of BMI may decrease or disappear or to limited variability within the study population, as the majority of participants were overweight or obese.
Conclusion
This study found an association between biomarker levels of HSP27 and Sema3A with presence of DPN. Moreover, age and duration are critical risk factors for uncontrolled diabetes, as corroborated by the regression coefficient values. More research needs to be done on different populations, looking for other factors that can be biomarkers in diagnosis and treatment.
Acknowledgments
I extend my deepest thanks to Dr. Hassan Al-Naqeeb for his generous cooperation in the collection of samples. I would like to express my sincere gratitude to the neurologists for their valuable assistance in performing nerve examinations and nerve conduction studies for the patients. Special thanks are also due to Dr. Ahmed Al-Ibrahimi and Dr. Durgham for their assistance in examining the samples.
Financial Disclosure
This research received no grant.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Written informed consent was obtained from all participants prior to sample collection and data analysis.
Author Contributions
Iman Naeem Taher: methodology, formal analysis, visualization, and writing – original draft. Dr. Narjis Hadi Al-Saadi: conceptualization, supervision, validation, writing – review & editing, and proofreading.
Data Availability
The authors declare that the data supporting the findings of this study are available within the article.
| References | ▴Top |
- Mallick-Searle T, Adler JA. Update on treating painful diabetic peripheral neuropathy: a review of current US guidelines with a focus on the most recently approved management options. J Pain Res. 2024;17:1005-1028.
doi pubmed - Roikjer J, Morch CD, Ejskjaer N. Diabetic peripheral neuropathy: diagnosis and treatment. Curr Drug Saf. 2021;16(1):2-16.
doi pubmed - Zhu J, Hu Z, Luo Y, Liu Y, Luo W, Du X, Luo Z, et al. Diabetic peripheral neuropathy: pathogenetic mechanisms and treatment. Front Endocrinol (Lausanne). 2023;14:1265372.
doi pubmed - Tesfaye S, Kempler P. Conventional management and current guidelines for painful diabetic neuropathy. Diabetes Res Clin Pract. 2023;206(Suppl 1):110765.
doi pubmed - Elafros MA, Andersen H, Bennett DL, Savelieff MG, Viswanathan V, Callaghan BC, Feldman EL. Towards prevention of diabetic peripheral neuropathy: clinical presentation, pathogenesis, and new treatments. Lancet Neurol. 2022;21(10):922-936.
doi pubmed - Wang W, Ji Q, Ran X, Li C, Kuang H, Yu X, Fang H, et al. Prevalence and risk factors of diabetic peripheral neuropathy: A population-based cross-sectional study in China. Diabetes Metab Res Rev. 2023;39(8):e3702.
doi pubmed - Li C, Wang W, Ji Q, Ran X, Kuang H, Yu X, Fang H, et al. Prevalence of painful diabetic peripheral neuropathy in type 2 diabetes mellitus and diabetic peripheral neuropathy: A nationwide cross-sectional study in mainland China. Diabetes Res Clin Pract. 2023;198:110602.
doi pubmed - Yang K, Wang Y, Li YW, Chen YG, Xing N, Lin HB, Zhou P, et al. Progress in the treatment of diabetic peripheral neuropathy. Biomed Pharmacother. 2022;148:112717.
doi pubmed - Jang HN, Oh TJ. Pharmacological and nonpharmacological treatments for painful diabetic peripheral neuropathy. Diabetes Metab J. 2023;47(6):743-756.
doi pubmed - Sloan G, Alam U, Selvarajah D, Tesfaye S. The treatment of painful diabetic neuropathy. Curr Diabetes Rev. 2022;18(5):e070721194556.
doi pubmed - Abrams RMC, Pedowitz EJ, Simpson DM. A critical review of the capsaicin 8% patch for the treatment of neuropathic pain associated with diabetic peripheral neuropathy of the feet in adults. Expert Rev Neurother. 2021;21(3):259-266.
doi pubmed - Liu Y, Liu Y, Deng J, Li W, Nie X. Fibroblast growth factor in diabetic foot ulcer: progress and therapeutic prospects. Front Endocrinol (Lausanne). 2021;12:744868.
doi pubmed - Wu Y, Li Y, Jiang T, Yuan Y, Li R, Xu Z, Zhong X, et al. Reduction of cellular stress is essential for Fibroblast growth factor 1 treatment for diabetic nephropathy. J Cell Mol Med. 2018;22(12):6294-6303.
doi pubmed - Li R, Wang B, Wu C, Li D, Wu Y, Ye L, Ye L, et al. Acidic fibroblast growth factor attenuates type 2 diabetes-induced demyelination via suppressing oxidative stress damage. Cell Death Dis. 2021;12(1):107.
doi pubmed - Zou Y, Shi H, Liu N, Wang H, Song X, Liu B. Mechanistic insights into heat shock protein 27, a potential therapeutic target for cardiovascular diseases. Front Cardiovasc Med. 2023;10:1195464.
doi pubmed - Holguin BA, Hildenbrand ZL, Bernal RA. Insights into the role of heat shock protein 27 in the development of neurodegeneration. Front Mol Neurosci. 2022;15:868089.
doi pubmed - Yin C, Lv J, Huang S, Lang C, Zhao Y, Wang G, Kan J, et al. Therapeutic targets in diabetic peripheral neuropathy: heat shock proteins. Front Endocrinol (Lausanne). 2025;16:1729488.
doi pubmed - Lu Q, Zhu L. The role of semaphorins in metabolic disorders. Int J Mol Sci. 2020;21(16).
doi pubmed - Shen M, Zhou C, Tian Y, Shang T, Qingyun L, Ming M, Ding F, et al. Effects of Semaphorin3A on the growth of sensory and motor neurons. Exp Cell Res. 2023;424(2):113506.
doi pubmed - Maalmi H, Strom A, Petrera A, Hauck SM, Strassburger K, Kuss O, Zaharia OP, et al. Serum neurofilament light chain: a novel biomarker for early diabetic sensorimotor polyneuropathy. Diabetologia. 2023;66(3):579-589.
doi pubmed - Kim AS, Lee JM. The limited role of serum neurofilament light chain in predicting pain severity of patients with diabetic polyneuropathy. Sci Rep. 2024;14(1):15612.
doi pubmed - von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, Initiative S. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453-1457.
doi pubmed - Abisambra JF, Blair LJ, Hill SE, Jones JR, Kraft C, Rogers J, Koren J, 3rd, et al. Phosphorylation dynamics regulate Hsp27-mediated rescue of neuronal plasticity deficits in tau transgenic mice. J Neurosci. 2010;30(46):15374-15382.
doi pubmed - Vidyasagar A, Wilson NA, Djamali A. Heat shock protein 27 (HSP27): biomarker of disease and therapeutic target. Fibrogenesis Tissue Repair. 2012;5(1):7.
doi pubmed - Benn SC, Perrelet D, Kato AC, Scholz J, Decosterd I, Mannion RJ, Bakowska JC, et al. Hsp27 upregulation and phosphorylation is required for injured sensory and motor neuron survival. Neuron. 2002;36(1):45-56.
doi pubmed - Costigan M, Mannion RJ, Kendall G, Lewis SE, Campagna JA, Coggeshall RE, Meridith-Middleton J, et al. Heat shock protein 27: developmental regulation and expression after peripheral nerve injury. J Neurosci. 1998;18(15):5891-5900.
doi pubmed - Stenberg L, Hazer Rosberg DB, Kohyama S, Suganuma S, Dahlin LB. Injury-induced HSP27 expression in peripheral nervous tissue is not associated with any alteration in axonal outgrowth after immediate or delayed nerve repair. Int J Mol Sci. 2021;22(16):8624.
doi pubmed - Chiu MH, Gershkovich B, Yu IL, O'Brien ER, Deng J, McDonald B. Heat shock protein 27 in the pathogenesis of COVID-19 and non-COVID acute respiratory distress syndrome. Cell Stress Chaperones. 2023;28(6):877-887.
doi pubmed - Yang Y, Guan W, Sheng XM, Gu HJ. Role of Semaphorin 3A in common psychiatric illnesses such as schizophrenia, depression, and anxiety. Biochem Pharmacol. 2024;226:116358.
doi pubmed - Wu LY, Li M, Qu ML, Li X, Pi LH, Chen Z, Zhou SL, et al. High glucose up-regulates Semaphorin 3A expression via the mTOR signaling pathway in keratinocytes: a potential mechanism and therapeutic target for diabetic small fiber neuropathy. Mol Cell Endocrinol. 2018;472:107-116.
doi pubmed - Winberg ML, Noordermeer JN, Tamagnone L, Comoglio PM, Spriggs MK, Tessier-Lavigne M, Goodman CS. Plexin A is a neuronal semaphorin receptor that controls axon guidance. Cell. 1998;95(7):903-916.
doi pubmed - Kiseleva EP, Rutto KV. Semaphorin 3A in the immune system: twenty years of study. Biochemistry (Mosc). 2022;87(7):640-657.
doi pubmed - Ji JD, Park-Min KH, Ivashkiv LB. Expression and function of semaphorin 3A and its receptors in human monocyte-derived macrophages. Hum Immunol. 2009;70(4):211-217.
doi pubmed - Gordon BA. Neurofilaments in disease: what do we know? Curr Opin Neurobiol. 2020;61:105-115.
doi pubmed - Morgenstern J, Groener JB, Jende JME, Kurz FT, Strom A, Gopfert J, Kender Z, et al. Neuron-specific biomarkers predict hypo- and hyperalgesia in individuals with diabetic peripheral neuropathy. Diabetologia. 2021;64(12):2843-2855.
doi pubmed - Maatta LL, Andersen ST, Parkner T, Hviid CVB, Bjerg L, Kural MA, Charles M, et al. Serum neurofilament light chain - A potential biomarker for polyneuropathy in type 2 diabetes? Diabetes Res Clin Pract. 2023;205:110988.
doi pubmed - Barro C, Zetterberg H. Neurological symptoms and blood neurofilament light levels. Acta Neurol Scand. 2021;144(1):13-20.
doi pubmed - Chen K, Rao Z, Dong S, Chen Y, Wang X, Luo Y, Gong F, et al. Roles of the fibroblast growth factor signal transduction system in tissue injury repair. Burns Trauma. 2022;10:tkac005.
doi pubmed - Kulebyakin KY, Nimiritsky PP, Makarevich PI. Growth factors in regeneration and regenerative medicine: "the Cure and the Cause". Front Endocrinol (Lausanne). 2020;11:384.
doi pubmed - Jamal SB, Hockman D. Fgf1. Differentiation. 2024;139:100802.
doi pubmed - Fan L, Ding L, Lan J, Niu J, He Y, Song L. Fibroblast growth factor-1 improves insulin resistance via repression of JNK-mediated inflammation. Front Pharmacol. 2019;10:1478.
doi pubmed - Forthmann B, Aletta JM, Lee YW, Terranova C, Birkaya B, Stachowiak EK, Stachowiak MK, et al. Coalition of nuclear receptors in the nervous system. J Cell Physiol. 2015;230(12):2875-2880.
doi pubmed - Lu B, Yang Z, Wang M, Yang Z, Gong W, Yang Y, Wen J, et al. High prevalence of diabetic neuropathy in population-based patients diagnosed with type 2 diabetes in the Shanghai downtown. Diabetes Res Clin Pract. 2010;88(3):289-294.
doi pubmed - Abdissa D. Prevalence and associated factors of painful diabetic peripheral neuropathy among diabetic patients on follow up at Jimma University Medical Center. J Diabetes Metab Disord. 2020;19(2):1407-1413.
doi pubmed - Kiani J, Moghimbeigi A, Azizkhani H, Kosarifard S. The prevalence and associated risk factors of peripheral diabetic neuropathy in Hamedan, Iran. Arch Iran Med. 2013;16(1):17-19.
pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.