| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Review
Volume 16, Number 1, February 2026, pages 1-14
Imeglimin: Discovery, Pharmacology, and Trials
Amedeo Lonardoa, c, Ralf Weiskirchenb
aAOU Modena, Ospedale Civile di Baggiovara (-2023), Modena 41100,
Italy
bInstitute of Molecular Pathobiochemistry, Experimental Gene Therapy, and
Clinical Chemistry (IFMPEGKC), RWTH University Hospital Aachen, Pauwelsstr. 30, D-52074 Aachen,
Germany
cCorresponding Author: Amedeo Lonardo, AOU Modena, Ospedale Civile di
Baggiovara (-2023), Modena 41100, Italy
Manuscript submitted November 25, 2025, accepted January 13, 2026, published online February 25,
2026
Short title: Imeglimin: Discovery, Pharmacology and Trials
doi:
https://doi.org/10.14740/jem1605
| Abstract | ▴Top |
This review briefly summarizes imeglimin’s discovery, development, properties, mechanisms, and core findings on efficacy and safety from major studies. Imeglimin is a first-in-class, orally available tetrahydro-triazine that was designed from the metformin scaffold to address the pathophysiological defects of β-cell dysfunction and insulin resistance in type 2 diabetes mellitus (T2DM). Pre-clinical work shows that imeglimin partially inhibits mitochondrial complex I, corrects complex III deficiency, lowers reactive oxygen species, and boosts NAD+-dependent ATP generation. This enhances glucose-stimulated insulin secretion and preserves β-cell mass. Additionally, the compound augments endogenous glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) release and improves hepatic and skeletal-muscle insulin signaling, resulting in combined insulinotropic and insulin-sensitizing actions. Pharmacokinetic studies reveal rapid absorption, an elimination half-life of approximately 9–12 h, high oral bioavailability, and predominantly renal excretion with no clinically meaningful interactions with metformin or sitagliptin. In phase 2 and 3 trials, the optimized regimen of 1,000 mg twice daily consistently lowers glycated hemoglobin (HbA1c) by approximately 0.8–0.9% as monotherapy and provides additional reductions of 0.4–0.6% when combined with metformin or insulin. It maintains placebo-like tolerability and a minimal risk of hypoglycemia. Long-term data confirm durable glycemic efficacy, a neutral cardiovascular profile with no QT/QTc prolongation, and predominantly mild gastrointestinal adverse events. Real-world evidence supports sustained HbA1c lowering, modest weight loss, and lipid improvements over 12 months, independent of age, sex, body mass index, or renal function. Multivariate and cluster analyses suggest that older age, therapy-naive status, and lower baseline HbA1c predict a more pronounced response, highlighting the importance of patient stratification. Research on imeglimin for T2DM is constrained by small sample sizes, mainly Japanese data, lack of cardiovascular outcome trials, limited comparisons with other antidiabetic drugs, and insufficient long-term safety information. More studies are needed to clarify its efficacy and safety profiles.
Keywords: Imeglimin; Discovery; Pharmacology; T2DM
| Introduction | ▴Top |
According to the International Diabetes Federation, type 2 diabetes mellitus (T2DM) currently affects approximately 589 million adults worldwide and is projected to exceed 853 million by 2050, and of these, four in five adults with diabetes live in low- and middle-income countries (Fig. 1) [1]. These surging prevalence and incidence rates impose an ever-increasing burden of micro- and macrovascular complications on healthcare systems and societies alike [2]. Despite the availability of multiple antidiabetic drug classes, including metformin, sulphonylureas, thiazolidinediones, dipeptidyl-peptidase-4 inhibitors (DPP-4i), glucagon-like peptide-1 receptor agonists (GLP-1RAs) and sodium–glucose co-transporter-2 inhibitors (SGLT2i), a substantial proportion of patients fail to attain or maintain durable glycemic control [3]. This failure is often because traditional agents do not adequately address the complex, multifactorial pathophysiology of T2DM or are limited by tolerability issues such as gastrointestinal disturbance, weight gain, or hypoglycemia, resulting in poor long-term patient adherence to therapy. Consequently, there remains an unmet need for therapies capable of simultaneously improving β-cell function and insulin sensitivity with an acceptable safety profile that may improve patient compliance with drug prescriptions.
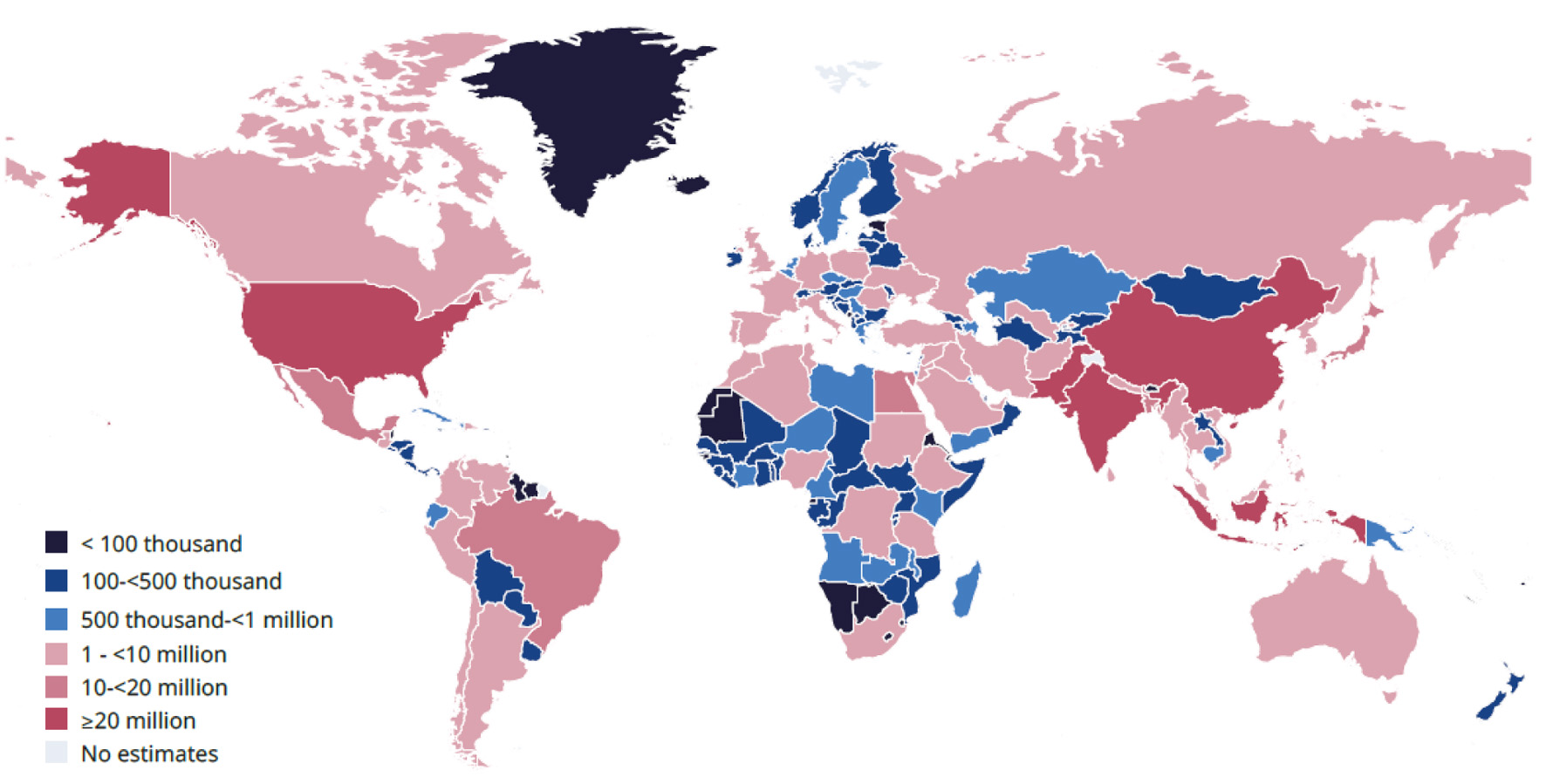 Click for large image |
Figure 1. Estimated number of adults (20–79 years old) with diabetes by country in 2024. The International Diabetes Federation (IDF) has granted permission for copyright for the requested purpose. |
Imeglimin, a first-in-class, orally administered small molecule belonging to the tetrahydro-triazine family, was developed to address a therapeutic gap. It was rationally derived from the biguanide scaffold that serves as the foundation for metformin [4, 5]. Imeglimin was designed to have a dual mode of action: 1) enhancing glucose-stimulated insulin secretion and preserving β-cell mass, and 2) improving insulin action in hepatic and skeletal muscle tissues through targeted modulation of mitochondrial bioenergetics. Pre-clinical studies have shown that imeglimin partially inhibits mitochondrial complex I while correcting complex III dysfunction [4, 6]. This pharmacological activity helps to rebalance electron transport, reduce reactive oxygen species generation (ROS), and increase nicotinamide adenine dinucleotide (NAD+)-dependent ATP production [7]. These mitochondrial effects lead to restored β-cell responsiveness and improved peripheral glucose disposal, setting imeglimin apart from existing antidiabetic agents. Imeglimin has been formally approved and marketed only in Japan although it may be used in some countries without formal approval.
Over the past decade, a comprehensive clinical development program has evaluated imeglimin in various treatment settings. Phase 2 and 3 randomized controlled trials (RCTs) have shown that imeglimin 1,000 mg twice daily reduces glycated hemoglobin (HbA1c) by around 0.8–0.9% as monotherapy or even higher values [8, 9]. When combined with metformin, DPP-4i, or insulin, imeglimin provides additional HbA1c reductions of 0.4–0.6%, while maintaining a safety profile similar to placebo and a minimal risk of hypoglycemia [10]. Long-term extension studies and real-world data support the long-lasting efficacy of imeglimin in managing blood sugar levels. Additionally, imeglimin has been associated with modest weight loss, improvements in lipid profiles, and enhancements of cardia and renal function [5].
This narrative review aims to synthesize the current body of evidence on imeglimin, including its discovery and medicinal-chemistry evolution, physicochemical and pharmacokinetic properties, mitochondrial and incretin-based mechanisms of action, and the efficacy-and-safety findings from pivotal clinical trials and observational studies. Special attention is given to pharmacokinetic interactions, predictors of therapeutic response, and potential positioning within contemporary T2DM treatment algorithms. By integrating chemical, biological, and clinical insights, we aim to provide clinicians, researchers, and policymakers with a comprehensive appraisal of the therapeutic potential of imeglimin and the remaining knowledge gaps that require future investigation.
| Method | ▴Top |
To assemble the evidence for this narrative review, we conducted a thorough search of the MEDLINE database using keywords such as “imeglimin,” “PXL-008,” “ETC-1502,” “type 2 diabetes,” “clinical trial,” “pharmacology,” “mitochondria,” and “β-cell” in various combinations. The time frame considered was 2012–2025 and only articles in English were assessed in more detail. Additionally, we cross-referenced references cited in retrieved articles and relevant review papers to include any additional reports. The eligibility criteria for inclusion in this review were: 1) original experimental or clinical data on imeglimin, 2) peer-reviewed full-text publications, and 3) studies reporting chemical, pharmacokinetic, mechanistic, efficacy, safety, or real-world outcomes. In synthesizing the literature, RCTs, phase II/III studies, and meta-analyses were given the most weight for conclusions regarding efficacy and safety. In particular, we assessed 109 articles that were found in PubMed using the search term: imeglemin AND “type 2 diabetes.” Mechanistic, pharmacokinetic, and early-phase studies were primarily used to support biological plausibility and contextual interpretation. When discussing predictors of therapeutic response, priority was given to studies employing multivariable modeling or data-driven clustering over unadjusted subgroup analyses, and to associations that were consistent across independent datasets. For real-world data, we emphasized prospective or longitudinal cohorts with clearly defined inclusion criteria, standardized outcome measures, and follow-up of at least several months. We highlighted findings that aligned with randomized trial data while treating isolated or heterogeneous observations as hypothesis-generating.
| Discovery | ▴Top |
Imeglimin can be synthesized similarly to phenformin and metformin and has emerged from a rational drug discovery program that aimed to preserve the beneficial mitochondrial and insulin-sensitizing properties of metformin while addressing its limitations in potency and tolerability [11, 12]. Medicinal chemistry campaigns focused on combining the guanidine motif of biguanides with hetero-aromatic scaffolds, leading to a new molecule with a tetrahydro-triazin ring. This effort ultimately produced imeglimin as the primary clinical candidate (Fig. 2).
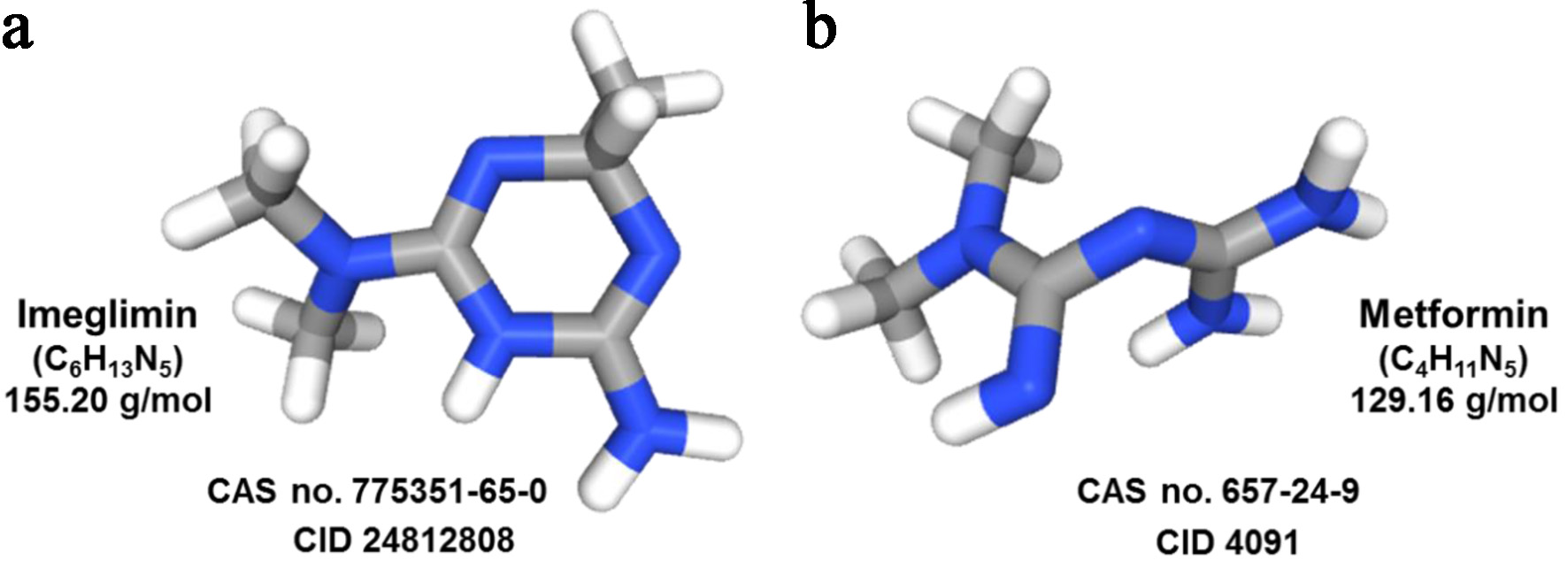 Click for large image |
Figure 2. Chemical structures of imeglimin and metformin. This illustration shows the chemical structures of imeglimin (a) and metformin (b) highlighting the medicinal chemistry continuity and the shared guanidine-derived motif. Imeglimin incorporates this motif into a tetrahydro-triazine scaffold, resulting in a larger ring-fused “sum structure” with dual insulinotropic and insulin-sensitizing activity, while metformin consists of a smaller open-chain biguanide that mainly exerts insulin-sensitizing effects. The structures were taken from the PubChem database with given Compound Identifier (CID) accession numbers. |
Pre-clinical screening has shown that this scaffold selectively and reversibly modulates mitochondrial complex I activity, restores complex III function, and reduces ROS generation. This translates into enhanced β-cell preservation and improved insulin action. These mechanistic features distinguish imeglimin from classical biguanides and justify its advancement into clinical development.
| Pharmacology | ▴Top |
Table 1 provides a comprehensive summary of the main chemical, pharmacological, biochemical, and clinical characteristics that distinguish imeglimin from other glucose-lowering medications. This information is based on a variety of sources [4, 7, 8, 13–16], including the development history of this compound, preclinical studies on its mechanism of action, formal trials in phases 2 and 3, programs examining its interaction within the body, and real-world data. The table organizes key details into four categories: chemistry, pharmacology, biology/biochemistry, and clinical science, to illustrate how imeglimin progresses from molecular design to clinical use. It covers important factors such as molecular weight, solubility in water, recommended oral dose (1,000 mg twice a day), absorption, distribution, metabolism, and excretion (ADME) process, its effects on mitochondria and incretin systems, HbA1c reduction in various treatment scenarios, and its favorable safety profile, including its lack of negative impact on the heart. The table also includes predictive factors for patient response to imeglimin, such as age, prior treatment history, and initial HbA1c levels.
 Click to view |
Table 1. Key Chemical, Pharmacological,
Biochemical, and Clinical Characteristics of Imeglimin |
| Efficacy, Safety, and Pharmacokinetic Profile of Imeglimin in T2DM Management | ▴Top |
Evidence from clinical trials
Overview of clinical evidence
Numerous clinical studies, as summarized in Table 2 [10, 13, 17–35], have evaluated the efficacy, safety, and pharmacokinetic characteristics of imeglimin for the treatment of T2DM. The evidence indicates that imeglimin enhances glycemic control [10, 22, 26, 29, 34], improves insulin secretion and sensitivity [26], and exhibits favorable tolerability both as monotherapy and when combined with other antidiabetic agents [17, 22].
 Click to view |
Table 2. Antidiabetic Efficacy and Safety of
Imeglimin (Evidence From Clinical Trials) |
Comparative efficacy and combination therapies
Comparative studies have shown that imeglimin is as effective as metformin in reducing glycemic area under the curve (AUC), fasting glucose, and HbA1c, while being better tolerated, which supports its use in combination therapies for T2DM [17]. Additionally, adding imeglimin to metformin significantly reduces HbA1c and fasting glucose compared to a placebo, while also improving the proinsulin/insulin ratio and maintaining a similar safety profile [10]. When used with insulin therapy, imeglimin leads to substantial and lasting decreases in HbA1c over periods up to 52 weeks, with a safety profile comparable to placebo and only mild episodes of hypoglycemia [23, 25].
Predictors of therapeutic response
Recent statistical advancements, including cluster and multivariate analyses have identified variables like older age, lack of prior therapy, and lower baseline HbA1c as predictors of a positive response to imeglimin. These predictors have shown varying outcomes across different patient subgroups [29].
Mechanisms of action and metabolic effects
Studies using hyperglycemic clamp techniques have shown that imeglimin significantly enhances insulin secretion and beta-cell sensitivity, leading to its hypoglycemic effects without affecting glucagon levels [18]. Further research indicates that imeglimin not only has direct insulinotropic effects but also stimulates the secretion of GLP-1 and GIP [32]. When combined with metformin, imeglimin may cause mild gastrointestinal symptoms, especially diarrhea, in patients with less than 5 years of disease duration. However, improvements in glycemic control are still apparent even in those experiencing these side effects [33].
Pharmacokinetic interactions
Pharmacokinetic evaluations have shown that co-administering imeglimin with metformin or sitagliptin does not significantly alter systemic drug exposure [19]. Additionally, cimetidine-induced inhibition of the multidrug and toxin extrusion transporter 1 (MATE1) leads to a slight increase in imeglimin concentration, but without notable clinical consequences [20].
Cardiovascular and overall safety
In terms of cardiovascular safety, therapeutic and supratherapeutic doses of imeglimin do not prolong the QT/QTc interval, indicating no significant impact on cardiac parameters like heart rate and electrocardiogram (ECG) intervals [13]. Imeglimin, whether used alone or in combination with other antidiabetic agents, consistently improves HbA1c levels with strong tolerability, minimal risk of hypoglycemia, and an optimal dosage of 1,000 mg twice daily to reduce gastrointestinal side effects [22, 25]. Notably, significant hypoglycemic effects are observed regardless of gastrointestinal symptoms or diarrhea; however, individuals experiencing such symptoms may achieve greater reductions in HbA1c [25, 34].
Evidence from meta-analytic reviews
A limited number of meta-analytic reviews have been conducted based on published original clinical trials (Table 3) [5, 16, 36–38]. The main findings are summarized and discussed below.
 Click to view |
Table 3. Antidiabetic Efficacy and Safety of
Imeglimin (Evidence From Meta-Analytic Reviews) |
Glycemic control and clinical benefits
Imeglimin has been shown to effectively and safely improve glycemic control in patients with T2DM [36]. Clinical evidence indicates significant reductions in both HbA1c and fasting plasma glucose (FPG) levels. These findings highlight the favorable impact of imeglimin on key glycemic parameters, supporting its role as an effective therapeutic agent in diabetes management [16, 37].
Effects on insulin resistance and lipid profile
Despite the observed improvements in glycemic control, imeglimin did not yield significant changes in insulin resistance, as measured by the homeostasis model assessment of insulin resistance (HOMA-IR), nor did it produce notable improvements in lipid parameters [36]. The meta-analyses summarized in Table 3 indicate inconsistent or neutral effects on insulin resistance and lipid parameters.
Tolerability and safety profile
Meta-analyses of RCTs have shown that imeglimin significantly reduces HbA1c levels while maintaining an acceptable tolerability profile. The incidence of adverse events (AEs) was similar to placebo, indicating a favorable safety profile for this medication. Specifically, imeglimin given at a dose of 1,000 mg twice daily seems to offer the best therapeutic benefits for glycemic control without compromising patient safety [16]. This dosing regimen has consistently proven effective in clinical studies, further supporting its potential as a standard treatment option.
Enhancement of β-cell function
In addition to improving glycemic control, imeglimin has shown effectiveness in enhancing β-cell function, which is essential for maintaining normal glucose levels in individuals with T2DM [38]. Importantly, these benefits come without significant AEs, emphasizing the safety of the drug [37].
| Limitations and Future Perspectives | ▴Top |
It is essential to recognize specific limitations prevalent in current research, such as small sample sizes and patient heterogeneity across several studies [38]. Additional concerns in published literature include a predominance of Japanese datasets, the lack of cardiovascular outcome trials, insufficient direct comparisons with GLP-1RAs or SGLT2i, and limited long-term safety data extending beyond 2–3 years. These issues highlight the need for larger and more comprehensive clinical trials to further clarify the role of imeglimin in the management of T2DM and to confirm its long-term efficacy and safety. Looking ahead, it is recommended that future research addresses current limitations by implementing stringent methodologies, enrolling broader patient populations, and incorporating longer follow-up durations. These measures are critical for accurately characterizing the therapeutic profile of imeglimin and ensuring its adoption in clinical practice is based on comprehensive and reliable evidence. A thoughtful approach to these factors will facilitate the optimization of treatment protocols for patients with T2DM and enhance the clinical utility of this innovative therapy.
| Conclusions | ▴Top |
Imeglimin is a first-in-class novel oral glucose-lowering drug derived from metformin that presents an innovative approach to managing T2DM. Unlike traditional treatments, imeglimin targets key pathophysiological pathways by enhancing glucose-stimulated insulin secretion (GSIS) and maintaining β-cell mass. It also improves insulin action, potentially inhibiting hepatic glucose production, and enhances insulin signaling in both hepatic and skeletal muscle tissues. This leads to improved insulin secretion and sensitivity, as well as enhanced glucose utilization in peripheral tissues [4, 39].
Imeglimin may address mitochondrial dysfunction, a key pathogenic factor in T2DM, by rebalancing respiratory chain activity. Specifically, it partially inhibits complex I and corrects deficient complex III, resulting in less oxidative stress and helping to prevent cell death. In rodent models of T2DM, imeglimin improves glucose-stimulated ATP production and boosts NAD+ synthesis through the salvage pathway, supporting increased insulin secretion. Additionally, imeglimin helps preserve β-cell mass and uniquely targets defective cellular energy metabolism, distinguishing from other major diabetes therapies [4].
Multiple key phase III clinical trials have demonstrated statistically significant and clinically relevant reductions in glucose levels alongside an overall positive safety and tolerability profile. Notably, there have been no occurrences of severe hypoglycemia [40]. Additional extensive studies involving varied populations are necessary to validate these findings [5].
Acknowledgments
None to declare.
Financial Disclosure
The authors have no financial interests to disclose.
Conflict of Interest
The authors have nothing to declare.
Author Contributions
AL and RW both participated equally in all steps of this study, from the conception of the article, to data collection and analysis, writing and editing the first draft, final editing, and revising the manuscript based on reviewers’ comments.
Data Availability
The authors declare that the data supporting the findings of this study are available within the article.
Use of Artificial Intelligence Tools
During the preparation of this manuscript, the authors used the free online tool “Edit My English” to enhance readability and ensure that the language is devoid of grammar, spelling, and punctuation errors. After using this tool, the authors carefully reviewed and edited the content as necessary, taking full responsibility for the content of the article.
Abbreviations
ADME: absorption, distribution, metabolism, and excretion; AE: adverse event; AUC: area under the curve; bid: twice daily; BMI: body mass index; CGM: continuous glucose monitoring; CI: confidence interval; CKD: chronic kidney disease; Cmax: maximum plasma concentration; ΔΔQTcF: time-matched, placebo-subtracted, baseline-adjusted QTc interval (Fridericia-corrected); DPP-4i: dipeptidyl-peptidase-4 inhibitor; ECG: electrocardiogram; FPG: fasting plasma glucose; GI: gastrointestinal; GIP: glucose-dependent insulinotropic polypeptide; GLP-1: glucagon-like peptide-1; GLP-1 RA: GLP-1 receptor agonist; GSIS: glucose-stimulated insulin secretion; HbA1c: glycated hemoglobin; HDL-C: high-density lipoprotein cholesterol; HOMA-β: homeostatic model assessment of β-cell function; HOMA-IR: homeostatic model assessment of insulin resistance; HR: heart rate; ISR: insulin secretion rate; LDL-C: low-density lipoprotein cholesterol; LSM: least-squares mean; MAGE: mean amplitude of glycemic excursions; MATE: multidrug and toxin extrusion transporter; NAD+: nicotinamide adenine dinucleotide (oxidized form); NNT: number needed to treat; OCT: organic cation transporter; OD: once daily; OGTT: oral glucose tolerance test; OHA: oral hypoglycemic agent; OR: odds ratio; PG: plasma glucose; QT/QTc: interval from Q-wave onset to T-wave end/heart-rate-corrected QT; QTcF: QT interval corrected by the Fridericia formula; RCT: randomized controlled trial; ROS: reactive oxygen species; SMD: standardized mean difference; T2DM: type 2 diabetes mellitus; TEAE: treatment-emergent adverse event; TIR: total insulin response; t½: terminal elimination half-life; Tmax: time to reach Cmax
| References | ▴Top |
- International Diabetes Federation. IDF Diabetes Atlas, 11th edn. Brussels, Belgium: International Diabetes Federation, 2025. Available from: https://diabetesatlas.org/.
- Forray AI, Gavrilas L, Craciun AE, Borzan CM. The global burden of
disease: a focus on type II diabetes. In: Preedy VR, Patel VB. (eds) Handbook of Public Health
Nutrition. Springer, Cham. 2025.
doi - Polonsky WH, Henry RR. Poor medication adherence in type 2 diabetes:
recognizing the scope of the problem and its key contributors. Patient Prefer Adherence.
2016;10:1299-1307.
doi pubmed - Hallakou-Bozec S, Vial G, Kergoat M, Fouqueray P, Bolze S, Borel AL,
Fontaine E, et al. Mechanism of action of Imeglimin: A novel therapeutic agent for type 2
diabetes. Diabetes Obes Metab. 2021;23(3):664-673.
doi pubmed - Song Q, Mae R, Kutbi E, AlJurayyan AN, Abu-Zaid A, Jamilian P,
Falahatzadeh M. Imeglimin as an effective therapeutic approach in management of type 2 diabetes
mellitus: an umbrella review and systematic review, meta-regression and meta-analysis. Diabetol
Metab Syndr. 2025;17(1):357.
doi pubmed - Yanai H, Adachi H, Hakoshima M, Katsuyama H. Glucose-lowering effects
of imeglimin and its possible beneficial effects on diabetic complications. Biology (Basel).
2023;12(5):726.
doi pubmed - Hallakou-Bozec S, Kergoat M, Fouqueray P, Bolze S, Moller DE.
Imeglimin amplifies glucose-stimulated insulin release from diabetic islets via a distinct
mechanism of action. PLoS One. 2021;16(2):e0241651.
doi pubmed - Shireen SM, Bhavya E, Parthiban R. Imeglimin systematic review: a
novel therapeutic approach for type 2 diabetes-unveiling benefits on beta-cell function, insulin
sensitivity, and potential long-term glycaemic control (HbA1c). Eur J Clin
Pharmacol. 2025;81(12):1713-1731.
doi pubmed - Babazono T, Osonoi T, Okamoto H, Onishi Y, Nakamoto S, Kashima M,
Kawanami D, et al. Long-term safety and efficacy of imeglimin in Japanese individuals with type
2 diabetes and chronic kidney disease: A 52-week postmarketing clinical study (TWINKLE).
J Diabetes Investig. 2025;16(10):1808-1819.
doi pubmed - Fouqueray P, Pirags V, Inzucchi SE, Bailey CJ, Schernthaner G,
Diamant M, Lebovitz HE. The efficacy and safety of imeglimin as add-on therapy in patients with
type 2 diabetes inadequately controlled with metformin monotherapy. Diabetes Care.
2013;36(3):565-568.
doi pubmed - Yendapally R, Sikazwe D, Kim SS, Ramsinghani S, Fraser-Spears R,
Witte AP, La-Viola B. A review of phenformin, metformin, and imeglimin. Drug Dev Res.
2020;81(4):390-401.
doi pubmed - Swain J, Jadhao P, Sravya SL, Teli B, Lavanya K, Singh J, Sahoo A, et
al. Mitochondrial dysfunction and imeglimin: a new ray of hope for the treatment of type-2
diabetes mellitus. Mini Rev Med Chem. 2024;24(17):1575-1589.
doi pubmed - Dubourg J, Perrimond-Dauchy S, Felices M, Bolze S, Voiriot P,
Fouqueray P. Absence of QTc prolongation in a thorough QT study with imeglimin, a first in class
oral agent for type 2 diabetes mellitus. Eur J Clin Pharmacol.
2020;76(10):1393-1400.
doi pubmed - Tomita Y, Hansson E, Mazuir F, Wellhagen GJ, Ooi QX, Mezzalana E,
Kitamura A, et al. Imeglimin population pharmacokinetics and dose adjustment predictions for
renal impairment in Japanese and Western patients with type 2 diabetes. Clin Transl Sci.
2022;15(4):1014-1026.
doi pubmed - Chevalier C, Fouqueray P, Bolze S. Imeglimin: a clinical pharmacology
review. Clin Pharmacokinet. 2023;62(10):1393-1411.
doi pubmed - Hagi K, Nitta M, Watada H, Kaku K, Ueki K. Efficacy, safety and
tolerability of imeglimin in patients with type 2 diabetes mellitus: A meta-analysis of
randomized controlled trials. J Diabetes Investig. 2023;14(11):1246-1261.
doi pubmed - Pirags V, Lebovitz H, Fouqueray P. Imeglimin, a novel glimin oral
antidiabetic, exhibits a good efficacy and safety profile in type 2 diabetic patients. Diabetes
Obes Metab. 2012;14(9):852-858.
doi pubmed - Pacini G, Mari A, Fouqueray P, Bolze S, Roden M. Imeglimin increases
glucose-dependent insulin secretion and improves beta-cell function in patients with type 2
diabetes. Diabetes Obes Metab. 2015;17(6):541-545.
doi pubmed - Fouqueray P, Perrimond-Dauchy S, Bolze S. Imeglimin does not induce
clinically relevant pharmacokinetic interactions when combined with either metformin or
sitagliptin in healthy subjects. Clin Pharmacokinet. 2020;59(10):1261-1271.
doi pubmed - Chevalier C, Perrimond-Dauchy S, Dubourg J, Fouqueray P, Bolze S.
Lack of drug-drug interaction between cimetidine, a renal transporter inhibitor, and imeglimin,
a novel oral antidiabetic drug, in healthy volunteers. Eur J Drug Metab
Pharmacokinet. 2020;45(6):725-733.
doi pubmed - Chevalier C, Dubourg J, Bolze S, Fouqueray P. Pharmacokinetics of
imeglimin in subjects with moderate hepatic impairment. Clin Pharmacokinet.
2021;60(4):485-490.
doi pubmed - Dubourg J, Ueki K, Grouin JM, Fouqueray P. Efficacy and safety of
imeglimin in Japanese patients with type 2 diabetes: A 24-week, randomized, double-blind,
placebo-controlled, dose-ranging phase 2b trial. Diabetes Obes Metab.
2021;23(3):800-810.
doi pubmed - Dubourg J, Fouqueray P, Thang C, Grouin JM, Ueki K. Efficacy and
safety of imeglimin monotherapy versus placebo in Japanese Patients With Type 2 Diabetes (TIMES
1): a double-blind, randomized, placebo-controlled, parallel-group, multicenter phase 3 trial.
Diabetes Care. 2021;44(4):952-959.
doi pubmed - Dubourg J, Fouqueray P, Quinslot D, Grouin JM, Kaku K. Long-term
safety and efficacy of imeglimin as monotherapy or in combination with existing antidiabetic
agents in Japanese patients with type 2 diabetes (TIMES 2): A 52-week, open-label, multicentre
phase 3 trial. Diabetes Obes Metab. 2022;24(4):609-619.
doi pubmed - Reilhac C, Dubourg J, Thang C, Grouin JM, Fouqueray P, Watada H.
Efficacy and safety of imeglimin add-on to insulin monotherapy in Japanese patients with type 2
diabetes (TIMES 3): A randomized, double-blind, placebo-controlled phase 3 trial with a 36-week
open-label extension period. Diabetes Obes Metab. 2022;24(5):838-848.
doi pubmed - Theurey P, Thang C, Pirags V, Mari A, Pacini G, Bolze S,
Hallakou-Bozec S, et al. Phase 2 trial with imeglimin in patients with Type 2 diabetes indicates
effects on insulin secretion and sensitivity. Endocrinol Diabetes Metab. 2022;5(6):e371.
doi pubmed - Hagi K, Kochi K, Watada H, Kaku K, Ueki K. Effect of patient
characteristics on the efficacy and safety of imeglimin monotherapy in Japanese patients with
type 2 diabetes mellitus: A post-hoc analysis of two randomized, placebo-controlled trials.
J Diabetes Investig. 2023;14(9):1101-1109.
doi pubmed - Takahashi A, Nomoto H, Onishi K, Manda S, Miya A, Kameda H, Nakamura
A, et al. A comparative study of the effects of imeglimin add-on or metformin dose escalation on
glycaemic variability in subjects with type 2 diabetes treated with low-dose metformin
(MEGMI-CGM study). Diabetes Obes Metab. 2024;26(8):3471-3474.
doi pubmed - Hagi K, Kochi K, Watada H, Kaku K, Ueki K. Factors contributing to
the clinical effectiveness of imeglimin monotherapy in Japanese patients with type 2 diabetes
mellitus. J Diabetes Investig. 2024;15(9):1239-1247.
doi pubmed - Katsuyama H, Hakoshima M, Heshiki T, Iida S, Adachi H, Yanai H.
Real-world effectiveness of imeglimin in patients with type 2 diabetes: A retrospective
longitudinal study in Japan. Diabetes Res Clin Pract. 2024;213:111752.
doi pubmed - Hagi K, Kochi K, Watada H, Kaku K, Ueki K. Differences in imeglimin
response in subgroups of patients with type 2 diabetes stratified by data-driven cluster
analysis: A post-hoc analysis of imeglimin clinical trial data. Diabetes Obes Metab.
2024;26(9):3732-3742.
doi pubmed - Usui R, Hamamoto Y, Imura M, Omori Y, Yamazaki Y, Kuwata H, Tatsuoka
H, et al. Differential effects of imeglimin and metformin on insulin and incretin secretion-An
exploratory randomized controlled trial. Diabetes Obes Metab. 2025;27(2):856-865.
doi pubmed - Ito J, Hagi K, Kochi K, Ueki K, Watada H, Kaku K. Gastrointestinal
symptoms in patients receiving imeglimin in combination with metformin: A post-hoc analysis of
imeglimin clinical trial data. J Diabetes Investig. 2025;16(4):629-638.
doi pubmed - Takahashi A, Nomoto H, Yokoyama H, Yokozeki K, Furusawa S, Oe Y,
Kameda R, et al. Efficacy of imeglimin treatment versus metformin dose escalation on glycemic
control in subjects with type 2 diabetes treated with a dipeptidyl peptidase-4 inhibitor plus
low-dose metformin: A multicenter, prospective, randomized, open-label, parallel-group
comparison study (MEGMI study). Diabetes Obes Metab. 2025;27(3):1466-1476.
doi pubmed - Kaku K, Shimoda M, Osonoi T, Iwamoto M, Kaneto H. Efficacy and safety
of imeglimin add-on to DPP-4 inhibitor therapy in Japanese patients with type 2 diabetes
mellitus: An interim analysis of the randomised, double-blind FAMILIAR trial. Diabetes Obes
Metab. 2025;27(6):3212-3222.
doi pubmed - Abdelhaleem IA, Salamah HM, Alsabbagh FA, Eid AM, Hussien HM, Mohamed
NI, Ebada MA. Efficacy and safety of imeglimin in patients with type 2 diabetes mellitus: A
systematic review and meta-analysis of randomized clinical trials. Diabetes Metab Syndr.
2021;15(6):102323.
doi pubmed - Singh AK, Singh A, Singh R, Misra A. Efficacy and safety of imeglimin
in type 2 diabetes: A systematic review and meta-analysis of randomized placebo-controlled
trials. Diabetes Metab Syndr. 2023;17(2):102710.
doi pubmed - Tewari J, Qidwai KA, Tewari A, Rana A, Singh V, Tewari V, Mateen R,
et al. Efficacy and safety of imeglimin, a novel oral agent in the management of type 2 diabetes
mellitus: a systematic review and meta-analysis. Naunyn Schmiedebergs Arch Pharmacol.
2025;398(10):14501-14513.
doi pubmed - Hou T, Zhang J, Shi W. Mechanisms and clinical perspectives on
imeglimin for insulin resistance in obese patients. Eur J Pharmacol.
2025;1003:177937.
doi pubmed - Krishnan K, Raman S, Anand Moses CR, Rajesh RP, Gupta A, Mudaliar V,
Vimal J. Phase 3 efficacy and safety trial of proposed liraglutide biosimilar for reduction of
glycosylated hemoglobin (HbA1c) in patients with Type 2 diabetes mellitus. Diabetes Res Clin
Pract. 2024;207:111034.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution 4.0 International
License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any
medium, including commercial use, provided the original work is properly
cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.