| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Review
Volume 16, Number 2, April 2026, pages 51-69
Key Physiopathological Pathways and Emerging Therapeutic Strategies in Type 2 Diabetes Mellitus
Komal Awasthia , Pratyush Singha
, Ashwin Kumar Shuklaa
, Kauser Usmanb
, Monisha Banerjeea, c
aMolecular and Human Genetics Laboratory, Department of Zoology, Faculty of Science, University of Lucknow, Lucknow, India
bDepartment of Medicine, King George’s Medical University, Lucknow, India
cCorresponding Author: Monisha Banerjee, Molecular and Human Genetics Laboratory, Department of Zoology, Faculty of Science, University of Lucknow, Lucknow 226007, India
Manuscript submitted December 15, 2025, accepted February 25, 2026, published online April 8, 2026
Short title: Key Pathways and Therapies in T2DM
doi: https://doi.org/10.14740/jem1622
| Abstract | ▴Top |
Type 2 diabetes mellitus (T2DM) is a progressive metabolic disorder initiated by insulin resistance, leading to compensatory hyperinsulinemia, β-cell exhaustion, and chronic hyperglycemia. It is a major global health burden associated with macrovascular and microvascular complications. Dysregulation of the renin-angiotensin system (RAS), Antioxidant pathway, inflammation, and vitamin D signaling interact in a self-perpetuating metabolic loop driving disease progression. Hepatic insulin resistance plays a central role, while glucotoxic pathways distinguish T2DM from obesity. This review aims to examine the molecular cross-talk among the RAS, antioxidant pathway, inflammation, and vitamin D signaling pathways in T2DM pathogenesis. It seeks to clarify how their interactions drive insulin resistance, β-cell dysfunction, and complications, thereby supporting the development of more precise, mechanism-based therapeutic strategies. The RAS plays a central role in T2DM through overactivation of the angiotensin-converting enzyme/angiotensin II/angiotensin II receptor type 1 (ACE/Ang II/AT1R) axis, promoting insulin resistance, β-cell apoptosis, oxidative stress (OS), inflammation, and vascular damage, while the ACE2/Ang-(1–7)/Mas axis offers protective effects. Ang II–induced ROS production disrupts antioxidant defenses and accelerates glucotoxicity. Chronic low-grade inflammation, marked by increased interleukin-6 (IL-6) and reduced interleukin-10 (IL-10), further worsens metabolic dysfunction. Vitamin D receptor (VDR) signaling counteracts these effects by suppressing renin, reducing Ang II activity, enhancing antioxidant enzymes, and modulating inflammation. The liver serves as a key site where these pathways converge. Therapeutic strategies—including ACE inhibitors, angiotensin II receptor blockers (ARBs), GLP-1 receptor agonists, sodium-glucose cotransporter 2 (SGLT2) inhibitors, dipeptidyl peptidase-4 (DPP-4) inhibitors, antioxidants, and vitamin D—target these interconnected mechanisms, with responses influenced by disease stage and sex differences. T2DM develops through interconnected dysregulation of four key pathways: the RAS, OS–antioxidant imbalance, chronic inflammation, and vitamin D/VDR signaling. These pathways interact to promote insulin resistance, β-cell dysfunction, and vascular complications, driving progression from prediabetes to overt disease. Therapeutic strategies targeting these mechanisms—including RAS inhibitors, antioxidant-based interventions, metformin, and vitamin D supplementation—offer complementary benefits but have limitations and potential adverse effects. Treatment responses may vary by biological factors such as sex and disease stage, highlighting the need for well-designed clinical trials to enable precise, mechanism-based management.
Keywords: Type 2 diabetes mellitus; Renin-angiotensin system pathway; Antioxidant genes; Inflammatory pathway; Vitamin D receptor; Drugs and therapies
| Introduction | ▴Top |
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder primarily driven by insulin resistance (IR), which precedes and contributes to impaired glucose regulation. In the early stages of T2DM, peripheral tissues such as skeletal muscle, adipose tissue, and liver become less responsive to normal circulating insulin levels, leading to compensatory increases in insulin secretion from pancreatic β cells to maintain normoglycemia [1–3]. Over time, persistent IR places excessive demand on β cells, eventually resulting in β-cell dysfunction and insufficient insulin production. According to the International Diabetes Federation (IDF) Diabetes Atlas, 11th edition [4], 11.1% of adults aged 20–79 years are currently living with diabetes, corresponding to approximately 589 million individuals worldwide, a figure projected to rise to 852.5 million by 2050. T2DM accounts for more than 90% of all diabetes cases globally. Based on disability-adjusted life years (DALYs), a standard metric used to quantify global disease burden, diabetes is ranked as the eighth leading cause of disease burden worldwide and is projected to become the second leading cause by 2050 [4, 5]. The urgency of understanding T2DM pathophysiology was further highlighted during the coronavirus disease 2019 (COVID-19) pandemic, where T2DM patients experienced significantly higher mortality rates [6, 7]. This heightened vulnerability was driven largely by the mechanisms discussed in this review, including dysregulated renin-angiotensin system (RAS) signaling (via the angiotensin-converting enzyme 2 (ACE2) receptor), chronic systemic inflammation, and prevalent vitamin D deficiency, all of which increased the viral impact. As discussed in the beginning, IR represents the central initiating defect in T2DM pathogenesis, with β-cell exhaustion arising secondary to sustained metabolic stress [3, 8].
T2DM is associated with both macrovascular and microvascular complications. The macrovascular complications related to T2DM include coronary artery disease (CAD), peripheral arterial disease (PAD), and ischemic strokes promoted by diabetes-mediated endothelial dysfunction and atherosclerosis [9]. The microvascular complications caused by diabetes are diabetic nephropathy, retinopathy, and neuropathy [9]. While numerous physiological systems are implicated in the multi-factor pathogenesis of T2DM, this review specifically focuses on the RAS) antioxidant, inflammatory, and vitamin D receptor (VDR) signaling pathways [10–13]. The rationale for selecting these specific systems lies in the profound interconnectivity and their collective role in forming a constant cycle of metabolic dysfunction. Rather than acting in isolation, these four pathways intersect to drive the transition from early IR and β-cell exhaustion to chronic macro- and microvascular complications, such as nephropathy and retinopathy.
Distinguishing pathobiological mechanisms: obesity vs. T2DM
While obesity and T2DM share common features, such as chronic low-grade inflammation (meta-inflammation) and RAS overactivation, certain mechanisms are specific to the transition to a diabetic state [14, 15]. In individuals with obesity alone, compensatory hyperinsulinemia often maintains normoglycemia [16]. However, the progression to T2DM is defined by the progressive failure and apoptosis of pancreatic beta-cells.
Furthermore, once chronic hyperglycemia is established, it triggers “T2DM-specific” metabolic problems collectively known as glucotoxicity [17]. These include overactivation of the aldose reductase (polyol) pathway, increased formation of advanced glycation end-products (AGEs), and activation of protein kinase C (PKC). Unlike simple obesity, these hyperglycemia-driven pathways are the primary drivers of the microvascular damage (retinopathy, nephropathy, and neuropathy) that distinguishes the diabetic state. Thus, while the “metabolic loop” of RAS and oxidative stress (OS) begins in obesity, its role in beta-cell exhaustion and secondary glucotoxic pathways is what specifically defines T2DM pathogenesis [18].
| Methods | ▴Top |
A comprehensive literature search was conducted to identify relevant studies focusing on the pathophysiology and therapeutic strategies of T2DM, specifically regarding RAS, OS, inflammation, and vitamin D signaling. We searched electronic databases including PubMed, Google Scholar, Scopus, and Web of Science for articles published up to December 2023.
The search utilized combinations of the following keywords: “Type 2 Diabetes Mellitus,” “Renin-Angiotensin System,” “Oxidative Stress,” “Inflammatory Cytokines,” “Vitamin D Receptor (VDR),” and “Metabolic Pathways.” Inclusion criteria focused on peer-reviewed original research, clinical trials, and high-impact review articles published in English. Articles were selected based on their relevance to the molecular “cross-talk” between the targeted pathways. This systematic approach was employed to ensure a comprehensive and objective synthesis of the existing literature while minimizing selection bias.
| Role of RAS Pathway and T2DM | ▴Top |
RAS is an essential system in the body that aids in controlling blood pressure and fluid balance [19]. It is one of the most important mechanisms concerning hypertension, essential for regulating the dilation and constriction of blood vessels [20]. The systemic RAS mainly helps maintain blood pressure. In contrast, localized RAS components manage processes like OS, inflammation, and fibrosis [20]. The organs that possess their own local RAS systems are the kidneys, pancreas, intestines, respiratory system, and heart.
The RAS system is linked to more than just hypertension. It also plays a role in conditions like COVID-19 and T2DM [20]. There are two subsystems in RAS, classical and alternative subsystems [21]. The classical subsystem mainly consists of the participation of proteins, ACE, angiotensin II (Ang II), and angiotensin II receptor type 1 (AT1R) [21]. Thus, the classical subsystem can be called the ACE/Ang II/AT1R axis. On the other hand, the alternative subsystem consists mainly of the proteins, ACE2, angiotensin 1-7 (Ang 1-7), and MAS receptor [21]. Thus, it could be called the ACE2/Ang 1-7/MAS axis.
The hormone renin, of the RAS pathway, is obtained from the precursor, prorenin [22]. The renin/prorenin ratio (R/PR) is 10 in healthy individuals, whereas it is 40–50 in T2DM patients [21]. Thus, clearly, the RAS pathway is somehow related to T2DM development (Fig. 1). In addition to the above-mentioned case, there are various links of the RAS pathway with glucose metabolism [21].
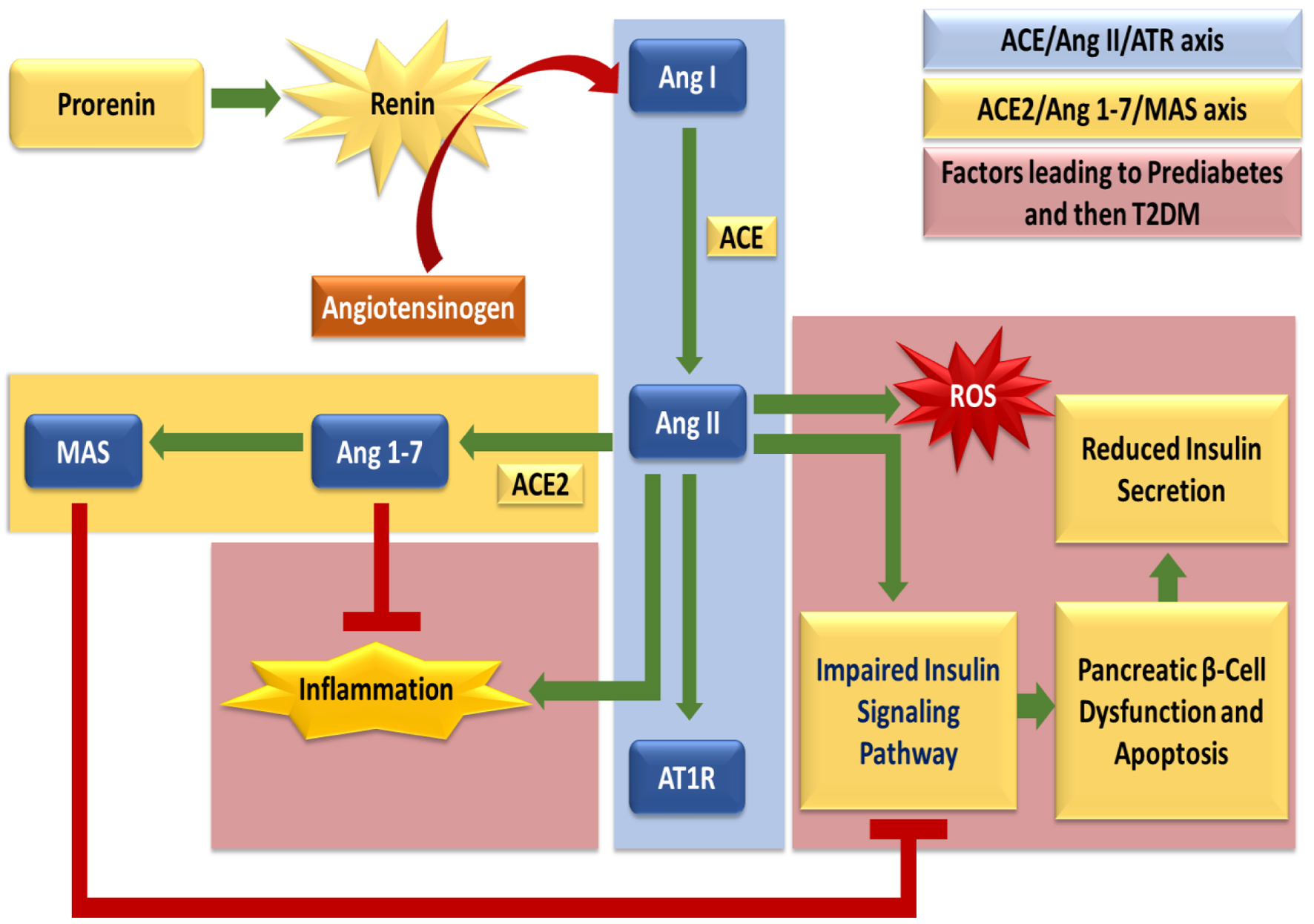 Click for large image | Figure 1. The diagram depicts the balance between the classical ACE/Ang II/AT1R axis and the protective ACE2/Ang 1-7/MAS axis of the renin-angiotensin system (RAS). Ang II promotes oxidative stress, inflammation, impaired insulin signaling, and β-cell dysfunction, thereby driving progression to T2DM, whereas ACE2/Ang 1-7/MAS counteract these effects and preserve metabolic homeostasis. |
RAS components play a significant role in the development of T2DM through various mechanisms. Ang II promotes IR by impairing insulin signaling pathways in target tissues like muscle, liver, and adipose tissue [23]. It contributes to pancreatic β-cell dysfunction and apoptosis, leading to reduced insulin secretion [22]. Ang II also induces inflammation and OS, which are key factors in the pathogenesis of T2DM [22]. The ACE2/Ang1-7/Mas axis has protective effects against T2DM (Fig. 1). Ang1-7 has anti-inflammatory, anti-fibrotic, and vasodilatory properties that can improve insulin sensitivity [22]. Activating the Mas receptor with Ang 1-7 can help reduce the harmful effects of Ang II on IR and pancreatic function. However, the local RAS in adipose tissue can lead to obesity, which is a major risk factor for T2DM [22]. Overactivation of RAS in adipose tissue leads to IR, glucose intolerance, and hypertension [24]. Angiotensinogen (AGT) expression is upregulated in obese individuals, contributing to increased Ang II levels and metabolic complications [25].
RAS genes and prediabetes
People with impaired glucose tolerance (IGT) and/or impaired fasting glucose (IFG) are referred to as having “prediabetes.” It suggests an increased susceptibility to T2DM and its complications [3]. In a study on Sprague-Dawley rats, the expression levels of RAS pathway components, such as renin, ACE, AT1R, and Ang II, were found to be higher in pre-diabetic rats than in non-pre-diabetic rats in most body tissues [26]. The ACE/Ang II/AT1R axis is upregulated in the cases of mild hyperglycemia, leading to increased NADPH oxidase (NOX) with downstream effects resulting in IR as seen in T2DM and prediabetes [27].
Adipose tissue secretes some anti-hyperglycemic hormones, such as omentin, adiponectin, and leptin, but with a high-calorie diet, their levels decrease, leading to over-nutrition and adiposity [27]. The relationship between over-nutrition and prediabetes risk factors (including upregulated RAS pathway) has been found to be bidirectional [27]. Mature adipocytes produce angiotensin 2 receptor (AT2R). This helps insulin-sensitive mature adipocytes grow and allows preadipocytes to differentiate (Fig. 2). As a result, it enhances triglyceride metabolism and insulin signaling [27].
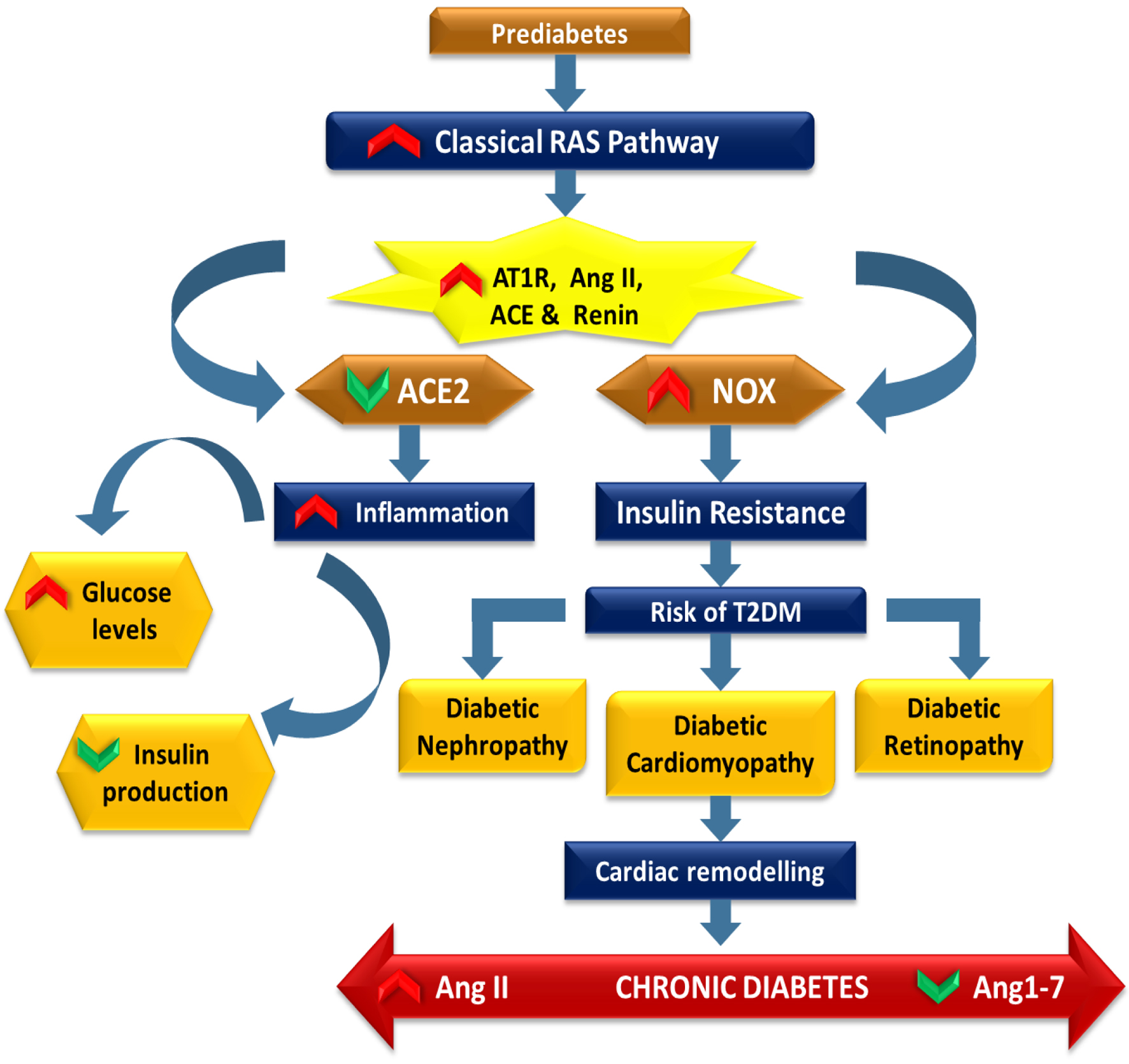 Click for large image | Figure 2. The diagram depicts the role of the classical RAS in the progression from prediabetes to T2DM. Upregulation of renin, ACE, Ang II, and AT1R enhances oxidative stress via NOX activation and promotes inflammation and insulin resistance. Reduced ACE2 activity weakens protective Ang 1-7 signaling, worsening glucose dysregulation. Persistent metabolic imbalance increases the risk of T2DM and complications, including nephropathy, cardiomyopathy, retinopathy, and cardiac remodeling, driven by dominance of the ACE/Ang II/AT1R axis. |
As implied earlier, renin, ACE, AT1R, and Ang II are higher in pre-diabetics. These three genes are related to the classical RAS pathway. Since, ACE2 enzyme is related with the conversion of the components of classical to alternate pathway, downregulation of which (thus, upregulation of the classical pathway) will lead to prediabetes and ultimately diabetes (Fig. 2). Moreover, ACE2 is the enzyme that obstructs Ang II signaling and thus, gene therapy of ACE2 reduces damaging effects of Ang II [28]. Although it is thought that ACE2 is an important player in the RAS pathway that affects the development of T2DM, not much is known about the low levels of ACE2 in diabetics. One of the possible mechanisms leading to low ACE2 in diabetes is its cleavage by a disintegrin and metalloprotease 17 (ADAM17) [29].
RAS genes and T2DM
In diabetic patients, the RAS pathway helps develop complications like diabetic nephropathy [30]. The RAS pathway contributes to renal fibrosis and glomerulosclerosis, which are major factors in the development of diabetic nephropathy [31]. Both tissue and circulatory RAS are hyperactivated in diabetic nephropathy, which causes local tissue damage via Ang II [32]. This activation contributes to persistent hypertension in diabetes individuals by raising blood pressure, causing proteinuria, and lowering glomerular filtration rate [30]. An early RAS inhibitor intervention in diabetic individuals with albuminuria is thought to slow the evolution of diabetic nephropathy [33].
RAS activation is also an important factor in the development of diabetic cardiomyopathy [34]. Ang II affects the heart negatively through IR and cardiac remodeling, but Ang II’s effects are opposed by Ang 1-7, which positively affects the cardiovascular system [34]. In the retina, abnormal ACE expression affects the blood flow and the structure of the vasculature, posing as an etiological factor for diabetic retinopathy [35]. Ang II and ACE raise vascular endothelial growth factors (VEGFs). VEGFs play a key role in the abnormal angiogenesis linked to diabetic retinopathy. The local expression of AT1R, Ang II, ACE, and renin has been seen to be elevated in people with diabetic retinopathy [36].
Ang II prevents the increased insulin secretion that occurs from pancreatic islets in hyperglycemic conditions [20]. Ang II also induces NOX production, which is responsible for the increase in OS by superoxide radical production [20]. Thus, Ang II is the major diabetogenic component of the RAS pathway (Fig. 2).
In summary, the overactivation of the localized RAS, particularly the Ang II/AT1R axis, is a primary driver of IR and vascular damage in T2DM. Therapeutic targeting of this system through ACE inhibitors or angiotensin II receptor blockers (ARBs) remains a cornerstone in preventing diabetic complications (Table 1).
 Click to view | Table 1. Summary of Key Pathways, Genes, and Pathophysiological Implications in T2DM |
| Role of Antioxidant Pathway and T2DM | ▴Top |
Reactive oxygen species (ROS) are generated primarily in the mitochondria as by-products of oxygen metabolism [36]. The most well-known ROS include superoxide, singlet oxygen, hydrogen peroxide, etc. [37]. Elevated ROS is related to various diseases like cardiovascular disorders (CVDs), cancer, and diabetes [38]. To fight ROS, the body has an antioxidant system. This includes enzymes like catalase (CAT), glutathione peroxidase (GPx), and superoxide dismutase (SOD). These enzymes turn harmful ROS into harmless products [39]. Furthermore, OS, caused by ROS overproduction and diminished antioxidant enzyme efficiency that disrupts the ROS-antioxidant balance, is a critical factor in T2DM pathogenesis [38, 40].
For xenobiotics that cause OS, the cytosol contains dimeric enzymes called glutathione-S-transferases (GSTs). These enzymes help attach xenobiotics to a water-soluble compound known as glutathione (GSH) [37]. The various isoforms of GSTs are named after Greek letters, alpha, kappa, mu, omega, pi, sigma, theta, and zeta [41]. The genes GST Theta 1 (GSTT1), GST Mu 1 (GSTM1), and GST Pi 1 (GSTP1) are involved in the development of T2DM and its various complications [37].
Antioxidant genes and prediabetes
ROS are countered by both endogenous (such as SOD, GPx, CAT) and exogenous (such as vitamins, carotenoids, polyphenols) antioxidants [42]. Decreased dietary (i.e., exogenous) antioxidants have been found to be associated with prediabetes [42]. Thus, dietary intake of antioxidant-rich foods is successful in controlling prediabetes. One such food, whose prediabetes progression-slowing properties have been reviewed earlier, is blueberry [43].
As mentioned before, there is IFG in prediabetes, meaning fasting glucose levels are higher than normal but below the diabetic range (Fig. 3). High glucose levels boost insulin production in β cells. However, as prediabetes advances to diabetes, ROS reduce gene expression. This leads to less insulin secretion and, eventually, β-cell apoptosis. This process pushes the body into a diabetic state [44].
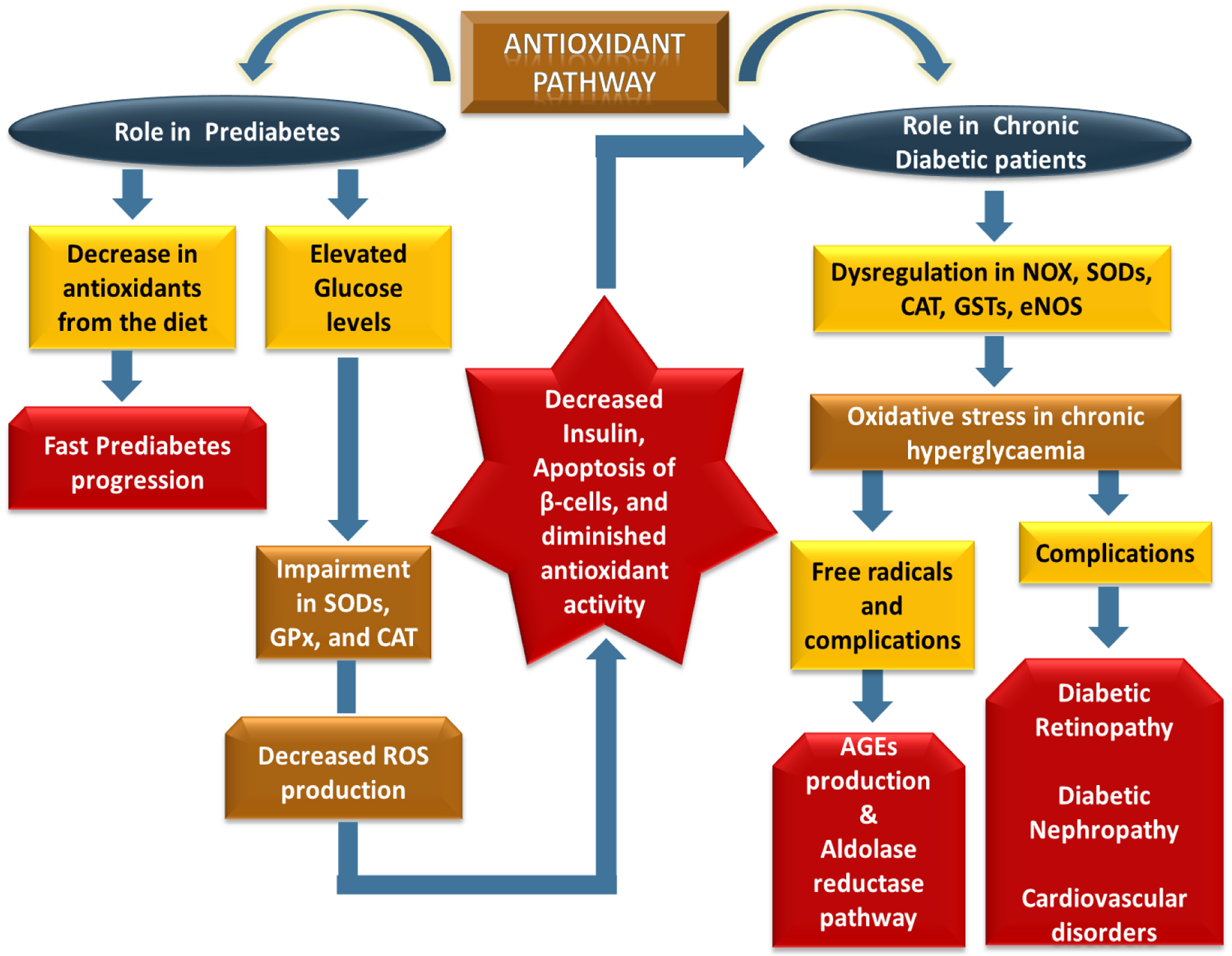 Click for large image | Figure 3. This schematic depicts progressive antioxidant pathway dysregulation from prediabetes to T2DM. In prediabetes, reduced dietary antioxidants and hyperglycemia impair SOD, GPx, and CAT activity, promoting β-cell dysfunction, reduced insulin secretion, and apoptosis. In diabetes, persistent hyperglycemia disrupts NOX, SODs, CAT, GSTs, and eNOS, increasing oxidative stress and free radical generation. This activates AGEs and the polyol pathway, contributing to diabetic retinopathy, diabetic nephropathy, and cardiovascular complications. |
The genetic makeup of key antioxidant enzymes influences the effectiveness of the antioxidant pathway in prediabetes. The genes SOD1, SOD2, and SOD3 make various SOD enzymes. Their single-nucleotide polymorphisms (SNPs) are associated with a higher risk of T2DM [34]. GPx-1, GPx-2, GPx-3, GPx-4, GSTT1, GSTM1, GSTP1, CAT, and eNOS (endothelial nitric oxide synthase; NOS-III) genes play roles in antioxidant defense and are linked to T2DM pathogenesis [38, 45, 46]. Variations in these genes may affect antioxidant capacity even in the pre-diabetic stage, influencing progression to diabetes.
Antioxidant genes and T2DM
Antioxidants prevent the development of diabetes-related complications in T2DM patients [47]. Some of the main issues faced by diabetic patients, such as diabetic nephropathy, retinopathy, CVDs, and stroke, are linked to OS [48]. The etiology of these issues involves both mitochondrial and non-mitochondrial sources of ROS, in addition to other stress factors, including inflammation and dyslipidemia [48]. According to Figure 3, excessive ROS production and diminished antioxidant activity in diabetes contribute to elevated OS levels [41, 49].
Chronic hyperglycemia causes both macrovascular and microvascular problems. It does this through OS, which creates harmful free radicals in the body [50]. Free radicals are key in starting and worsening diabetic complications. They disrupt three main pathways: the PKC pathway, the aldose reductase pathway, and AGE production [51]. For example, ROS makes the aldose reductase pathway faulty, leading to the accumulation of sorbitol in the retina, which results in diabetic retinopathy [51]. Diabetic neuropathy is another example. It affects half of the people with diabetes. This condition is caused by OS that leads to neuron death. Also, there is a reduced ability to repair and regenerate nerves [51].
At the genetic level, the diabetic state is characterized by the dysregulation of the same antioxidant genes discussed earlier (SOD, GPx, CAT, GSTs, eNOS), as well as by the upregulation of pro-oxidant genes (Table 1). For example, NOX, a promoter of OS, has several isoforms—NOX1, NOX2, NOX3, NOX4, NOX5, DUOX1, and DUOX2 [52]. Of these, NOX2 is notably upregulated in both T2DM mice and in the kidney walls of patients with diabetic nephropathy [52]. Additionally, besides eNOS, there are inducible nitric oxide synthase (iNOS/NOS-II) and neuronal nitric oxide synthase (nNOS/NOS-III). These also significantly impact conditions like hypercholesterolemia, hypertension, and diabetes [38, 52]. Dysregulation of these genes, either by mutation or altered expression, increases OS and the progression of diabetic complications.
To conclude, OS acts as a bridge between hyperglycemia and cellular damage. The exhaustion of the antioxidant defense system and the rise in ROS production create a toxic environment that accelerates β-cell failure and endothelial dysfunction.
| Role of Inflammatory Pathway and T2DM | ▴Top |
There are many risk factors associated with the development of T2DM, but the most important one is excess fat in the body, especially in the abdominal region [53]. T2DM induced by obesity is influenced mainly by two factors: IR and inflammation [54]. Interleukins are a group of cytokines (secreted proteins/signaling molecules) expressed by white blood cells (leukocytes) that mediate immune responses and play a definite role in the pathogenesis of T2DM [55]. People with T2DM have increased levels of inflammatory cytokines, the most studied of which is the pro-inflammatory cytokine, interleukin-6 (IL-6) [54]. IL-6 levels are higher in obese individuals and are known to enhance pancreatic β-cell apoptosis [56].
Anti-inflammatory cytokines play an important but context-dependent role in IR and the pathogenesis of T2DM. Interleukin-10 (IL-10) is generally considered protective due to its ability to suppress inflammation; however, evidence suggests that IL-10 may also modulate metabolic pathways in a manner that can inhibit diet-induced IR, indicating a complex and context-specific role in T2DM development [54, 57]. Similarly, interleukin-22 (IL-22), a member of the IL-10 cytokine family, has been implicated in glucose homeostasis, with plasma IL-22 levels showing a negative dose–response relationship with fasting plasma glucose (FPG), suggesting a potential protective effect under physiological conditions [58]. Beyond these cytokines, other anti-inflammatory mediators such as interleukin-1 (IL-1) influence key metabolic processes, including glucose regulation, blood pressure, iron metabolism, and bone remodeling. In addition, adiponectin levels and genetic variants have been consistently associated with an increased risk of T2DM, further highlighting the multifaceted role of inflammatory regulation in disease progression [59].
Cytokine genes and prediabetes
IR results when immune cells infiltrate adipocytes, contributing towards the development of prediabetes [60]. Among cytokines, IL-6 induces compensatory hyperinsulinemia and hyperglycemia. The insulin overload is sensed by T regulatory (T reg) cells present in adipocytes with their insulin receptors, leading to the release of anti-inflammatory IL-10 [60]. Usually, there is a converse relationship between the levels of serum IL-10 and hyperinsulinemia and IR. It was observed that levels of IL-10 were not elevated in pre-diabetics, but the same was not the case for T2DM patients [61].
IL-22, though promoting both pro- and anti-inflammatory cytokines, ultimately has beneficial effects against the development of prediabetes [62]. This is supported by findings that mice deficient in the IL-22 receptor were more prone to the development of IR and glucose intolerance [63]. In a study on the Han Chinese population, plasma IL-22 levels during fasting were found to be lower in pre-diabetic patients than in those with normal glucose levels [62].
At the genetic level, the genes encoding these cytokines—such as IL-6, IL-10, and IL-22—play a crucial role in modulating the inflammatory environment in prediabetes. Increased plasma IL-6, for example, is associated with IGT and serves as an etiological factor for the development of T2DM [55]. The IL-10 gene product protects the body. IL-10 effectively calms inflammatory responses from macrophages and lymphocytes. It also inhibits pro-inflammatory cytokines like IL-6 and tumor necrosis factor-alpha (TNF-α) [64]. The balance of these cytokine gene expressions determines progression or protection in the pre-diabetic state.
Cytokine genes and T2DM
Inflammation plays an important role not just in the onset of diabetes but also in the development of its complications [65]. The pro-inflammatory cytokine IL-6 is thought to promote the development of both micro- and macrovascular complications in T2DM [66]. Systemic increases in IL-6 levels are associated with complications like diabetic nephropathy [67], but only weakly influence the development of diabetic retinopathy [68]. Albuminuria is the gold standard for diagnosing diabetic nephropathy. However, plasma IL-6 levels above 10.5 pg/mL may help tell apart T2DM patients with nephropathy from those without it [65].
At the genetic level, T2DM shows increased pro-inflammatory cytokine genes. IL-6 is a key player. It affects glucose balance by acting on adipocytes, muscle cells, liver cells, neuroendocrine cells, and pancreatic β cells [69]. In contrast, anti-inflammatory cytokine genes such as IL-10 are often downregulated in T2DM, as plasma IL-10 levels are significantly reduced in these patients [70]. This reduction removes a critical brake on inflammation, allowing unchecked production of pro-inflammatory cytokines and exacerbating diabetic complications (Fig. 4). IL-22 has both pro- and anti-inflammatory effects. It can change how adipose tissue works by increasing local IL-1β production. This raises anti-adipogenic responses and worsens IR in obese patients with T2DM [58, 62, 71].
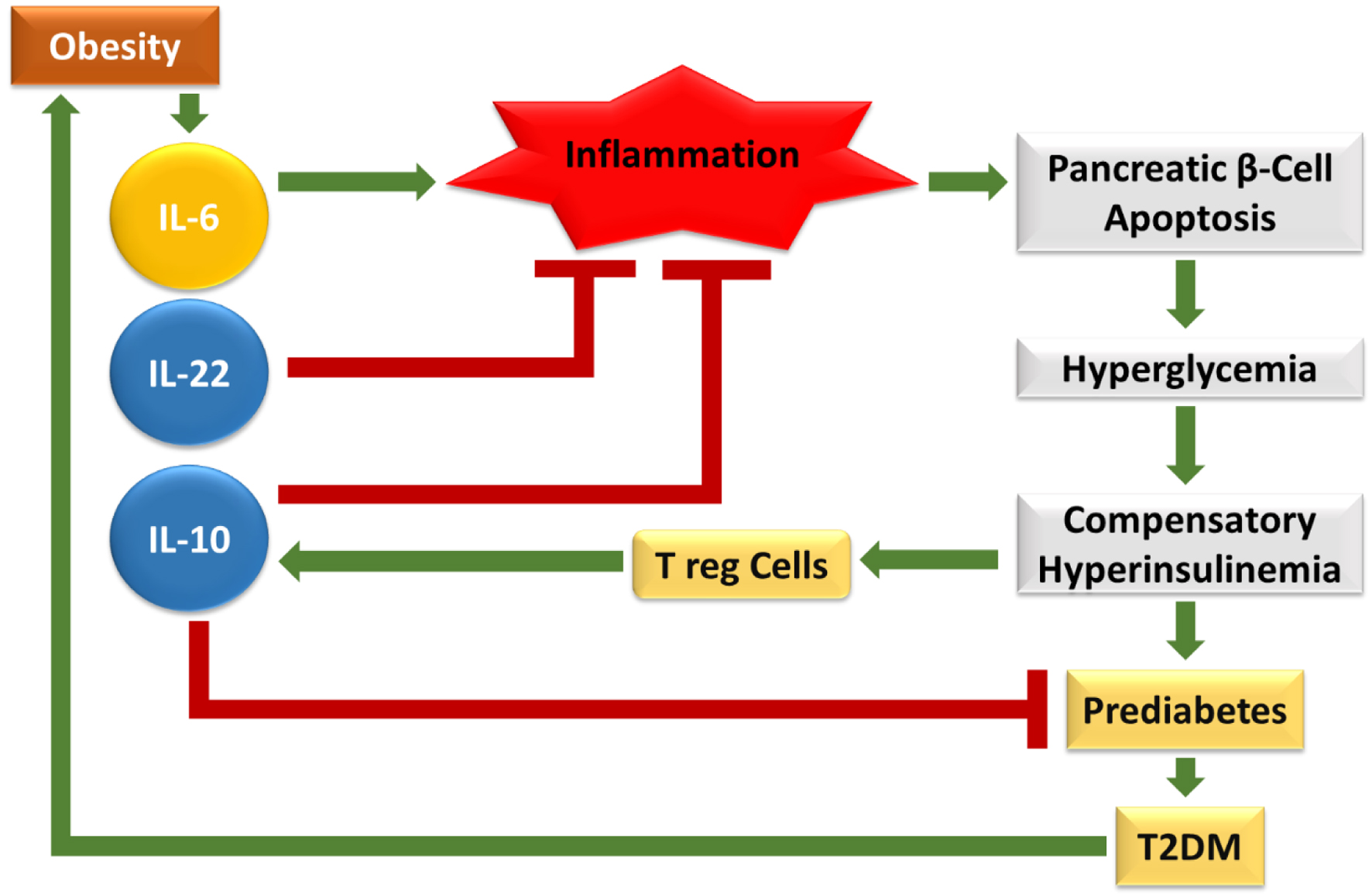 Click for large image | Figure 4. The given figure depicts how obesity-induced cytokine imbalance drives T2DM progression. Elevated IL-6 promotes chronic inflammation, leading to pancreatic β-cell apoptosis, hyperglycemia, compensatory hyperinsulinemia, prediabetes, and ultimately T2DM. Anti-inflammatory cytokines (IL-10, IL-22) and Treg cells counteract inflammation and protect β-cells. However, in obesity, pro-inflammatory dominance sustains immune dysregulation, accelerating β-cell dysfunction and metabolic deterioration. |
Overall, chronic low-grade inflammation, characterized by the elevation of pro-inflammatory cytokines like TNF-α and IL-6, perpetuates a cycle of insulin desensitization. Modulating these inflammatory pathways offers a promising secondary strategy for T2DM management (Table 1).
| Role of VDR and T2DM | ▴Top |
The expression of VDR is observed, though at variable levels, in all nucleated cells of humans [72]. Vitamin D has an important role in the secretion of insulin [73]. VDR variations have been linked with the development of glucose intolerance and insufficient insulin secretion, thus leading to T2DM development [74]. The VDRs expressed on pancreatic β cells are the ones at fault for the development of glucose intolerance and insulin insensitivity [73]. Since vitamin D is involved in maintaining the endocrine secretion of the pancreas, it has been seen that high vitamin D levels pose a protective mechanism against T2DM [75]. A deficiency of vitamin D is also a risk factor for obesity and, in turn, for diabetes [76].
VDR genes and prediabetes
Vitamin D is associated with the development of both IGT and IR, the hallmarks of prediabetes [77]. In the pancreatic β cells, there are both cytosolic and nuclear VDRs. According to Figure 5, the nuclear VDRs are activated by 1,25-dihydroxy vitamin D (1,25(OH)2D3), with the downstream signaling affecting secretion of insulin [77]. It is this secretion of insulin that, when impaired in the early pre-diabetic phase, ultimately leads to the diabetic state [77]. In pre-diabetics, supplementation with vitamin D has been found to sustain higher serum 1,25(OH)2D3 levels and thus, increases the chance of regressing to ideal glucose regulation [78].
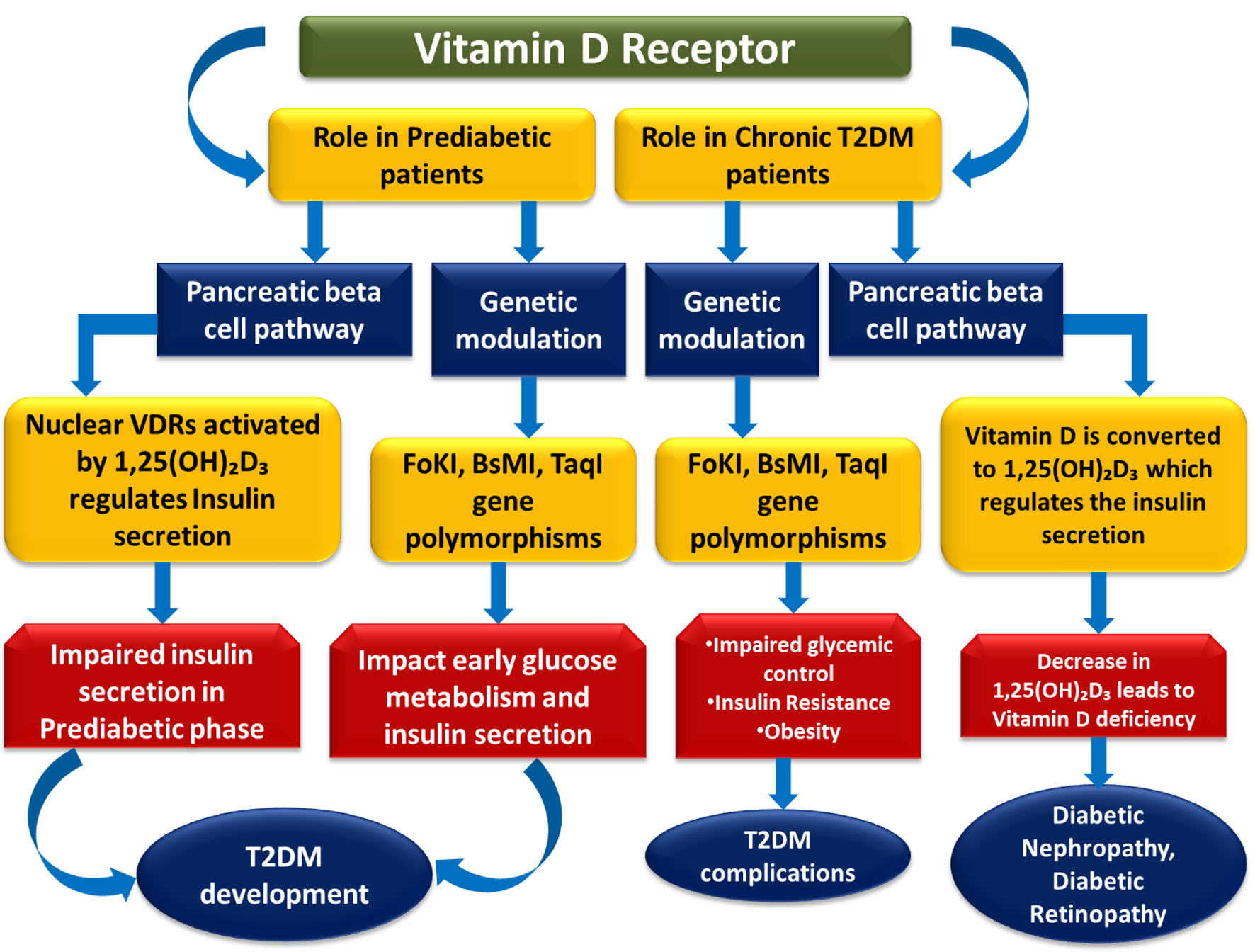 Click for large image | Figure 5. This figure depicts the role of VDR signaling in the progression from prediabetes to T2DM. Activation of nuclear VDRs by 1,25(OH)2D3 regulates pancreatic β-cell function and insulin secretion, while VDR polymorphisms (FokI, BsmI, TaqI) influence glucose metabolism. Impaired signaling contributes to insulin deficiency, insulin resistance, poor glycemic control, and vitamin D deficiency, ultimately leading to T2DM development and complications such as nephropathy and retinopathy. |
VDR gene polymorphisms may impact the risk of prediabetes. They change the VDR function in β cells, and how the body uses vitamin D. Most studies focus on T2DM. However, polymorphisms like FokI, BsmI, and TaqI may also impact early glucose metabolism and insulin secretion. This can increase the risk of prediabetes [75, 79, 80]. These variants may alter the receptor’s activity, affecting the efficiency of vitamin D signaling in maintaining glucose homeostasis (Fig. 5).
VDR genes and T2DM
VDRs are highly expressed in the kidneys, and they are the classic targets for 1,25(OH)2D3 [81]. The conversion of vitamin D into 1,25(OH)2D3 takes place in the kidneys, but in diabetic nephropathy, the kidneys are compromised, and enough 1,25(OH)2D3 production does not occur [81]. Vitamin D deficiency also plays a role in the development as well as progression of diabetic retinopathy, potentially by protecting retinal microvessels against diabetes-induced damage [82].
Gene variants of VDR, such as FokI, BsmI, and TaqI, have been studied across diverse populations for associations with T2DM and its complications [75, 79, 82]. Although these associations are sometimes population-specific and controversial, some studies report that these variants may influence glycemic control, IR, and even obesity among T2DM patients [83]. For example, BsmI was associated with IR among T2DM patients with poor glycemic control, and FokI with obesity [83]. Thus, VDR gene polymorphisms may contribute to the clinical heterogeneity of T2DM and its complications by altering the receptor’s function and downstream effects on glucose metabolism and tissue protection (Fig. 5).
In brief, vitamin D signaling through the VDR serves as a critical protective mechanism. By regulating insulin secretion and suppressing both RAS and inflammatory pathways, the VDR acts as a master metabolic modulator whose deficiency significantly worsens T2DM outcomes (Table 1).
| The Central Role of Liver in T2DM Pathogenesis | ▴Top |
The liver is a critical metabolic hub that maintains glucose homeostasis by balancing glucose uptake, glycogen synthesis, and gluconeogenesis [84]. In T2DM, hepatic IR leads to uncontrolled endogenous glucose production, which is a primary cause of fasting hyperglycemia. The physiological pathways discussed in this review, namely, RAS, OS, inflammation, and vitamin D signaling, all converge within the liver to worsen metabolic dysfunction [85].
For instance, localized hepatic RAS overactivation, particularly Ang II, has been shown to impair insulin signaling and promote hepatic fibrosis [86]. Simultaneously, chronic systemic inflammation driven by cytokines like IL-6 directly targets liver cells, further impairing their responsiveness to insulin [87]. Moreover, the liver is a key site for vitamin D metabolism and expresses VDRs; vitamin D deficiency is often linked with non-alcoholic fatty liver disease (NAFLD), which further compounds IR in T2DM patients [88]. By integrating these mechanisms, it becomes clear that hepatic dysregulation is not merely a consequence of T2DM but a primary regulator of its progression [84].
| Correlation Between RAS, Antioxidant, Inflammation, and Vitamin D Signaling Pathways in the Pathogenesis of T2DM | ▴Top |
The components of the RAS pathway also give rise to inflammation through various mechanisms. Ang II leads to the induction of immune cell differentiation and thus, the augmentation of cytokines that promote inflammation [89]. Ang II contributes to hypertension by acting through both immune cell differentiation and the induction of pro-inflammatory cytokines [90]. Apart from Ang II activating pro-inflammatory cytokines, reciprocally, they also lead to the enhancement of expression of RAS components [89]. Ang II either increases the number of inflammatory cytokines by promoting their differentiation from hematopoietic stem cells (HSCs), suggesting its pro-inflammatory activity. Alternatively, when Ang II binds to AT1R, it induces polarization of M2 macrophages, which in turn have anti-inflammatory activities [91]. Thus, Ang II could be considered as a “double-edged sword,” which has both pro- and anti-inflammatory effects [92].
From an inflammation point of view, the classical RAS subsystem/pathway can be called the “pro-inflammatory pathway” or the “pathological pathway,” whereas the alternative pathway could be called simply the “anti-inflammatory pathway” [93]. ACE2, Ang 1-7, aldosterone, renin, and Mas receptor are the other elements of the RAS pathway that impact inflammation [92]. These components use a different mechanism to offset the inflammatory effects of Ang II [92].
The ACE2/Ang 1-7/MAS receptor axis has protective anti-inflammatory qualities. This alternative pathway helps with: vasodilation, reducing cell proliferation, lowering inflammation, cutting OS, preventing hypertrophy, and reducing fibrosis [92].
The inflammatory responses can be stimulated by ROS through pro-inflammatory factors, protein kinases, gene expression, and transcription factors [94]. The participation of ROS is not just in the initiation of inflammation, but also in its progression and resolution [95]. During its progression stage, ROS participates in inflammation during “respiratory burst” [95]. Through the respiratory burst, small-sized microorganisms lead to the production of ROS inside cells. It inhibits the expression of interleukin-1 beta (IL-1β) and thus restricts the recruitment of new neutrophils. In contrast, large pathogens cause extracellular ROS to be released, which increases the expression of IL-1β and results in the formation of neutrophil clusters surrounding the pathogen [96]. As we have discussed in the above headings, Ang-II, a component of RAS, has both pro- and anti-inflammatory cytokine-promoting activity [92]. ROS also promotes pro-inflammatory cytokines, as implied before [36, 95]. Clearly, there are cross-links between RAS, antioxidants, and inflammatory pathways. Ang II leads to the formation of oxidant species, thus causing OS inside the vascular system [97]. Due to this OS, nuclear factor-kappa B (NF-κB) is activated, which causes the production of inflammatory cytokines [97].
The ligand for VDR, i.e., vitamin D, in its biologically active form, 1,25-dihydroxy vitamin D (1,25(OH)2D3), can inhibit renin gene expression [98], thus affecting the RAS pathway. Vitamin D influences the RAS pathway. It also helps protect against T2DM by reducing inflammation. This anti-inflammatory action has anti-diabetogenic effects, as we noted earlier. Apart from that, it has been seen that high serum 1,25(OH)2D3 concentrations were associated independently with a lower risk of microvascular complications of T2DM, such as diabetic nephropathy and neuropathy, though not with diabetic retinopathy [99, 100].
Apart from renin, the other components of RAS, whose expression is suppressed by vitamin D, are Ang II and ACE [76]. In the inflammatory pathway, vitamin D leads to a decrease in the production of T helper type 1 cells (Th1 cells) and thus suppresses the progression of inflammation by reducing pro-inflammatory cytokines [101]. One such pro-inflammatory cytokine that is downregulated by vitamin D is IL-6 [77].
Thus, the RAS pathway, VDR, and inflammation are interrelated as far as the pathogenesis of T2DM is concerned. Vitamin D also has antioxidant properties, partially mediated by VDR [102]. It is said to induce the expression of antioxidant enzymes like GPx and SOD and substrates like GSH [103]. Vitamin D also represses the expression of NOX, which is a critical promoter of OS [103]. The classical pathway, i.e., the ACE/Ang II/AT1R axis, is impacted by active vitamin D, i.e., 1,25(OH)2D3, at various steps. Even before the classical pathway commences, 1,25(OH)2D3 inhibits renin expression [98], and then 1,25(OH)2D3 also inhibits components of the axis like ACE and Ang II [76]. The same 1,25(OH)2D3 reduces pro-inflammatory cytokines and ROS. It does this by lowering Th1 cell production. Without this, Th1 cells would create more pro-inflammatory cytokines and ROS [76]. The pro-inflammatory cytokines are also produced by the classical RAS pathway, as Ang II promotes the differentiation of immune cells from HSCs, which are in turn responsible for the production of the pro-inflammatory cytokines [89]. Ang II boosts pro-inflammatory cytokines. It does this by increasing ROS, which then triggers cytokine production through NF-κB [97]. Although the classical pathway is mainly considered to be pro-inflammatory, it also has anti-inflammatory action through the polarization of M2 macrophages [91].
Vitamin D acts on VDRs only in its active form, i.e.1,25(OH)2D3, which is formed by the action of the enzyme 1α-hydroxylase [104]. When 1,25(OH)2D3 binds to VDR, the receptor pairs with retinoid X receptor (RXR). This action influences gene expression and impacts various non-genomic cellular pathways [103]. Research with VDR knock-out mice and 1α-hydroxylase-deficient mice shows that vitamin D reduces both circulating and tissue RAS pathways [104]. Other than RAS, the various cellular effects of active vitamin D include its antioxidant properties and its anti-inflammatory action [105]. The high anti-inflammatory and antioxidant properties of vitamin D are due to an increase in the formation of GSH [105]. The way by which vitamin D modulates GSH levels is by affecting glutathione peroxidase 1 (GPx1), SOD, and glutathione reductase (GR) expression [105].
Ang II can cause kidney damage by promoting inflammation. This happens when macrophages and T-lymphocytes enter the kidneys. These cells release pro-inflammatory cytokines, such as TNF-α and IL-6. In rats treated with Ang II, researchers saw an increase in the gene expression of these cytokines [106]. Inflammation plays a key role in diabetic nephropathy [106]. RAS interacts with innate immunity by helping express Toll-like receptors (TLRs) [107]. TLRs can act either through the myeloid factor 88 (MyD88) dependent or independent pathway, ultimately activating NF-κB, which ultimately leads to the production of chemokines and pro-inflammatory cytokines [107].
In summary, the information shows a complex web of pathways. Components activate and repress each other through genomic and non-genomic routes (Fig. 6). The ROS produced by Ang II action also acts through NF-κB to produce pro-inflammatory cytokines promoting inflammation [107]. NF-κB activation by Ang II is not just by the AT1R-NOX-ROS pathway described above, but also through upregulation of TLRs, bringing even components like MyD88 into the picture [107]. Ang II promotes inflammation, but it also has anti-inflammatory effects. Through its receptor, AT1R, it helps polarize M2 macrophages [91]. This makes Ang II a “double-edged sword” [92]. Vitamin D, the “sunshine vitamin,” also comes barging in this whole haphazard web through its active form, 1,25(OH)2D3 [108]. 1,25(OH)2D3 acts through VDR, which in turn exhibits its effects by dimerizing with RXR [104]. The VDR-RXR dimer acts through various genomic and non-genomic pathways [105]. One genomic pathway activated by the VDR-RXR dimer inhibits renin gene expression [98]. On the other hand, non-genomic pathways include inhibiting RAS components like ACE and Ang II [76]. They also reduce OS and inflammation by boosting GSH expression [105]. Thus, we can see that neither of the pathways acts in isolation, and all the pathways concerned with T2DM pathogenesis are tangled among themselves by a web of interactions (Fig. 6).
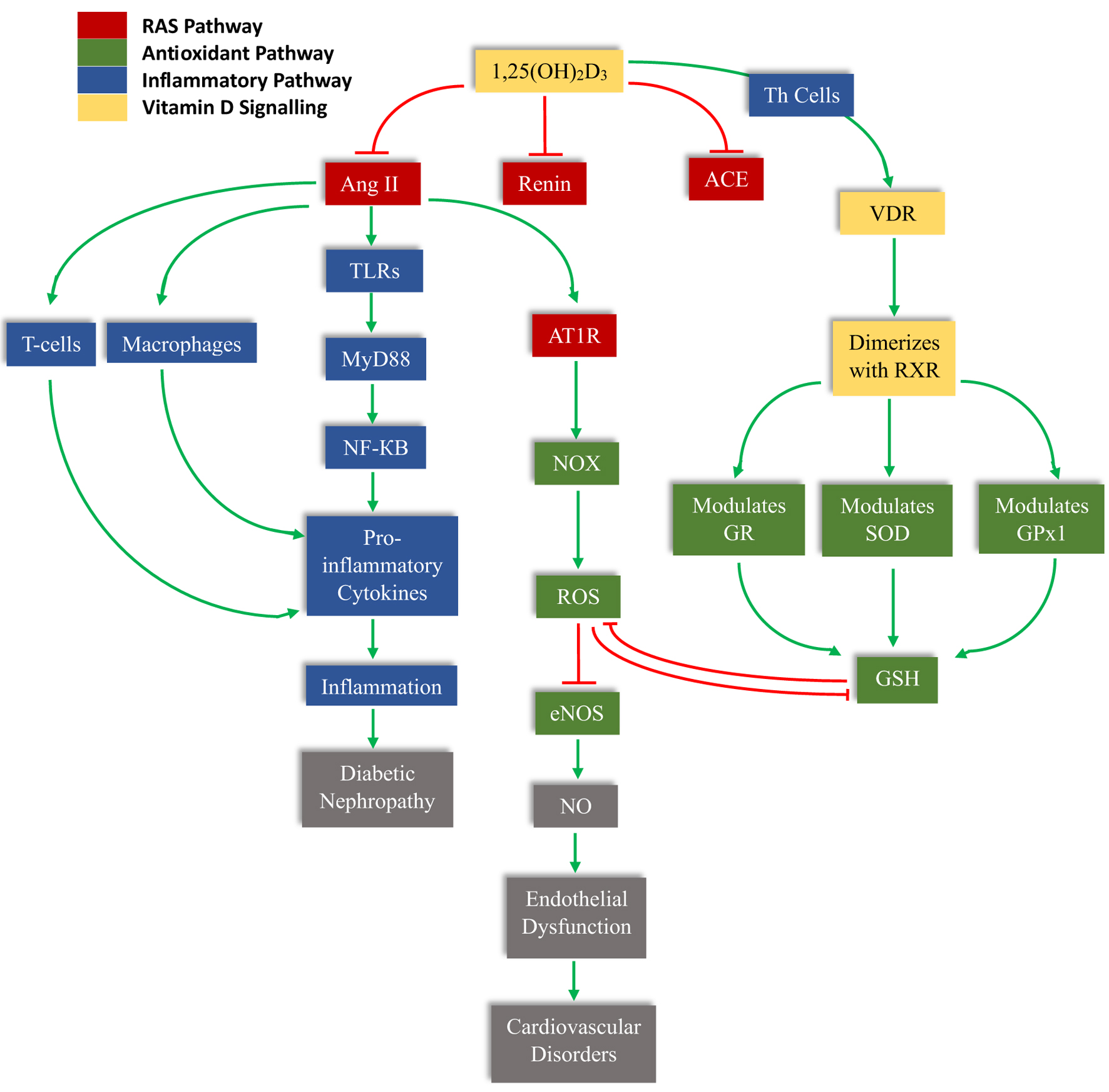 Click for large image | Figure 6. The schematic illustrates the central role of 1,25(OH)2D3 (calcitriol) in modulating RAS activation, immune signaling, oxidative stress, and antioxidant defense. Calcitriol suppresses renin and ACE expression, thereby reducing Ang II formation. Ang II activates AT1R, stimulating NADPH oxidase (NOX)–mediated ROS generation. Excess ROS decreases eNOS activity and nitric oxide (NO) bioavailability, leading to endothelial dysfunction and subsequent cardiovascular disorders. Ang II also activates TLRs/MyD88/NF-κB signaling in T cells and macrophages, enhancing pro-inflammatory cytokine production, inflammation, and progression to diabetic nephropathy. Concurrently, calcitriol binds to the VDR, which dimerizes with RXR and transcriptionally regulates antioxidant enzymes, including GR, SOD, and GPx1, thereby restoring glutathione (GSH) levels and attenuating ROS. Green arrows indicate activation or stimulation, while red blunt arrows represent inhibition. |
| Drugs and Therapies for T2DM Based on Various Pathways | ▴Top |
RAS pathway in T2DM treatment
T2DM is linked to low levels of ACE2 protein. Angiotensin-converting enzyme inhibitors (ACEis) can help. They downregulate the classical RAS pathway, easing the burden on the ACE2 enzyme. This makes ACEi effective for managing an overactive RAS pathway [49]. Other than ACEi, ARBs inhibit AT1R and thus also downregulate the overactive classical RAS pathway [49]. Importantly, sex acts as a biological variable in RAS regulation. Experimental and clinical evidence suggests that estrogen enhances ACE2 expression and promotes the protective ACE2/Ang 1-7/Mas receptor axis, whereas androgens may favor classical RAS activation. Consequently, men with T2DM may exhibit relatively greater RAS overactivity and cardiovascular risk, while postmenopausal women may lose estrogen-mediated RAS protection, influencing therapeutic responsiveness to ACEi/ARBs [109]. ACE2 is the receptor for the COVID-19 virus (SARS-CoV-2). Use of ACEi has been suggested to upregulate ACE2 expression, a mechanism that has raised concerns regarding a potential association with adverse outcomes in diabetic patients infected with COVID-19. Some observational evidence has reported a higher in-hospital mortality among diabetic COVID-19 patients receiving ACEi therapy [110]. However, these findings remain context-specific and inconclusive, as other factors such as disease severity, comorbidities, and treatment setting may influence outcomes. In contrast, ARBs were not found to significantly alter the mortality risk in this patient population [110], indicating differential effects within RAS-modulating therapies. Apart from all that, as already mentioned before, gene therapy of ACE2 reduces the damaging effects of Ang II, as well [28].
Albuminuria in T2DM is often associated with a high risk of development of CVDs and nephropathy [110]. Notably, sex differences have been reported in the progression of diabetic kidney disease, with men generally exhibiting faster progression to end-stage renal disease (ESRD), whereas women may demonstrate delayed onset but experience increased risk after menopause. These differences may partly reflect sex hormone–mediated modulation of intrarenal RAS activity. ACEi and ARBs are therefore widely used to reduce the risk of CVDs and nephropathy in T2DM patients with albuminuria. However, ACEi and ARBs can become less effective over time, as aldosterone levels may return to baseline after long-term RAS inhibition [110]. Combining mineralocorticoid receptor antagonists (MRAs) like eplerenone and spironolactone with RAS inhibitors can lower albuminuria and proteinuria significantly. However, MRAs may cause side effects such as hypotension, hyperkalemia, and gynecomastia [111], and these risks may vary by sex due to differences in hormonal milieu and body composition.
As already mentioned, albuminuria associated with T2DM leads to diabetic nephropathy, which has the risk of progressing into ESRD [112]. The various ACEi used in the treatment of diabetes associated albuminuria include drugs like captopril and ramipril, while ARBs studied for the same include losartan, irbesartan, VALsartan, olmesartan, and telmisartan [112]. Losartan also inhibits the formation of abnormal blood vessels in diabetic retinopathy patients [35]. The ramipril also reduces the risk of major cardiovascular events by 25% [35]. Direct renin inhibitors, such as aliskiren, have also been studied for potential benefit in diabetic nephropathy [112]. While current strategies effectively slow the progression of diabetic nephropathy, future therapeutic approaches may benefit from incorporating sex-stratified analyses combinations we could explore various combinations of ACEis, ARBs, and adjunctive therapies [63].
Antioxidant pathway in T2DM treatment
Prediabetic individuals and patients with early-stage T2DM often rely primarily on lifestyle modifications, whereas those with long-standing disease generally require hypoglycemic agents, with or without insulin, to enhance insulin secretion and improve insulin sensitivity [113].
Sex-related differences in body fat distribution, mitochondrial function, and OS burden may influence disease progression and treatment response, with visceral adiposity—more prevalent in men—being strongly associated with OS and IR. Several hypoglycemic agents, including metformin, also exhibit antioxidant properties that may contribute to their pleiotropic metabolic benefits [113]. Although metformin remains a first-line and effective therapy for T2DM, its use is frequently associated with gastrointestinal adverse effects [114], and some studies suggest potential sex-related variation in tolerability and pharmacokinetics [115].
In addition to glucose-lowering drugs, statins—widely prescribed for dyslipidemia—have been reported to exert antioxidant effects, potentially offering vascular protection in diabetic patients [116]. Exogenous antioxidants such as vitamins C and E are commonly included under the umbrella of “antioxidant therapy” and have been explored for their potential role in mitigating OS in T2DM [113]. However, despite strong mechanistic and preclinical evidence, clinical outcomes of antioxidant supplementation in T2DM remain inconsistent, with limited and controversial efficacy reported in large-scale human studies [113]. Similarly, several phytochemicals, including cyanidin, resveratrol, caffeic acid, anthocyanins, glabridin, and calycosin, have shown promise in experimental models by activating endogenous antioxidant defense pathways, but their therapeutic benefits in clinical diabetes management require further validation through well-designed clinical trials [113].
Inflammatory pathway in T2DM treatment
Metformin is not only an anti-hyperglycemic agent but also exerts anti-inflammatory effects [117]. It reduces inflammation primarily through activation of AMP kinase (AMPK), which decreases NF-κB phosphorylation and lowers pro-inflammatory cytokines such as IL-6 [118]. Sex hormones significantly influence inflammatory responses, with estrogen generally exerting anti-inflammatory effects and testosterone modulating cytokine production differently. Therefore, inflammatory burden and response to anti-inflammatory therapies in T2DM may vary between men and women [119].
Besides metformin, other drugs also help reduce inflammation in T2DM management. GLP-1 receptor agonists (GLP-1 RAs) enhance glucose-dependent insulin secretion, suppress glucagon, slow gastric emptying, and promote weight loss, while exerting anti-inflammatory effects by reducing C-reactive protein (CRP), IL-6, and TNF-α levels [120]. Sodium-glucose cotransporter 2 (SGLT2) inhibitors promote glycosuria, natriuresis, and caloric loss for glycemic control and cardiovascular/renal protection, alongside lowering inflammatory markers like ferritin, CRP, leptin, and plasminogen activator inhibitor-1 (PAI-1) [121]. Dipeptidyl peptidase-4 (DPP-4) inhibitors prolong incretin activity to improve postprandial glycemia with low hypoglycemia risk and demonstrate anti-inflammatory benefits by decreasing high-sensitivity C-reactive protein (hs-CRP), TNF-α, IL-6, and IL-1β [122]. These agents offer cardioprotective advantages beyond glycemic control, positioning GLP-1 RAs and SGLT2 inhibitors as preferred options in patients with atherosclerotic CVDs, heart failure, or chronic kidney disease per guidelines, while DPP-4 inhibitors serve as effective add-ons [120, 121, 123]. Emerging evidence suggests that cardiovascular and renal benefits of GLP-1 RAs and SGLT2 inhibitors are observed in both sexes, although absolute risk reductions may differ due to baseline risk variation [124].
Many phytochemicals with anti-inflammatory activity also show promise as possible therapeutic agents for the treatment of T2DM in the future [125]. Alkaloids like berberine also lead to a decrease in the transcriptional activity of NF-κb [126]. In mice with diabetes, treatment with berberine showed downregulation of pro-inflammatory cytokines, including IL-6 [126]. Flavonoids like curcumin also work through decreasing NF-κB activity, among other mechanisms [127]. Curcumin admixture in the diet of obese mice showed improvement against glucose intolerance [127]. Given known sex differences in cardiovascular risk profiles, careful consideration of sex-specific risk–benefit balance is warranted.
But not all anti-inflammatory drugs/agents are beneficial for diabetics. The main over-the-counter “anti-inflammatory” drugs are non-steroidal anti-inflammatory drugs (NSAIDs). These can increase the risk of heart failure in patients with T2DM [128]. These drugs work by inhibiting cyclooxygenase (COX)-1 and -2 and thus work mechanistically differently than the above-mentioned inflammatory pathway [129].
VDR in T2DM treatment
We have talked about VDR’s role in insulin secretion. This shows how proper vitamin D signaling can help prevent T2DM. Sex differences in vitamin D metabolism, body fat distribution, and hormonal interactions may affect VDR-mediated metabolic responses. Theoretically, we can also make out how the activation of VDR can be done by either its natural ligand or other various agonists. Thus, these ligands could also act as possible drugs/therapy for the prevention and/or treatment of T2DM. Alfacalcidiol, a vitamin D analogue and VDR agonist, significantly lowers blood glucose levels. It also decreases fasting serum insulin and reduces IR in rats [130]. Not only that, but alfacalcidiol also acted synergistically with metformin as an anti-hyperglycemic agent [130].
Another potential intervention explored is vitamin D supplementation. Evidence suggests that prediabetics receiving medium to high doses of vitamin D exhibited a reduced risk of progression to T2DM, whereas no comparable benefit was observed with lower doses [130]. Short-term intervention studies have reported modest improvements in glycemic parameters following vitamin D supplementation; however, findings from long-term trials remain inconsistent. Several studies demonstrated no significant effect on hemoglobin A1c (HbA1c) levels over extended follow-up periods, and in instances where HbA1c reductions were reported, corresponding changes in FPG were not evident [131]. These mixed outcomes may be attributed to substantial variability in study design, including differences in baseline vitamin D status, dosage regimens, duration of supplementation, population characteristics and importantly, sex distribution across studies, and glycemic endpoints assessed. Collectively, the available evidence indicates that while vitamin D supplementation may offer short-term metabolic benefits in selected populations, its long-term efficacy in sustained glycemic control and T2DM prevention remains inconclusive [131].
When thinking about vitamin D for diabetes, it is important to consider the risk of overdose. Too much vitamin D can cause side effects. These include high blood pressure, heart rhythm problems, and increased urination. It may also lead to high calcium levels, possible kidney failure, dehydration, and increased thirst [108]. Other signs of vitamin D overdose can include: abdominal pain, pancreatitis, vomiting, confusion, depression, and coma [108]. Thus, while VDR-targeted therapies remain promising, future clinical trials should systematically evaluate sex as a biological variable to optimize dosing, efficacy, and safety in T2DM management.
| Conclusion | ▴Top |
In conclusion, T2DM emerges from a complex and self-perpetuating interaction among four major physiological pathways: the RAS, OS-antioxidant imbalance, chronic inflammation, and vitamin D/VDR signaling [10, 13]. While each pathway independently contributes to insulin resistance, β-cell dysfunction, and vascular injury, their extensive molecular cross-talk amplifies metabolic derangements and accelerates progression from prediabetes to overt T2DM and its complications [75, 90, 91, 97, 104, 106, 107, 132, 133]. Therapeutic strategies targeting these interconnected mechanisms offer complementary benefits. RAS-modulating agents such as ACEis and ARBs remain central in limiting vascular and renal complications [134], and ACE2-based gene therapy represents a potential future approach [28]. Antioxidant-focused interventions, including dietary antioxidants and phytochemicals, may mitigate OS, while established agents such as metformin exert additional antioxidant and anti-inflammatory effects [135]. Similarly, vitamin D supplementation and VDR agonists may help restore impaired vitamin D signaling and modulate RAS and inflammatory pathways [129, 130]. However, these interventions are not without limitations or adverse effects, and therapeutic responses may vary due to biological factors such as sex, baseline metabolic status, and disease stage [107, 110, 113]. Therefore, well-designed, large-scale clinical trials—incorporating pathway-based and sex-stratified analyses—are essential to validate these adjunctive strategies and to develop more precise, mechanism-driven approaches for the long-term management of T2DM.
Acknowledgments
Komal Awasthi, Pratyush Singh, and Ashwin Kumar Shukla acknowledge fellowships from the University of Lucknow, Lucknow, UGC, New Delhi, and the Research and Development Scheme (R&D), Government of Uttar Pradesh, Lucknow, India, respectively. The authors also acknowledge the Department of Biotechnology (DBT), Department of Science and Technology (DST), Indian Council of Medical Research (ICMR), New Delhi, India, and Centre of Excellence, Higher Education, for research grants to the Molecular and Human Genetics Lab, Department of Zoology, University of Lucknow, Lucknow.
Financial Disclosure
No funding was received to assist with the preparation of this manuscript.
Conflict of Interest
The authors have no conflict of interest.
Author Contributions
Komal Awasthi: conceptualization, writing- original draft, literature review, visualization, validation. Pratyush Singh: writing- original draft, literature review. Ashwin Kumar Shukla: literature review, visualization, review, and editing. Kauser Usman: supervision and clinical input. Monisha Banerjee: conceptualization, supervision, review, and final editing. All authors reviewed and approved the final version of the manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
T2DM: type 2 diabetes mellitus; IR: insulin resistance; IDF: International Diabetes Federation; DALYs: disability-adjusted life years; CAD: coronary artery disease; PAD: peripheral arterial disease; RAS: renin-angiotensin system; VDRs: vitamin D receptors; AGEs: advanced glycation end-products; PKC: protein kinase C; OS: oxidative stress; COVID-19: coronavirus disease 2019; ACE: angiotensin-converting enzyme; Ang II: angiotensin II; AT1R: angiotensin II receptor type 1; ACE2: angiotensin converting enzyme 2; Ang 1-7: angiotensin 1-7; R/PR ratio: renin/prorenin ratio; AGT: angiotensinogen; IGT: impaired glucose tolerance; IFG: impaired fasting glucose; NOX: NADPH oxidase; AT2R: angiotensin II receptor type 2; ADAM 17: a disintegrin and metalloprotease 17; VEGFs: vascular endothelial growth factors; ROS: reactive oxygen species; CVDs: cardiovascular disorders; CAT: catalase; GPx: glutathione peroxidase; SOD: superoxide dismutase; GSTs: glutathione-S-transferases; GSH: glutathione; GSTT1: GST Theta 1; GSTM1: GST Mu 1; GSTP1: GST Pi 1; SNPs: single nucleotide polymorphisms; eNOS/NOS-III: endothelial nitric oxide synthase; iNOS/NOS-I: inducible nitric oxide synthase; nNOS/NOS-II: neural nitric oxide synthase; IL-1: interleukin-1; IL-6: interleukin-6; IL-10: interleukin-10; IL-22: interleukin-22; FPG: fasting plasma glucose; T reg cells: T regulatory cells; 1,25(OH)2D3: 1, 25-dihydroxy vitamin D; NAFLD: non-alcoholic fatty liver disease; HSCs: hematopoietic stem cells; IL-1β: interleukin-1 beta; NF-κB: nuclear factor-kappa B; Th1 cells: T helper type 1 cells; RXR: retinoid X receptor; GPx1: glutathione peroxidase 1; GR: glutathione reductase; TLRs: Toll-like receptors; MyD88: myeloid factor 88; ACEis: ACE inhibitors; ARBs: angiotensin II receptor blockers; MRAs: mineralocorticoid receptor antagonists; ESRD: end-stage renal disease; AMPK: AMP kinase; GLP-1 RAs: GLP-1 receptor agonists; CRP: C-reactive protein; TNF-α: tumor necrosis factor-alpha; SGLT2: sodium-glucose cotransporter 2; PAI-1: plasminogen activator inhibitor-1; DPP-4: dipeptidyl peptidase-4; hs-CRP: high-sensitivity C-reactive protein; NSAIDs: non-steroidal anti-inflammatory drugs; COX: cyclooxygenase; HbA1c: hemoglobin A1c
| References | ▴Top |
- Shukla AK, Shamsad A, Kushwah AS, Singh S, Usman K, Banerjee M. CD36 gene variant rs1761667 (G/A) as a biomarker in obese type 2 diabetes mellitus cases. Egypt J Med Hum Genet. 2024; 25:9.
doi - Venkatesan SK. Classification of diabetes mellitus: a review. Int J Diabetes Metab. 2021;1:20-22.
- International Diabetes Federation. IDF Diabetes Atlas, 10th edition Brussels, Belgium: 2021. Available at: https://www.diabetesatlas.org.
- International Diabetes Federation. IDF Diabetes Atlas, 11th edition Brussels, Belgium: 2025. Available at: https://www.diabetesatlas.org.
- G. B. D. Diseases Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396(10258):1204-1222.
doi pubmed - Shukla AK, Awasthi K, Singh P; Usman K, Banerjee M. Assessment of diabetes risk in Lucknow and surrounding areas using the Indian Diabetes Risk Score (IDRS): a population-based study. Int J Diabetes Dev Ctries. 2025.
doi - Shukla AK, Awasthi K, Usman K, Banerjee M. Circulating MiRNAs targeting antioxidant genes: potential biomarkers for type 2 diabetes mellitus. Mol Biol Rep. 2025;52(1):648.
doi pubmed - DeFronzo RA, Ferrannini E. Pathogenesis of type 2 diabetes mellitus. Med Clin N Am. 2021;105:231-250.
doi - Zakir M, Ahuja N, Surksha MA, Sachdev R, Kalariya Y, Nasir M, Kashif M, et al. Cardiovascular complications of diabetes: from microvascular to macrovascular pathways. Cureus. 2023;15(9):e45835.
doi pubmed - Ribeiro-Oliveira A, Jr., Nogueira AI, Pereira RM, Boas WW, Dos Santos RA, Simoes e Silva AC. The renin-angiotensin system and diabetes: an update. Vasc Health Risk Manag. 2008;4(4):787-803.
pubmed - Al-Mansoori L, Al-Jaber H, Prince MS, Elrayess MA. Role of inflammatory cytokines, growth factors and adipokines in adipogenesis and insulin resistance. Inflammation. 2022;45(1):31-44.
doi pubmed - Singh A, Kukreti R, Saso L, Kukreti S. Mechanistic insight into oxidative stress-triggered signaling pathways and type 2 diabetes. Molecules. 2022;27(3):950.
doi pubmed - Mitri J, Pittas AG. Vitamin D and diabetes. Endocrinol Metab Clin North Am. 2014;43(1):205-232.
doi pubmed - Yadav U, Kumar N, Sarvottam K. Role of obesity related inflammation in pathogenesis of peripheral artery disease in patients of type 2 diabetes mellitus. J Diabetes Metab Disord. 2023;22(1):175-188.
doi pubmed - Varra FN, Varras M, Varra VK, Theodosis-Nobelos P. Molecular and pathophysiological relationship between obesity and chronic inflammation in the manifestation of metabolic dysfunctions and their inflammation-mediating treatment options (Review). Mol Med Rep. 2024;29(6):95.
doi pubmed - Inaishi J, Saisho Y. Beta-cell mass in obesity and type 2 diabetes, and its relation to pancreas fat: a mini-review. Nutrients. 2020;12(12):3846.
doi pubmed - Yamaguchi H, Nagai R. Insights from the fructose-derived product glucoselysine: Revisiting the polyol pathway in diabetic complications. J Diabetes Investig. 2025;16(4):569-577.
doi pubmed - Dinic S, Arambasic Jovanovic J, Uskokovic A, Mihailovic M, Grdovic N, Tolic A, Rajic J, et al. Oxidative stress-mediated beta cell death and dysfunction as a target for diabetes management. Front Endocrinol (Lausanne). 2022;13:1006376.
doi pubmed - Song Z, Xu Y, Bao L, Zhang L, Yu P, Qu Y, Zhu H, et al. From SARS to MERS, thrusting coronaviruses into the spotlight. Viruses. 2019;11(1):59.
doi pubmed - Shukla AK, Banerjee M. Angiotensin-converting-enzyme 2 and renin-angiotensin system inhibitors in COVID-19: an update. High Blood Press Cardiovasc Prev. 2021;28(2):129-139.
doi pubmed - Vargas Vargas RA, Varela Millan JM, Fajardo Bonilla E. Renin-angiotensin system: Basic and clinical aspects-a general perspective. Endocrinol Diabetes Nutr. 2022;69(1):52-62.
doi pubmed - Kanugula AK, Kaur J, Batra J, Ankireddypalli AR, Velagapudi R. Renin-angiotensin system: updated understanding and role in physiological and pathophysiological states. Cureus. 2023;15(6):e40725.
doi pubmed - Saiki A, Ohira M, Endo K, Koide N, Oyama T, Murano T, Watanabe H, et al. Circulating angiotensin II is associated with body fat accumulation and insulin resistance in obese subjects with type 2 diabetes mellitus. Metabolism. 2009;58(5):708-713.
doi pubmed - Schutten MT, Houben AJ, de Leeuw PW, Stehouwer CD. The link between adipose tissue renin-angiotensin-aldosterone system signaling and obesity-associated hypertension. Physiology (Bethesda). 2017;32(3):197-209.
doi pubmed - Giacchetti G, Faloia E, Sardu C, Camilloni MA, Mariniello B, Gatti C, Garrapa GG, et al. Gene expression of angiotensinogen in adipose tissue of obese patients. Int J Obes Relat Metab Disord. 2000;24(Suppl 2):S142-143.
doi pubmed - Mkhize BC, Mosili P, Ngubane PS, Sibiya NH, Khathi A. Diet-induced prediabetes: Effects on the activity of the renin-angiotensin-aldosterone system in selected organs. J Diabetes Investig. 2022;13(5):768-780.
doi pubmed - Cassandra Mkhize B, Mosili P, Sethu Ngubane P, Khathi A. The relationship between adipose tissue RAAS activity and the risk factors of prediabetes: a systematic review and meta-analysis. Adipocyte. 2023;12(1):2249763.
doi pubmed - Bindom SM, Hans CP, Xia H, Boulares AH, Lazartigues E. Angiotensin I-converting enzyme type 2 (ACE2) gene therapy improves glycemic control in diabetic mice. Diabetes. 2010;59(10):2540-2548.
doi pubmed - Shukla AK, Awasthi K, Usman K, Banerjee M. Role of renin-angiotensin system/angiotensin converting enzyme-2 mechanism and enhanced COVID-19 susceptibility in type 2 diabetes mellitus. World J Diabetes. 2024;15(4):606-622.
doi pubmed - Bahreini E, Rezaei-Chianeh Y, Nabi-Afjadi M. Molecular mechanisms involved in intrarenal renin-angiotensin and alternative pathways in diabetic nephropathy - a review. Rev Diabet Stud. 2021;17(1):1-10.
doi pubmed - Campbell DJ. Clinical relevance of local Renin Angiotensin systems. Front Endocrinol (Lausanne). 2014;5:113.
doi pubmed - Peti-Peterdi J, Kang JJ, Toma I. Activation of the renal renin-angiotensin system in diabetes—new concepts. Nephrol Dial Transplant. 2008;23(10):3047-3049.
doi pubmed - Xie X, Liu Y, Perkovic V, Li X, Ninomiya T, Hou W, Zhao N, et al. Renin-angiotensin system inhibitors and kidney and cardiovascular outcomes in patients with CKD: a Bayesian network meta-analysis of randomized clinical trials. Am J Kidney Dis. 2016;67(5):728-741.
doi pubmed - Batista JPT, Faria AOV, Ribeiro TFS, Simoes ESAC. The role of renin-angiotensin system in diabetic cardiomyopathy: a narrative review. Life (Basel). 2023;13(7).
doi pubmed - Rahimi Z, Moradi M, Nasri H. A systematic review of the role of renin angiotensin aldosterone system genes in diabetes mellitus, diabetic retinopathy and diabetic neuropathy. J Res Med Sci. 2014;19(11):1090-1098.
pubmed - Darenskaya MA, Kolesnikova LI, Kolesnikov SI. Oxidative stress: pathogenetic role in diabetes mellitus and its complications and therapeutic approaches to correction. Bull Exp Biol Med. 2021;171(2):179-189.
doi pubmed - Sies H, Jones DP. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol. 2020;21(7):363-383.
doi pubmed - Verma S, Sagar N, Vats P, Shukla KN, Abbas M, Banerjee M. Antioxidant enzyme levels as markers for type 2 diabetes mellitus. Int J Bioassays. 2013;2:685-690.
- Ozougwu JC. The role of reactive oxygen species and antioxidants in oxidative stress. Int J Res. 2016;3:1-8.
- Shukla AK, Awasthi K, Usman K, Banerjee M. Superoxide dismutase gene expression and promoter methylation as biomarkers for type 2 diabetes mellitus. Hum Gene. 2025;45:201427.
doi - Vats P, Kushwah AS, Banerjee M. Association of antioxidant gene variants with type 2 diabetes mellitus in different ethnic groups. Eur J Biomed Pharm Sci. 2017;4:290-298
- Cyunczyk M, Zujko ME, Jamiolkowski J, Zujko K, Lapinska M, Zalewska M, Kondraciuk M, et al. Dietary total antioxidant capacity is inversely associated with prediabetes and insulin resistance in Bialystok PLUS population. Antioxidants (Basel). 2022;11(2):283.
doi pubmed - Nunes S, Vieira P, Gomes P, Viana SD, Reis F. Blueberry as an attractive functional fruit to prevent (Pre)diabetes progression. Antioxidants (Basel). 2021;10(8):1162.
doi pubmed - Kaneto H, Katakami N, Matsuhisa M, Matsuoka TA. Role of reactive oxygen species in the progression of type 2 diabetes and atherosclerosis. Mediators Inflamm. 2010;2010:453892.
doi pubmed - Banerjee M, Vats P. Reactive metabolites and antioxidant gene polymorphisms in Type 2 diabetes mellitus. Redox Biol. 2014;2:170-177.
doi pubmed - Liu LS, Wang D, Tang R, Wang Q, Zheng L, Wei J, Li Y, et al. Individual and combined effects of the GSTM1, GSTT1, and GSTP1 polymorphisms on type 2 diabetes mellitus risk: A systematic review and meta-analysis. Front Genet. 2022;13:959291.
doi pubmed - Zhong O, Hu J, Wang J, Tan Y, Hu L, Lei X. Antioxidant for treatment of diabetic complications: A meta-analysis and systematic review. J Biochem Mol Toxicol. 2022;36(6):e23038.
doi pubmed - Iacobini C, Vitale M, Pesce C, Pugliese G, Menini S. Diabetic complications and oxidative stress: a 20-year voyage back in time and back to the future. Antioxidants (Basel). 2021;10(5).
doi pubmed - Shukla AK, Awasthi K, Usman K, Banerjee M. Pharmacogenetics of Metformin monotherapy: GSTM1/T1 polymorphisms and T2DM risk. Curr Pharmacogenomics Pers Med. 2024;21:100-112.
doi - Caturano A, D'Angelo M, Mormone A, Russo V, Mollica MP, Salvatore T, Galiero R, et al. Oxidative stress in type 2 diabetes: impacts from pathogenesis to lifestyle modifications. Curr Issues Mol Biol. 2023;45(8):6651-6666.
doi pubmed - Oguntibeju OO. Type 2 diabetes mellitus, oxidative stress and inflammation: examining the links. Int J Physiol Pathophysiol Pharmacol. 2019;11(3):45-63.
pubmed - Tabatabaei-Malazy O, Khodaeian M, Bitarafan F, Larijani B, M MA. Polymorphisms of antioxidant genes as a target for diabetes management. Int J Mol Cell Med. 2017;6(3):135-147.
doi pubmed - Blair M. Diabetes mellitus review. Urol Nurs. 2016;36.
doi - Saxena M, Srivastava N, Banerjee M. Association of IL-6, TNF-alpha and IL-10 gene polymorphisms with type 2 diabetes mellitus. Mol Biol Rep. 2013;40(11):6271-6279.
doi pubmed - Velikova TV, Kabakchieva PP, Assyov YS, Georgiev Tcapital A C. Targeting inflammatory cytokines to improve type 2 diabetes control. Biomed Res Int. 2021;2021:7297419.
doi pubmed - Hong EG, Ko HJ, Cho YR, Kim HJ, Ma Z, Yu TY, Friedline RH, et al. Interleukin-10 prevents diet-induced insulin resistance by attenuating macrophage and cytokine response in skeletal muscle. Diabetes. 2009;58(11):2525-2535.
doi pubmed - Cheng M, Zhou Y, Wang B, Mu G, Ma J, Zhou M, Wang D, et al. IL-22: A potential mediator of associations between urinary polycyclic aromatic hydrocarbon metabolites with fasting plasma glucose and type 2 diabetes. J Hazard Mater. 2021;401:123278.
doi pubmed - Banerjee M, Saxena M. Genetic polymorphisms of cytokine genes in type 2 diabetes mellitus. World J Diabetes. 2014;5(4):493-504.
doi pubmed - Grossmann V, Schmitt VH, Zeller T, Panova-Noeva M, Schulz A, Laubert-Reh D, Juenger C, et al. Profile of the immune and inflammatory response in individuals with prediabetes and type 2 diabetes. Diabetes Care. 2015;38(7):1356-1364.
doi pubmed - Moustafa SR. The immune-opioid axis in prediabetes: predicting prediabetes with insulin resistance by plasma interleukin-10 and endomorphin-2 to kappa-opioid receptors ratio. Diabetol Metab Syndr. 2021;13(1):61.
doi pubmed - Shen J, Fang Y, Zhu H, Ge W. Plasma interleukin-22 levels are associated with prediabetes and type 2 diabetes in the Han Chinese population. J Diabetes Investig. 2018;9(1):33-38.
doi pubmed - Wang X, Ota N, Manzanillo P, Kates L, Zavala-Solorio J, Eidenschenk C, Zhang J, et al. Interleukin-22 alleviates metabolic disorders and restores mucosal immunity in diabetes. Nature. 2014;514(7521):237-241.
doi pubmed - Bashir H, Majid S, Khan MS, Bhat MH, Hamid R, Ashraf R, Faiz S. Inter-relationship of Pro- and Anti- inflammatory Biomarkers with the development of Type 2 Diabetes Mellitus. Heliyon. 2022;8(11):e11329.
doi pubmed - King GL. The role of inflammatory cytokines in diabetes and its complications. J Periodontol. 2008;79(8 Suppl):1527-1534.
doi pubmed - Ha T, Binh G, Nguyen T, Huong T. The role of serum interleukine-6 level in type 2 diabetic nephropathy. Res J Biotechnol. 2021;16:2-5.
- Dalla Vestra M, Mussap M, Gallina P, Bruseghin M, Cernigoi AM, Saller A, Plebani M, et al. Acute-phase markers of inflammation and glomerular structure in patients with type 2 diabetes. J Am Soc Nephrol. 2005;16(Suppl 1):S78-82.
doi pubmed - Izuora KE, Chase HP, Jackson WE, Coll JR, Osberg IM, Gottlieb PA, Rewers MJ, et al. Inflammatory markers and diabetic retinopathy in type 1 diabetes. Diabetes Care. 2005;28(3):714-715.
doi pubmed - Kristiansen OP, Mandrup-Poulsen T. Interleukin-6 and diabetes: the good, the bad, or the indifferent? Diabetes. 2005;54(Suppl 2):S114-124.
doi pubmed - Ayelign B, Negash M, Andualem H, Wondemagegn T, Kassa E, Shibabaw T, Akalu Y, et al. Association of IL-10 (- 1082 A/G) and IL-6 (- 174 G/C) gene polymorphism with type 2 diabetes mellitus in Ethiopia population. BMC Endocr Disord. 2021;21(1):70.
doi pubmed - Dalmas E, Venteclef N, Caer C, Poitou C, Cremer I, Aron-Wisnewsky J, Lacroix-Desmazes S, et al. T cell-derived IL-22 amplifies IL-1beta-driven inflammation in human adipose tissue: relevance to obesity and type 2 diabetes. Diabetes. 2014;63(6):1966-1977.
doi pubmed - Jia J, Tao X, Tian Z, Liu J, Ye X, Zhan Y. Vitamin D receptor deficiency increases systolic blood pressure by upregulating the renin-angiotensin system and autophagy. Exp Ther Med. 2022;23(4):314.
doi pubmed - Manchanda PK, Bid HK. Vitamin D receptor and type 2 diabetes mellitus: growing therapeutic opportunities. Indian J Hum Genet. 2012;18(3):274-275.
doi pubmed - Palomer X, Gonzalez-Clemente JM, Blanco-Vaca F, Mauricio D. Role of vitamin D in the pathogenesis of type 2 diabetes mellitus. Diabetes Obes Metab. 2008;10(3):185-197.
doi pubmed - Bid HK, Konwar R, Aggarwal CG, Gautam S, Saxena M, Nayak VL, Banerjee M. Vitamin D receptor (FokI, BsmI and TaqI) gene polymorphisms and type 2 diabetes mellitus: a North Indian study. Indian J Med Sci. 2009;63(5):187-194.
pubmed - Yarahmadi A, Shahrokhi SZ, Azarpira N, Mostafavi-Pour Z. Vitamin D, renin-angiotensin system, and COVID-19 - their importance in diabetes. Clin Diabetol. 2022;11:45-51.
doi - Kharb S, Goel K, Rajput R. Role of Vitamin D receptor in prediabetes. BMJ Open Diabetes Res Care. 2020;10.
doi - Pittas AG, Kawahara T, Jorde R, Dawson-Hughes B, Vickery EM, Angellotti E, Nelson J, et al. Vitamin D and risk for type 2 diabetes in people with prediabetes: a systematic review and meta-analysis of individual participant data from 3 randomized clinical trials. Ann Intern Med. 2023;176(3):355-363.
doi pubmed - Yu F, Cui LL, Li X, Wang CJ, Ba Y, Wang L, Li J, et al. The genetic polymorphisms in vitamin D receptor and the risk of type 2 diabetes mellitus: an updated meta-analysis. Asia Pac J Clin Nutr. 2016;25(3):614-624.
doi pubmed - Galuska D, Pacal L, Kankova K. Pathophysiological Implication of Vitamin D in Diabetic Kidney Disease. Kidney Blood Press Res. 2021;46(2):152-161.
doi pubmed - Tecilazich F, Formenti AM, Giustina A. Role of vitamin D in diabetic retinopathy: Pathophysiological and clinical aspects. Rev Endocr Metab Disord. 2021;22(4):715-727.
doi pubmed - Gnanaprakash V, Bodhini D, Kanthimathi S, Ginivenisha K, Shanthirani CS, Anjana RM, Mohan V, et al. Association of vitamin D receptor (TaqI, BsmI, and FokI) polymorphisms with prediabetes and Type 2 diabetes in Asian Indians. J Diabetol. 2019;10:29-36.
doi - Zakaria WNA, Mohd Yunus N, Yaacob NM, Omar J, Wan Mohamed WMI, Sirajudeen KNS, Tuan Ismail TS. Association between vitamin D receptor polymorphisms (BsmI and FokI) and glycemic control among patients with type 2 diabetes. Int J Environ Res Public Health. 2021;18(4):1595.
doi pubmed - Satou R, Penrose H, Navar LG. Inflammation as a regulator of the renin-angiotensin system and blood pressure. Curr Hypertens Rep. 2018;20(12):100.
doi pubmed - Titchenell PM, Lazar MA, Birnbaum MJ. Unraveling the regulation of hepatic metabolism by insulin. Trends Endocrinol Metab. 2017;28(7):497-505.
doi pubmed - Andrade LJ, Oliveira GC, Bittencourt AM, Xavier IP, Oliveira LM. Hepatic insulin resistance: a liver-specific type 2 diabetes. SciELO Preprints;2024.
doi - Wei Y, Clark SE, Morris EM, Thyfault JP, Uptergrove GM, Whaley-Connell AT, Ferrario CM, et al. Angiotensin II-induced non-alcoholic fatty liver disease is mediated by oxidative stress in transgenic TG(mRen2)27(Ren2) rats. J Hepatol. 2008;49(3):417-428.
doi pubmed - Rehman K, Akash MSH, Liaqat A, Kamal S, Qadir MI, Rasul A. Role of interleukin-6 in development of insulin resistance and type 2 diabetes mellitus. Crit Rev Eukaryot Gene Expr. 2017;27(3):229-236.
doi pubmed - Hassnine AA, Elsayed AM, Abdelaziez SK, Higazi MM, Okaily NI, Soliman W. Relationship between level of serum VIT D and insulin resistance in patients with MAFLD. BMC Endocr Disord. 2025;25(1):176.
doi pubmed - Hahn AW, Jonas U, Buhler FR, Resink TJ. Activation of human peripheral monocytes by angiotensin II. FEBS Lett. 1994;347(2-3):178-180.
doi pubmed - Ma LJ, Corsa BA, Zhou J, Yang H, Li H, Tang YW, Babaev VR, et al. Angiotensin type 1 receptor modulates macrophage polarization and renal injury in obesity. Am J Physiol Renal Physiol. 2011;300(5):F1203-1213.
doi pubmed - Saravi B, Li Z, Lang CN, Schmid B, Lang FK, Grad S, Alini M, et al. The tissue renin-angiotensin system and its role in the pathogenesis of major human diseases: Quo Vadis? Cells. 2021;10(3):650.
doi pubmed - Unger T, Steckelings UM, dos Santos RA. The protective arm of the renin angiotensin system (RAS): functional aspects and therapeutic implications. Academic Press; 2015.
- Mehta MM, Weinberg SE, Chandel NS. Mitochondrial control of immunity: beyond ATP. Nat Rev Immunol. 2017;17(10):608-620.
doi pubmed - Chelombitko MA. Role of reactive oxygen species in inflammation: a minireview. Mosc Univ Biol Sci Bull. 2018;73:199-202.
doi - Warnatsch A, Tsourouktsoglou TD, Branzk N, Wang Q, Reincke S, Herbst S, Gutierrez M, et al. Reactive oxygen species localization programs inflammation to clear microbes of different size. Immunity. 2017;46(3):421-432.
doi pubmed - Husain K, Hernandez W, Ansari RA, Ferder L. Inflammation, oxidative stress and renin angiotensin system in atherosclerosis. World J Biol Chem. 2015;6(3):209-217.
doi pubmed - Zakhary CM, Rushdi H, Hamdan JA, Youssef KN, Khan A, Abdalla MA, Khan S. Protective role of vitamin D therapy in diabetes mellitus type II. Cureus. 2021;13(8):e17317.
doi pubmed - Chen X, Wan Z, Geng T, Zhu K, Li R, Lu Q, Lin X, et al. Vitamin D status, vitamin D receptor polymorphisms, and risk of microvascular complications among individuals with type 2 diabetes: a prospective study. Diabetes Care. 2023;46(2):270-277.
doi pubmed - Zhao WJ, Xia XY, Yin J. Relationship of serum vitamin D levels with diabetic microvascular complications in patients with type 2 diabetes mellitus. Chin Med J (Engl). 2021;134(7):814-820.
doi pubmed - Ardizzone S, Cassinotti A, Trabattoni D, Manzionna G, Rainone V, Bevilacqua M, Massari A, et al. Immunomodulatory effects of 1,25-dihydroxyvitamin D3 on TH1/TH2 cytokines in inflammatory bowel disease: an in vitro study. Int J Immunopathol Pharmacol. 2009;22(1):63-71.
doi pubmed - Mokhtari Z, Hekmatdoost A, Nourian M. Antioxidant efficacy of vitamin D. J Parathyr Dis. 2017;5:11-16.
- Sistanizad M, Kouchek M, Miri M, Salarian S, Shojaei S, Vasegh FM, Kafshgari HS, et al. High dose vitamin D improves total serum antioxidant capacity and ICU outcome in critically ill patients-A randomized, double-blind clinical trial. Eur J Integr Med. 2021;42:101271.
doi - Vaidya A, Williams JS. The relationship between vitamin D and the renin-angiotensin system in the pathophysiology of hypertension, kidney disease, and diabetes. Metabolism. 2012;61(4):450-458.
doi pubmed - Gu JC, Wu YG, Huang WG, Fan XJ, Chen XH, Zhou B, Lin ZJ, et al. Effect of vitamin D on oxidative stress and serum inflammatory factors in the patients with type 2 diabetes. J Clin Lab Anal. 2022;36(5):e24430.
doi pubmed - Maranduca MA, Tanase DM, Branisteanu DC, Serban DN, Branisteanu DE, Serban IL. Involvement of proinflammatory cytokines in angiotensin II-induced hypertension in rat. Exp Ther Med. 2020;20(4):3541-3545.
doi pubmed - Feng Q, Liu D, Lu Y, Liu Z. The interplay of renin-angiotensin system and toll-like receptor 4 in the inflammation of diabetic nephropathy. J Immunol Res. 2020;2020:6193407.
doi pubmed - Andrade M, Knight J, Bayram-Weston Z. Vitamin D: the ‘sunshine vitamin’, its role and the effects of deficiency. Nurs Times. 2024;120:18-23.
- Aghaaliakbari F, Abbasi MA, Ranjbar M, Jamshidi Makiani M, Farrokhpour M, Safarnezhad Tameshkel F, Karbalaie Niya MH, et al. Angiotensin converting enzyme inhibitors, a risk factor of poor outcome in diabetic patients with COVID-19 infection. Iran J Kidney Dis. 2020;14(6):482-487.
pubmed - Chappell MC. Renin-angiotensin system and sex differences in COVID-19: a critical assessment. Circ Res. 2023;132(10):1320-1337.
doi pubmed - An J, Niu F, Sim JJ. Cardiovascular and kidney outcomes of spironolactone or eplerenone in combination with ACEI/ARBs in patients with diabetic kidney disease. Pharmacotherapy. 2021;41(12):998-1008.
doi pubmed - Fried LF, Petruski-Ivleva N, Folkerts K, Schmedt N, Velentgas P, Kovesdy CP. ACE inhibitor or ARB treatment among patients with diabetes and chronic kidney disease. Am J Manag Care. 2021;27(20 Suppl):S360-S368.
doi pubmed - Choi SW, Ho CK. Antioxidant properties of drugs used in Type 2 diabetes management: could they contribute to, confound or conceal effects of antioxidant therapy? Redox Rep. 2018;23(1):1-24.
doi pubmed - Tarry-Adkins JL, Grant ID, Ozanne SE, Reynolds RM, Aiken CE. Efficacy and side effect profile of different formulations of metformin: a systematic review and meta-analysis. Diabetes Ther. 2021;12(7):1901-1914.
doi pubmed - Cangemi R, Loffredo L, Carnevale R, Perri L, Patrizi MP, Sanguigni V, Pignatelli P, et al. Early decrease of oxidative stress by atorvastatin in hypercholesterolaemic patients: effect on circulating vitamin E. Eur Heart J. 2008;29(1):54-62.
doi pubmed - Farkouh A, Riedl T, Gottardi R, Czejka M, Kautzky-Willer A. Sex-related differences in pharmacokinetics and pharmacodynamics of frequently prescribed drugs: a review of the literature. Adv Ther. 2020;37(2):644-655.
doi pubmed - Akpoveso OP, Ubah EE, Obasanmi G. Antioxidant phytochemicals as potential therapy for diabetic complications. Antioxidants (Basel). 2023;12(1):123.
doi pubmed - Kristofi R, Eriksson JW. Metformin as an anti-inflammatory agent: a short review. J Endocrinol. 2021;251(2):R11-R22.
doi pubmed - Sun Y, Li J, Xiao N, Wang M, Kou J, Qi L, Huang F, et al. Pharmacological activation of AMPK ameliorates perivascular adipose/endothelial dysfunction in a manner interdependent on AMPK and SIRT1. Pharmacol Res. 2014;89:19-28.
doi pubmed - Farkouh A, Baumgartel C, Gottardi R, Hemetsberger M, Czejka M, Kautzky-Willer A. Sex-related differences in drugs with anti-inflammatory properties. J Clin Med. 2021;10(7):1441.
doi pubmed - Ren Y, Chen Y, Zheng W, Kong W, Liao Y, Zhang J, Wang M, et al. The effect of GLP-1 receptor agonists on circulating inflammatory markers in type 2 diabetes patients: a systematic review and meta-analysis. Diabetes Obes Metab. 2025;27(7):3607-3626.
doi pubmed - Wang D, Liu J, Zhong L, Li S, Zhou L, Zhang Q, Li M, et al. The effect of sodium-glucose cotransporter 2 inhibitors on biomarkers of inflammation: a systematic review and meta-analysis of randomized controlled trials. Front Pharmacol. 2022;13:1045235.
doi pubmed - Shiber S, Sharabi A, Ayalon I, Naamany E, Grossman A, Molad Y. Anti-inflammatory and survival benefits of dipeptidyl peptidase 4 inhibitors among patients with gout, T2DM patients and chronic kidney disease. Exp Clin Endocrinol Diabetes. 2025;133(5):253-258.
doi pubmed - Xie D, Wang Q, Huang W, Zhao L. Dipeptidyl-peptidase-4 inhibitors have anti-inflammatory effects in patients with type 2 diabetes. Eur J Clin Pharmacol. 2023;79(10):1291-1301.
doi pubmed - Neuen BL, Fletcher RA, Heath L, Perkovic A, Vaduganathan M, Badve SV, Tuttle KR, et al. Cardiovascular, kidney, and safety outcomes with GLP-1 receptor agonists alone and in combination with SGLT2 inhibitors in type 2 diabetes: a systematic review and meta-analysis. Circulation. 2024;150(22):1781-1790.
doi pubmed - Xie X, Chang X, Chen L, Huang K, Huang J, Wang S, Shen X, et al. Berberine ameliorates experimental diabetes-induced renal inflammation and fibronectin by inhibiting the activation of RhoA/ROCK signaling. Mol Cell Endocrinol. 2013;381(1-2):56-65.
doi pubmed - Weisberg SP, Leibel R, Tortoriello DV. Dietary curcumin significantly improves obesity-associated inflammation and diabetes in mouse models of diabesity. Endocrinology. 2008;149(7):3549-3558.
doi pubmed - Holt A, Strange JE, Nouhravesh N, Nielsen SK, Malik ME, Schjerning AM, Kober L, et al. Heart failure following anti-inflammatory medications in patients with type 2 diabetes mellitus. J Am Coll Cardiol. 2023;81(15):1459-1470.
doi pubmed - Vishwakarma RK, Negi DS. The development of COX-1 and COX-2 inhibitors: a review. Int J Pharm Sci Res. 2020;11:3544.
doi - Abdel-Rehim WM, El-Tahan RA, El-Tarawy MA, Shehata RR, Kamel MA. The possible antidiabetic effects of vitamin D receptors agonist in rat model of type 2 diabetes. Mol Cell Biochem. 2019;450(1-2):105-112.
doi pubmed - Barbarawi M, Zayed Y, Barbarawi O, Bala A, Alabdouh A, Gakhal I, Rizk F, et al. Effect of vitamin D supplementation on the incidence of diabetes mellitus. J Clin Endocrinol Metab. 2020;105(8):2857-2868.
doi pubmed - Wu C, Qiu S, Zhu X, Li L. Vitamin D supplementation and glycemic control in type 2 diabetes patients: A systematic review and meta-analysis. Metabolism. 2017;73:67-76.
doi pubmed - Dikalov SI, Nazarewicz RR. Angiotensin II-induced production of mitochondrial reactive oxygen species: potential mechanisms and relevance for cardiovascular disease. Antioxid Redox Signal. 2013;19(10):1085-1094.
doi pubmed - Ding J, Yu M, Jiang J, Luo Y, Zhang Q, Wang S, Yang F, et al. Angiotensin II decreases endothelial nitric oxide synthase phosphorylation via AT(1)R Nox/ROS/PP2A pathway. Front Physiol. 2020;11:566410.
doi pubmed - Werner C, Baumhakel M, Teo KK, Schmieder R, Mann J, Unger T, Yusuf S, et al. RAS blockade with ARB and ACE inhibitors: current perspective on rationale and patient selection. Clin Res Cardiol. 2008;97(7):418-431.
doi pubmed - Dehkordi AH, Abbaszadeh A, Mir S, Hasanvand A. Metformin and its anti-inflammatory and anti-oxidative effects; new concepts. J Ren Inj Prev. 2018;8:54-61.
doi
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.