| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 16, Number 2, April 2026, pages 109-117
Impact of L-Carnitine on Reproductive Outcomes in Women With Polycystic Ovarian Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Eman Alshehria, Saeed Baradwanb, Majed Saeed Alshahranic, Hussein Sabbanb, Shimaa Mohamed Abdel-Kareemd, Marwa Fekry Mohamed El Sayedd, Ahmed Mohamed Abdelhakime, f
aDepartment of Obstetrics and Gynecology, College of Medicine, King Saud University, Riyadh, Saudi Arabia
bDepartment of Obstetrics and Gynecology, King Faisal Specialist Hospital and Research Center, Jeddah, Saudi Arabia
cDepartment of Obstetrics and Gynecology, Faculty of Medicine, Najran University, Najran, Saudi Arabia
dDepartment of Endocrinology and Metabolism, Faculty of Medicine for Girls, Al-Azhar University, Cairo, Egypt
eKasr Al-Ainy Faculty of Medicine, Cairo University, Cairo, Egypt
fCorresponding Author: Ahmed Mohamed Abdelhakim, Kasr Al-Ainy Faculty of Medicine, Cairo University, Cairo, Egypt
Manuscript submitted December 21, 2025, accepted February 25, 2026, published online April 8, 2026
Short title: L-Carnitine on Reproductive Outcomes in PCOS
doi: https://doi.org/10.14740/jem1624
| Abstract | ▴Top |
Background: Polycystic ovarian syndrome (PCOS) is one of the most common endocrine disorders affecting women of reproductive age and a major cause of anovulatory infertility. L-carnitine, a naturally occurring compound involved in energy metabolism and mitochondrial function, has gained attention for its potential role in enhancing ovarian function and improving reproductive outcomes. However, evidence regarding its efficacy during controlled ovarian stimulation remains limited and inconsistent.
Methods: A comprehensive literature search was conducted across multiple electronic databases from inception to May 2025 to identify eligible randomized controlled trials (RCTs) evaluating L-carnitine supplementation compared with placebo in women with PCOS undergoing controlled ovarian stimulation. Data were extracted and analyzed using RevMan software. Primary outcomes included ovulation rate and clinical pregnancy rate; secondary outcomes were endometrial thickness and number of mature follicles.
Results: Six RCTs comprising 485 participants met the inclusion criteria. Pooled analysis demonstrated that L-carnitine supplementation significantly improved ovulation and clinical pregnancy rates compared with placebo. Additionally, women receiving L-carnitine exhibited greater endometrial thickness and a higher number of mature follicles than those in the control group.
Conclusions: L-carnitine supplementation during controlled ovarian stimulation appears to enhance reproductive outcomes in women with PCOS by improving ovulation, endometrial receptivity, and follicular development. These findings suggest that L-carnitine may serve as a valuable adjunct therapy in fertility treatment protocols for women with PCOS.
Keywords: L-carnitine; Polycystic ovarian syndrome; Pregnancy rate
| Introduction | ▴Top |
Polycystic ovary syndrome (PCOS) is one of the most common endocrine disorders affecting women of reproductive age, with an estimated global prevalence of 5–15% [1]. The etiology of PCOS is multifactorial, involving a complex interplay of genetic, environmental, and lifestyle factors, though its precise pathogenesis remains unclear [2]. Clinically, PCOS is characterized by chronic anovulation, hyperandrogenism, insulin resistance, and menstrual irregularities [3, 4].
Women with PCOS frequently experience ovulatory dysfunction driven by excess body weight, elevated androgen concentrations, and increased luteinizing hormone (LH) secretion, contributing to subfertility and difficulty achieving pregnancy [5, 6]. Management typically focuses on lifestyle modification and pharmacological interventions such as ovulation-inducing agents, insulin sensitizers, and gonadotropins [7]. Emerging evidence further highlights the role of nutrition-related metabolic pathways in regulating ovarian physiology, raising interest in dietary supplements as adjunctive strategies to improve reproductive outcomes [8].
L-carnitine, the biologically active form of carnitine synthesized from lysine and methionine, plays a key role in mitochondrial fatty acid transport, cellular energy production, and metabolic regulation, with reported benefits on insulin sensitivity, oxidative stress, and glucose metabolism [9–11]. Notably, women with PCOS have been shown to have lower levels of total and free L-carnitine, a deficiency associated with elevated androgen and insulin levels that may contribute to the metabolic and reproductive disturbances characteristic of this syndrome [12].
Studies examining the effects of L-carnitine supplementation on reproductive outcomes in women with PCOS undergoing controlled ovarian stimulation (COS) have yielded inconsistent results. One study demonstrated that combining L-carnitine with letrozole for ovulation induction improves ovulation rates, clinical pregnancy rates, and endometrial thickness in PCOS patients [13]. In contrast, another study reported no significant benefits on pregnancy outcomes or endometrial development when L-carnitine was added to antagonist ovarian stimulation protocol [14]. Given the limited and conflicting evidence, a rigorous pooled analysis is needed to provide more reliable estimates of the effect of L-carnitine supplementation on fertility outcomes in this population. Consequently, we conducted a systematic review and meta-analysis to evaluate the impact of L-carnitine supplementation during COS on fertility outcomes in women with PCOS.
| Materials and Methods | ▴Top |
This systematic review and meta-analysis were conducted following the Cochrane Handbook for Systematic Reviews of Interventions. We adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement guidelines during the preparation of our review. All the data used in this review were extracted from published papers, making ethical approval unnecessary.
Search strategy
A comprehensive literature search was conducted across four electronic databases (PubMed, Scopus, Cochrane Library, and Web of Science) from inception through May 2025, with no restrictions on language or publication year. The search combined terms related to the intervention (Carnitine OR Levocarnitine OR L-carnitine OR L carnitine) and the condition (Polycystic ovary syndrome OR Polycystic ovarian syndrome OR PCOS OR Stein-Leventhal syndrome OR Stein Leventhal syndrome) using Boolean operators. Two reviewers (SB and MSA) independently conducted the search, with disagreements resolved through discussion and consensus.
Study selection
Studies were eligible for inclusion if they met the following criteria: 1) women diagnosed with PCOS according to the Rotterdam criteria; 2) intervention consisting of L-carnitine supplementation in combination with COS protocols; 3) comparator consisting of placebo alongside the same COS protocols; 4) reporting of ovulation rate and/or clinical pregnancy rate as outcomes; and 5) randomized controlled trial (RCT) study design.
Studies were excluded based on the following criteria: 1) non-randomized study designs including observational studies, case series, and case reports; 2) crossover studies without adequate washout periods; 3) review articles, systematic reviews, meta-analyses, editorials, and commentaries; 4) studies without extractable numerical data; 5) studies lacking a control or comparison group; and 6) insufficient reporting of selected outcomes (ovulation rate or clinical pregnancy rate).
All retrieved records were imported into EndNote X9 reference manager for duplicate removal and organization. Two reviewers (SB and MSA) independently screened titles and abstracts of all identified records against the eligibility criteria. Articles deemed potentially relevant were retrieved in full text and independently assessed for eligibility by the same two reviewers. Disagreements at both screening stages were resolved through discussion and consensus.
Data extraction
Data extraction was performed independently by two reviewers (SB and AMA) using a standardized extraction form developed in Microsoft Excel. Extracted information included study characteristics (first author, publication year, country), participant demographics (sample size, mean maternal age, body mass index), infertility duration, and intervention details (L-carnitine dosage, duration, COS protocol). Discrepancies between reviewers were resolved through discussion until consensus was achieved.
Primary outcomes were ovulation rate and clinical pregnancy rate. Ovulation rate was defined as the proportion of women achieving ovulation, confirmed by ultrasonographic evidence of follicular rupture and/or serum progesterone ≥ 3 ng/mL in the mid-luteal phase. Clinical pregnancy rate was defined as the proportion of women with ultrasonographic visualization of one or more gestational sacs with detectable fetal cardiac activity at 6–8 weeks’ gestation. Secondary outcomes included endometrial thickness (maximum anterior-posterior diameter in mm measured by transvaginal ultrasound on day of human chorionic gonadotropin (hCG) trigger) and number of mature follicles (follicles ≥ 17 mm diameter at final follicular assessment). All included studies used consistent definitions for these outcomes, enabling direct comparison and meta-analysis.
Risk of bias assessment
The risk of bias of the included trials was assessed using the Cochrane Risk of Bias tool [15]. Two reviewers (SB and AMA) independently evaluated the following domains: random sequence generation, allocation concealment, blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other potential sources of bias. Any discrepancies were resolved through discussion until consensus was achieved. Each domain was judged as having a low, high, or unclear risk of bias.
Data analysis
The meta-analysis was conducted using Review Manager (RevMan) software, version 5.4.0. Two reviewers (MSA and HS) independently performed the statistical analyses, resolving any discrepancies through discussion until consensus was achieved. Dichotomous variables were pooled as odds ratios (ORs) with 95% confidence intervals (CIs), while continuous variables were presented as mean differences (MDs) with 95% CIs. Random-effects models were applied for all outcomes to account for both within-study and between-study variability, providing more conservative effect estimates. Heterogeneity among studies was assessed using the Chi-square test (P < 0.1) and the I2 statistic [16]. For outcomes with I2 above 50%, sensitivity analyses were performed by sequentially excluding one study at a time to assess the influence of each individual study on the overall pooled estimate and to identify potential sources of heterogeneity. Statistical significance was defined as P < 0.05 for all outcomes.
| Results | ▴Top |
Results of the literature search and characteristics of the included studies
The selection process for this study is illustrated in the PRISMA flow diagram (Fig. 1). A total of 222 studies were initially retrieved through database searches. After screening titles and abstracts, 22 articles were assessed for full-text eligibility. Following full-text review, 16 studies were excluded due to ineligible study design, population, or outcomes. Finally, six RCTs met the inclusion criteria and were incorporated into both qualitative synthesis and quantitative meta-analysis [13, 14, 17–20]. These trials investigated L-carnitine supplementation at doses ranging from 1,000 to 3,000 mg/day. COS protocols varied among the studies, including letrozole [13], clomiphene citrate (CC) [17–19], and gonadotropin-releasing hormone (GnRH)-antagonist protocol [14, 20]. Across all six studies, a total of 485 participants were enrolled, with three trials conducted in Egypt [13, 17, 18] and three in Iran [14, 19, 20]. The characteristics of the included studies are presented in Table 1.
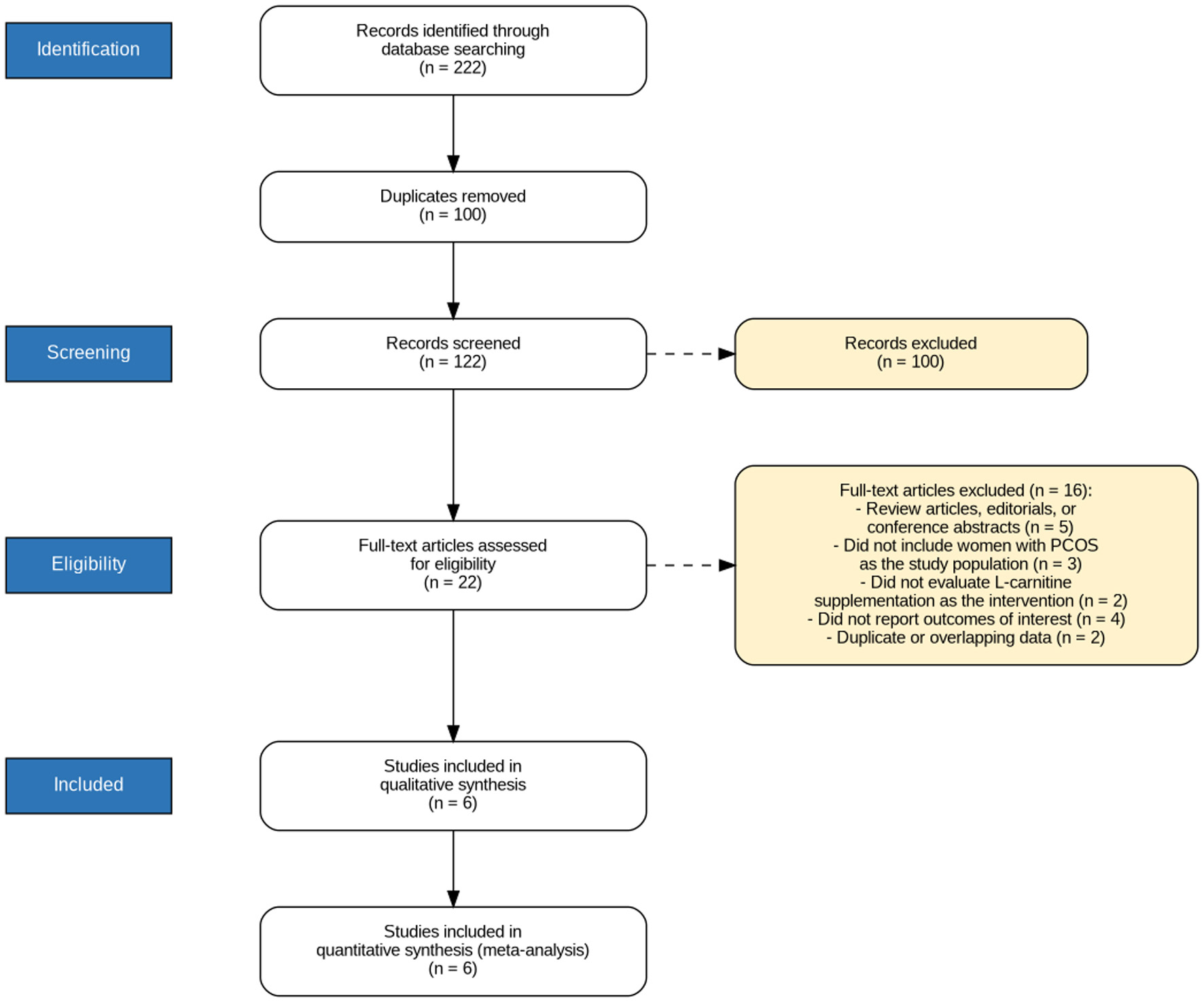 Click for large image | Figure 1. PRISMA flow diagram illustrating the systematic literature search and study selection process, including the number of records identified, screened, assessed for eligibility, and finally included in the meta-analysis. |
 Click to view | Table 1. Characteristics of the Included Studies |
Risk of bias of included studies
Risk of bias was assessed using the Cochrane Risk of Bias tool (RoB 1) for all six included studies (Fig. 2). All studies demonstrated low risk for random sequence generation, allocation concealment, and selective reporting. However, blinding was inadequate in several trials: three studies had high risk of performance bias [13, 17, 18] and four had high risk of detection bias [13, 17, 18, 20]. Additionally, one trial had high risk for incomplete outcome data [20], and one had unclear risk for other sources of bias [17].
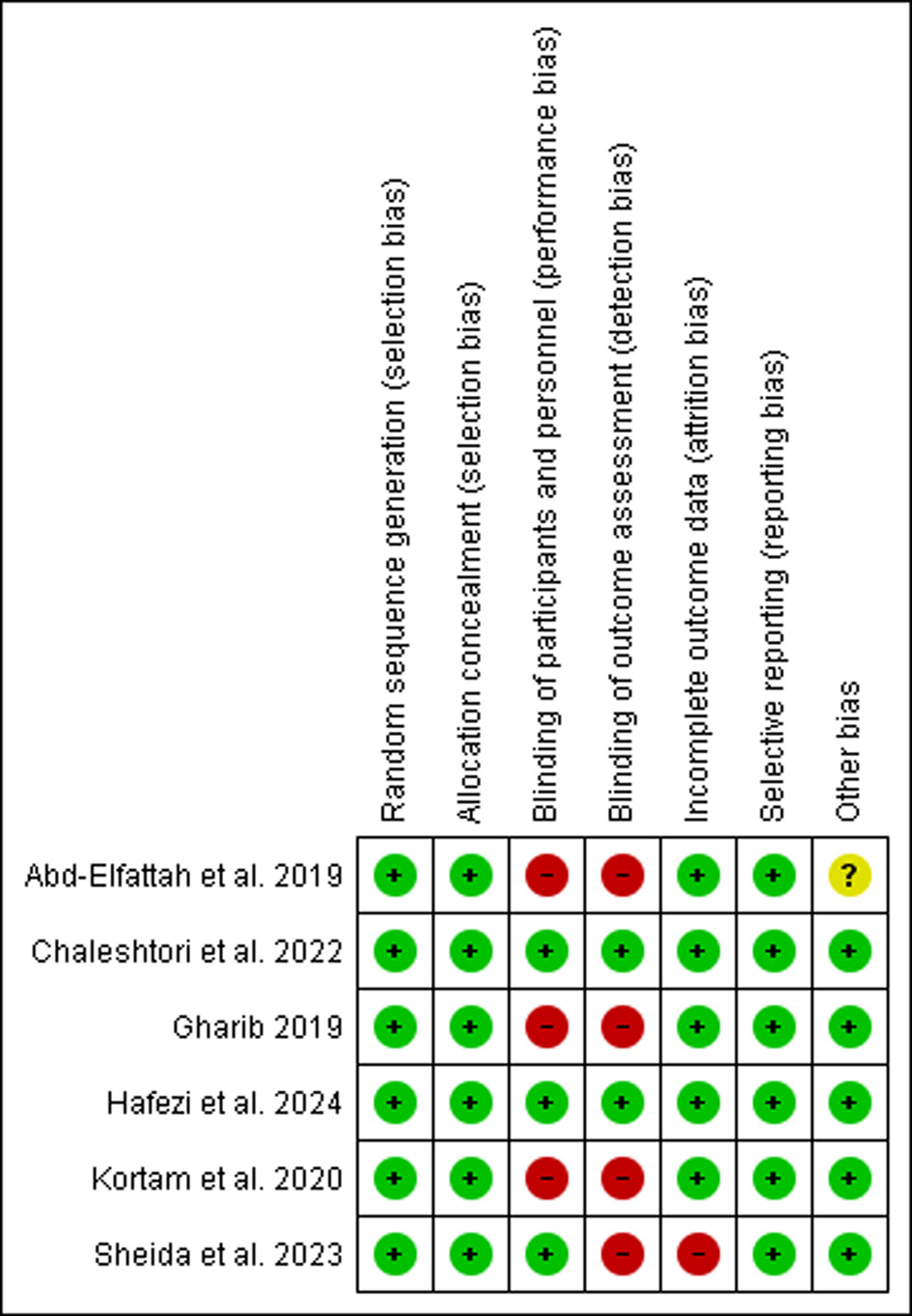 Click for large image | Figure 2. Risk of bias summary for the included randomized controlled trials assessed using the Cochrane Risk of Bias tool (RoB 1), displaying judgments across six domains for each included study. |
Outcomes
Ovulation rate
As shown in Figure 3, the ovulation rate was significantly higher in the L-carnitine group compared with the placebo group (OR = 4.78, 95% CI 2.95–7.75, P < 0.001). The included studies demonstrated low heterogeneity (P = 0.42, I2 = 0%).
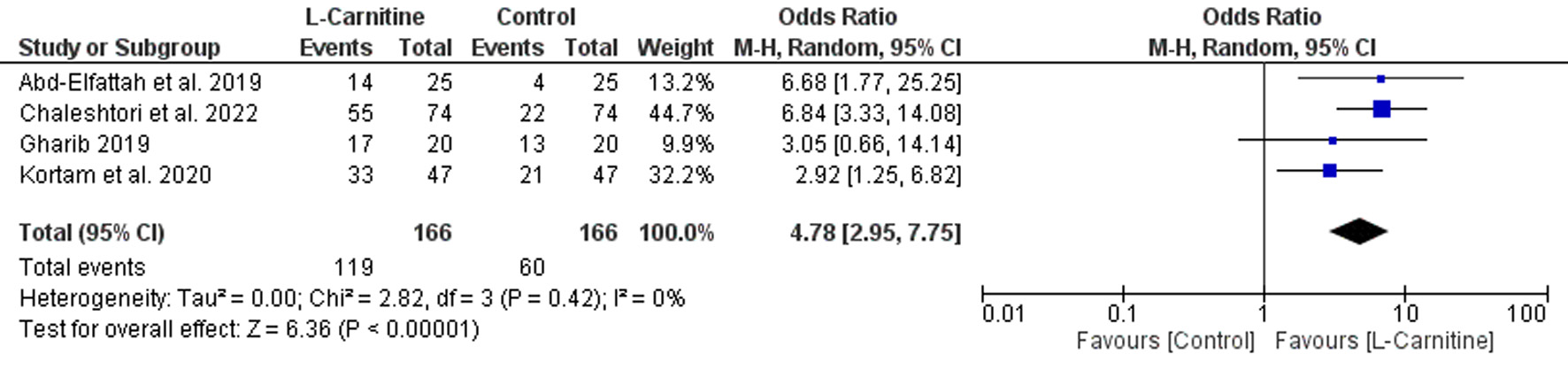 Click for large image | Figure 3. Forest plot of ovulation rate in women with PCOS receiving L-carnitine supplementation versus control during controlled ovarian stimulation. |
Clinical pregnancy rate
Figure 4 illustrates a significant increase in the clinical pregnancy rate among participants receiving L-carnitine compared with the control group (OR = 2.12, 95% CI 1.16–3.85, P = 0.01). The included studies demonstrated moderate heterogeneity (P = 0.15, I2 = 39%).
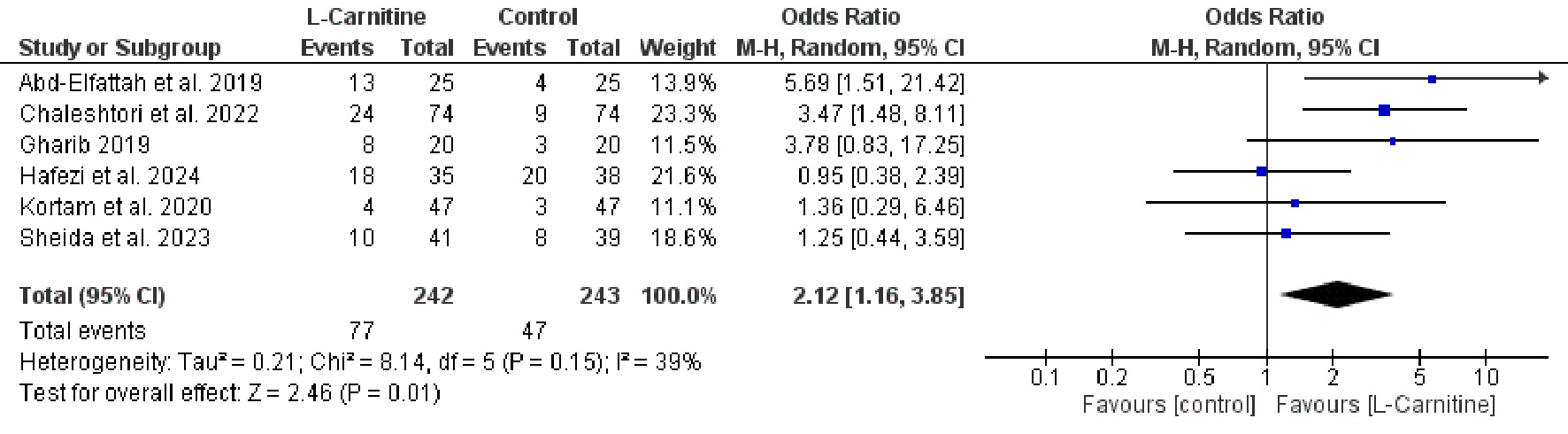 Click for large image | Figure 4. Forest plot of clinical pregnancy rate in women with PCOS receiving L-carnitine supplementation versus control during controlled ovarian stimulation. |
Endometrial thickness
The mean endometrial thickness was significantly greater in the L-carnitine group than in the control group (MD = 1.74, 95% CI 0.92–2.56, P < 0.001), as depicted in Figure 5. Substantial heterogeneity was observed among the studies (P = 0.002, I2 = 74%). Sensitivity analysis performed by excluding one trial [17] reduced heterogeneity to a moderate level (P = 0.14, I2 = 43%), while maintaining statistical significance and yielding a more conservative estimate of improvement in endometrial thickness (MD = 1.33, 95% CI 0.80–1.86, P < 0.001).
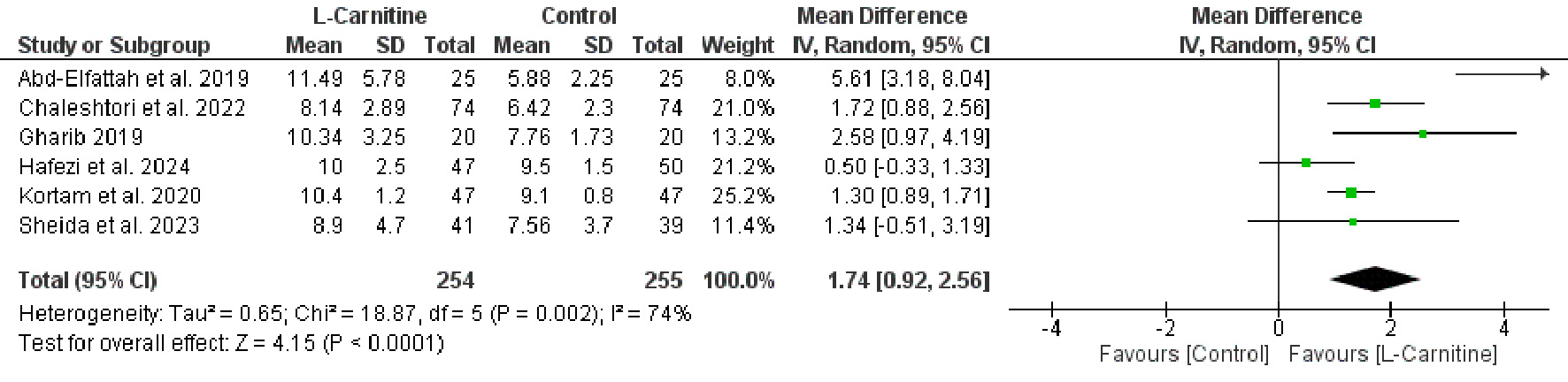 Click for large image | Figure 5. Forest plot of endometrial thickness in women with PCOS receiving L-carnitine supplementation versus control during controlled ovarian stimulation. |
Number of mature follicles
As shown in Figure 6, women receiving L-carnitine developed a significantly higher number of mature follicles compared with the control group (MD = 1.06, 95% CI 0.64–1.47, P < 0.001). The included studies demonstrated moderate heterogeneity (P = 0.12, I2 = 48%).
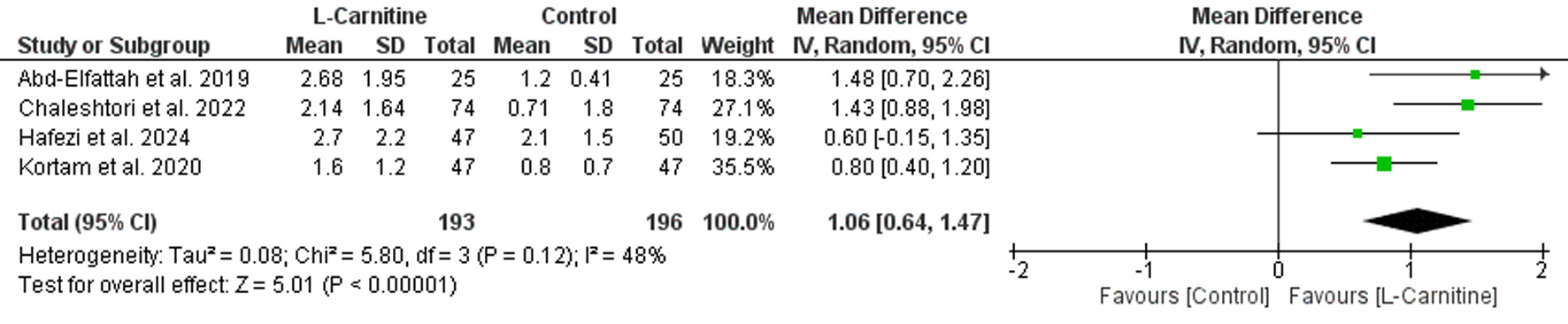 Click for large image | Figure 6. Forest plot of number of mature follicles in women with PCOS receiving L-carnitine supplementation versus control during controlled ovarian stimulation. |
| Discussion | ▴Top |
The results of this meta-analysis demonstrate that the addition of L-carnitine to COS protocols significantly enhances reproductive outcomes in women with PCOS. Participants receiving L-carnitine showed statistically significant improvements in both ovulation and clinical pregnancy rates compared with the control group. Moreover, L-carnitine supplementation was associated with increased endometrial thickness and a higher number of mature follicles, indicating improved ovarian responsiveness and endometrial receptivity.
Several studies have explored the benefits of L-carnitine supplementation on reproductive outcomes in women with PCOS. When combined with CC in clomiphene-resistant patients, L-carnitine significantly improved ovulation quality, clinical pregnancy rates, number of mature follicles, estradiol concentrations, and endometrial thickness compared with controls [17]. Another study confirmed improvements in ovulation rate, endometrial thickness, and pre-ovulatory follicle count, though the difference in pregnancy rates did not reach statistical significance [18]. In a clinical trial by Latifian et al [21], L-carnitine administration in women who previously failed CC and gonadotropin stimulation resulted in dominant follicle development in 64% of participants and a positive pregnancy test in 20%, alongside a significant increase in endometrial thickness.
A randomized trial found that adding L-carnitine to letrozole therapy significantly improved endometrial thickness, ovulation rate, and both chemical and clinical pregnancy rates in women with PCOS [13]. Similarly, a triple-blind randomized trial in clomiphene-resistant patients reported significant improvements in ovulation rate, endometrial thickness, serum progesterone, mature follicle count, and clinical pregnancy rate when L-carnitine was combined with CC [19]. In contrast, two studies evaluating L-carnitine as an adjunct to GnRH-antagonist protocols found no significant improvements in oocyte maturity, fertilization rate, endometrial thickness, or pregnancy outcomes [14, 20].
Repeated ovarian stimulation in women with PCOS reduces mitochondrial deoxyribonucleic acid (DNA) levels, increases oxidative stress markers such as 8-hydroxydeoxyguanosine, and disrupts mitochondrial distribution in oocytes, all of which may impair oocyte quality and assisted reproductive technology (ART) success rates [22–24]. L-carnitine supplementation may address these deficits by enhancing cellular energy production, reducing oxidative stress and lipotoxicity, and supporting oocyte maturation [25, 26]. Its mechanisms include facilitating palmitate transport into mitochondria, maintaining acetyl-coenzyme A (CoA)/CoA balance, scavenging reactive oxygen species, and reducing oocyte apoptosis [27, 28]. Additionally, L-carnitine downregulates pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), potentially creating a more favorable microenvironment for oocyte development and improving endometrial receptivity during the peri-implantation period [29].
L-carnitine also offers potential cost-effectiveness advantages in PCOS fertility management. As a widely available, low-cost supplement with a favorable safety profile, it requires no extensive monitoring or adverse event management. The improvements in ovulation and pregnancy rates demonstrated in this meta-analysis suggest a potential reduction in the number of stimulation cycles needed, which could decrease overall treatment costs and patient burden. These practical attributes make L-carnitine an easily integrated adjuvant option within existing fertility treatment protocols.
Our study had several notable strengths. We implemented a comprehensive search strategy across multiple databases and included only RCTs, which enhanced the reliability and validity of our findings. Adhering to PRISMA guidelines further underscored the methodological rigor of our research. Additionally, we carefully screened studies to minimize bias and ensure consistency in data extraction and analysis.
Several limitations warrant consideration. The small number of eligible trials and modest sample sizes may have reduced statistical power and limited the precision and generalizability of findings. Inadequate reporting of blinding procedures in some studies raises concerns about performance and detection bias. Additionally, inconsistent reporting of live birth and miscarriage outcomes, the most definitive fertility endpoints, precluded their adequate analysis, limiting firm conclusions about the ultimate clinical benefit of L-carnitine supplementation. Heterogeneity was observed across several outcomes, likely driven by differences in L-carnitine dosage, duration, and formulation, as well as variability in baseline patient characteristics such as body mass index (BMI), insulin resistance, metabolic profile, and PCOS severity. The included trials also employed heterogeneous ovarian stimulation protocols, representing distinct therapeutic contexts with differing pharmacologic mechanisms. The limited number of eligible studies precluded meaningful subgroup analyses by stimulation type or other clinical variables, restricting our ability to explore heterogeneity sources in depth. Pooled estimates should therefore be interpreted with caution. Finally, the exclusion of gray literature further raises concerns about publication bias and potential overestimation of treatment effects.
Future high-quality randomized trials with larger sample sizes are needed to assess varying L-carnitine doses and extended durations (e.g., 12 weeks) in women with PCOS, particularly those who are severely obese or have poor ovarian response. Emphasis should be placed on clinically meaningful outcomes such as ongoing pregnancy, miscarriage, and live birth rates, as these are the most relevant indicators of reproductive success. Beyond clinical efficacy, formal cost-effectiveness analyses are urgently needed, as economic evaluations comparing L-carnitine supplementation to standard protocols are currently lacking. Given the limited and inconsistent safety reporting across existing trials, which precluded a formal adverse event analysis, future investigations should systematically monitor and report adverse effects using standardized protocols to establish the safety profile of long-term L-carnitine supplementation.
Conclusions
Oral L-carnitine supplementation during COS significantly improves ovulation rate, clinical pregnancy rate, endometrial thickness, and the number of mature follicles in women with PCOS. These findings support its potential role as a safe, affordable, and effective adjuvant therapy in fertility treatment protocols. However, larger multicenter trials with standardized dosing protocols and longer follow-up periods are warranted to confirm and generalize these findings.
Acknowledgments
None to declare.
Financial Disclosure
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors have no conflict of interest.
Informed Consent
Not applicable.
Author Contributions
EA contributed to writing, review, and editing of the manuscript. SB designed the study, participated in data collection, and wrote the manuscript. MSA and HS participated in statistical analysis. SMAK and MFMES contributed to the critical revision and editing of the manuscript. AMA contributed to study design and data collection. All authors read and approved the final version of the manuscript for publication.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
ART: assisted reproductive technology; BMI: body mass index; CC: clomiphene citrate; CI: confidence interval; CoA: coenzyme A; COS: controlled ovarian stimulation; DNA: deoxyribonucleic acid; GnRH: gonadotropin-releasing hormone; hCG: human chorionic gonadotropin; IL-6: interleukin-6; LC: L-carnitine; LH: luteinizing hormone; MD: mean difference; NA: not available; OR: odds ratio; PCOS: polycystic ovarian syndrome; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analysis; RCT: randomized controlled trial; RoB: risk of bias; TNF-α: tumor necrosis factor-alpha
| References | ▴Top |
- Fu L, Xie N, Qu F, Zhou J, Wang F. The association between polycystic ovary syndrome and metabolic syndrome in adolescents: a systematic review and meta-analysis. Reprod Sci. 2023;30(1):28-40.
doi pubmed - Escobar-Morreale HF. Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat Rev Endocrinol. 2018;14(5):270-284.
doi pubmed - Sirmans SM, Pate KA. Epidemiology, diagnosis, and management of polycystic ovary syndrome. Clin Epidemiol. 2013;6:1-13.
doi pubmed - Chelikam N, Akella SA, Lakhanpal MR, et al. Role of ketogenic diets and intermittent fasting in neurologic diseases, cancers, and obesity: a systematic review of human studies. J Endocrinol Metab. 2024;14(3):103-127.
doi - Li M, Ruan X, Mueck AO. Management strategy of infertility in polycystic ovary syndrome. Glob Health J. 2022;6(2):70-74.
doi - Morshed MS, Banu H, Akhtar N, et al. Luteinizing hormone to follicle-stimulating hormone ratio significantly correlates with androgen level and manifestations are more frequent with hyperandrogenemia in women with polycystic ovary syndrome. J Endocrinol Metab. 2021;11(1):14-21.
doi - Singh S, Pal N, Shubham S, Sarma DK, Verma V, Marotta F, Kumar M. Polycystic ovary syndrome: etiology, current management, and future therapeutics. J Clin Med. 2023;12(4):1454.
doi pubmed - Agarwal A, Sengupta P, Durairajanayagam D. Role of L-carnitine in female infertility. Reprod Biol Endocrinol. 2018;16(1):5.
doi pubmed - Pillich RT, Scarsella G, Risuleo G. Reduction of apoptosis through the mitochondrial pathway by the administration of acetyl-L-carnitine to mouse fibroblasts in culture. Exp Cell Res. 2005;306(1):1-8.
doi pubmed - Talenezhad N, Mohammadi M, Ramezani-Jolfaie N, Mozaffari-Khosravi H, Salehi-Abargouei A. Effects of l-carnitine supplementation on weight loss and body composition: A systematic review and meta-analysis of 37 randomized controlled clinical trials with dose-response analysis. Clin Nutr ESPEN. 2020;37:9-23.
doi pubmed - Power RA, Hulver MW, Zhang JY, Dubois J, Marchand RM, Ilkayeva O, Muoio DM, et al. Carnitine revisited: potential use as adjunctive treatment in diabetes. Diabetologia. 2007;50(4):824-832.
doi pubmed - Fenkci SM, Fenkci V, Oztekin O, Rota S, Karagenc N. Serum total L-carnitine levels in non-obese women with polycystic ovary syndrome. Hum Reprod. 2008;23(7):1602-1606.
doi pubmed - Gharib WF. The effect of adding L-carnitine to induction of ovulation with letrozole among PCOS patients. Austin J Obstet Gynecol. 2019;6(3):1-4.
- Hafezi M, Arabipoor A, Ghaffari F, Vesali S, Zareei M, Hessari ZH. Adding L-carnitine to antagonist ovarian stimulation doesn't improve the outcomes of IVF/ ICSI cycle in patients with polycystic ovarian syndrome: a double-blind randomized clinical trial. J Ovarian Res. 2024;17(1):9.
doi pubmed - Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928.
doi pubmed - Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560.
doi pubmed - Abd-Elfattah AT, Hashish MAE, Elomda FAE aal, Megahed HI. Effect of adding L-carnitine to clomiphene resistant PCOs women on the ovulation and the pregnancy rate. Egypt J Hosp Med. 2019;76(5):4138-4143.
- Kortam M, Abdelrahman R, Fateen H. L-carnitine and clomiphene citrate for induction of ovulation in women with polycystic ovary syndrome: randomized controlled trial. Evid Based Womens Health J. 2020;10(1):1-7.
doi - Chaleshtori MH, Taheripanah R, Shakeri A. Clomiphene citrate (CC) plus L-Carnitine improves clinical pregnancy rate along with glycemic status and lipid profile in clomiphene-resistant polycystic ovary syndrome patients: A triple-blind randomized controlled clinical trial. Obes Med. 2022;34:100400.
doi - Sheida A, Davar R, Tabibnejad N, Eftekhar M. The effect of adding L-Carnitine to the GnRH-antagonist protocol on assisted reproductive technology outcome in women with polycystic ovarian syndrome: a randomized clinical trial. Gynecol Endocrinol. 2023;39(1):1878135.
doi pubmed - Latifian S, Hamdi K, Totakhneh R. Effect of addition of L-carnitine in polycystic ovary syndrome (PCOS) patients with clomiphene citrate and gonodotropin resistant. Int J Curr Res Acad Rev. 2015;3(8):469-476.
- Miyamoto K, Sato EF, Kasahara E, Jikumaru M, Hiramoto K, Tabata H, Katsuragi M, et al. Effect of oxidative stress during repeated ovulation on the structure and functions of the ovary, oocytes, and their mitochondria. Free Radic Biol Med. 2010;49(4):674-681.
doi pubmed - Finsterer J. Mitochondrial dysfunction in polycystic ovary syndrome. Reprod Sci. 2023;30(5):1435-1442.
doi pubmed - Ringseis R, Keller J, Eder K. Role of carnitine in the regulation of glucose homeostasis and insulin sensitivity: evidence from in vivo and in vitro studies with carnitine supplementation and carnitine deficiency. Eur J Nutr. 2012;51(1):1-18.
doi pubmed - Sangouni AA, Sasanfar B, Ghadiri-Anari A, Hosseinzadeh M. Effect of l-carnitine supplementation on liver fat content and cardiometabolic indices in overweight/obese women with polycystic ovary syndrome: A randomized controlled trial. Clin Nutr ESPEN. 2021;46:54-59.
doi pubmed - Samimi M, Jamilian M, Ebrahimi FA, Rahimi M, Tajbakhsh B, Asemi Z. Oral carnitine supplementation reduces body weight and insulin resistance in women with polycystic ovary syndrome: a randomized, double-blind, placebo-controlled trial. Clin Endocrinol (Oxf). 2016;84(6):851-857.
doi pubmed - Abdelrazik H, Sharma R, Mahfouz R, Agarwal A. L-carnitine decreases DNA damage and improves the in vitro blastocyst development rate in mouse embryos. Fertil Steril. 2009;91(2):589-596.
doi pubmed - Chang D, Kong F, Jiang W, Li F, Zhang C, Ding H, Kang Y, et al. Effects of L-carnitine administration on sperm and sex hormone levels in a male wistar rat reproductive system injury model in a high-altitude hypobaric hypoxic environment. Reprod Sci. 2023;30(7):2231-2247.
doi pubmed - Modanloo M, Shokrzadeh M. Analyzing mitochondrial dysfunction, oxidative stress, and apoptosis: potential role of L-carnitine. Iran J Kidney Dis. 2019;13(2):74-86.
pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.