| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 16, Number 2, April 2026, pages 70-81
From Inflammation to Endocrine Dysregulation: Thyroid Hormonal Alterations in COVID-19
Malvina Svilenova Todorovaa, b, e, Victoria Tsvetanova Tsvetkovaa, b, Milena Atanasova Atanasovac, Adelaida Lazarova Rusevad, Katya Nikolova Todorovaa, b
aClinic of Endocrinology, University Hospital “Georgi Stranski” - Pleven, Bulgaria
bDepartment of Cardiology, Pulmonology, Endocrinology and Rheumatology, Medical University - Pleven, Bulgaria
cDepartment of Anatomy, Histology, Cytology and Biology; Biology Division, Medical University - Pleven, Bulgaria
dDepartment of Clinical Immunology, Allergology and Clinical Laboratory, Clinical Laboratory Division, Medical University - Pleven, Bulgaria
eCorresponding Author: Malvina Svilenova Todorova, Department of Cardiology, Pulmonology, Endocrinology and Rheumatology, Medical University - Pleven, 5800 Pleven, Bulgaria
Manuscript submitted December 22, 2025, accepted March 27, 2026, published online April 8, 2026
Short title: Thyroid Alterations in COVID-19
doi: https://doi.org/10.14740/jem1625
| Abstract | ▴Top |
Background: Although thyroid dysfunction has been increasingly reported in coronavirus disease 2019 (COVID-19), the mechanisms linking severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection to thyroid hormonal disturbances remain incompletely understood. The study aimed to examine the relationship between inflammatory status and thyroid function in patients with active COVID-19 and in post-COVID individuals, compared with healthy individuals.
Methods: An observational study with a cross-sectional analytical approach was conducted in 101 participants allocated into three groups: patients with active COVID-19 (group 1, n = 33), individuals with thyroid dysfunction following recovery from COVID-19 (group 2, n = 35), and a non-infected reference group (group 3, n = 33). Thyroid parameters, including thyroid-stimulating hormone (TSH), free triiodothyronine (FT3), free thyroxine (FT4), thyroglobulin (Tg), and thyroid peroxidase (TPO); selected cytokines, including interleukin-17A (IL-17A), interleukin-10 (IL-10), interleukin-7 (IL-7), and interferon-gamma (IFN-γ); and markers related to hypoxia and antioxidant defense, including hypoxia-inducible factor 1-alpha (HIF-1α) and nuclear factor erythroid 2-related factor 2 (NFE2L2), were assessed.
Results: Significant between-group differences were observed for FT3 and TSH, with the lowest FT3 levels detected in the active COVID-19 group. Reduced FT3 was the most frequent abnormality in group 1, identified in 36.4% of patients, supporting a pattern consistent with euthyroid sick syndrome. TSH levels were significantly lower in group 1 than in group 3, while FT3 levels differed significantly between group 1 and both group 2 and group 3. HIF-1α and NFE2L2 levels were highest in active COVID-19, indicating marked intracellular hypoxia together with activation of the antioxidant defense response. Receiver operating characteristic analysis showed good discriminatory performance of HIF-1α for distinguishing active COVID-19 from the reference group (area under the curve = 0.771). Post-COVID patients showed the lowest median levels of IL-17A, IL-10, IL-7, and IFN-γ, together with a high frequency of autoimmune thyroid disorders, most commonly Hashimoto’s thyroiditis, present in 65.7% of patients.
Conclusion: SARS-CoV-2 infection appears to induce transient thyroid hormonal alterations primarily through inflammatory and hypoxia-related mechanisms. These findings support the concept of virus-induced euthyroid sick syndrome during acute infection and highlight the potential for post-infectious thyroid dysfunction.
Keywords: COVID-19; Thyroid function; FT3; TSH; HIF-1α; Inflammation; Post-COVID
| Introduction | ▴Top |
The coronavirus disease 2019 (COVID-19) pandemic, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has emerged as a multisystem disease affecting not only the respiratory tract but nearly all organs and physiological systems in the human body [1]. Increasing evidence indicates that, beyond its pulmonary manifestations and systemic inflammatory response, the viral infection exerts a substantial impact on the endocrine system. In particular, involvement and suppression of the hypothalamic–pituitary–thyroid (HPT) axis have been associated with worse clinical outcomes [2].
Although thyroid dysfunction in COVID-19 has been increasingly reported, the mechanisms linking SARS-CoV-2 infection to thyroid hormonal disturbances remain incompletely understood. In particular, the relative contribution of direct viral tropism, systemic inflammation, immune dysregulation, and hypoxia-related pathways across the acute and post-COVID phases has not been fully clarified. This unresolved interplay represents an important knowledge gap in understanding COVID-19–related endocrine dysfunction.
Direct viral invasion represents a key and early mechanism underlying endocrine dysfunction in COVID-19. This process is driven by the pronounced viral organotropism toward endocrine tissues, including the hypothalamus, pituitary gland, thyroid and adrenal glands, gonads, and pancreas, which express high levels of the key proteins mediating viral entry, namely angiotensin-converting enzyme 2 (ACE2) and transmembrane serine protease 2 (TMPRSS2) [3]. Importantly, the differential tissue distribution and expression levels of ACE2 across human organs have been associated with variability in viral tropism, disease severity, and clinical outcomes, reflecting a key determinant of organ-specific susceptibility to SARS-CoV-2 infection [4]. The interaction between the viral spike protein and ACE2, followed by TMPRSS2-mediated proteolytic priming, is essential for viral–host cell membrane fusion and subsequent cellular entry. Thus, TMPRSS2 expression represents a critical rate-limiting factor for viral entry and replication, as its proteolytic activity enables spike protein activation and efficient viral internalization. Importantly, emerging evidence suggests that TMPRSS2 expression is subject to post-transcriptional regulation by microRNAs, particularly miR-98-5p, which has been identified as a specific modulator of TMPRSS2 expression in human endothelial cells through direct interaction with TMPRSS2 mRNA [5]. These regulatory mechanisms suggest that both receptor availability (ACE2) and protease activity (TMPRSS2), together with their molecular regulation, may contribute to inter-individual variability in tissue susceptibility, viral load, and disease severity. Their presence in the hormone-producing tissues of the endocrine organs facilitates direct viral invasion, replication, and maintenance of local inflammation, with subsequent temporary or persistent disruption of hormonal homeostasis [6].
Beyond direct viral effects, immune-mediated mechanisms play a central role in the development of thyroid dysfunction in COVID-19. In particular, systemic inflammation and immune-mediated cytotoxicity may contribute substantially to thyroid injury. SARS-CoV-2–induced immune activation is characterized by increased activity of Th1 lymphocytes, natual killer (NK) cells, and CD8+ cytotoxic T cells, which release pro-inflammatory and cytotoxic mediators, including interferon-gamma (IFN-γ). This cytotoxic immune response promotes apoptotic and prothrombotic signaling within thyrocytes, contributing to ischemia, cellular injury, and tissue necrosis [7].
At the molecular level, the SARS-CoV-2–induced inflammatory response is driven by the activation of key transcription factors, particularly nuclear factor kappa-B (NF-κB), which orchestrates the expression of a broad range of pro-inflammatory cytokines, chemokines, and other inflammatory mediators [8]. Emerging evidence indicates that this inflammatory milieu may disrupt thyroid hormone homeostasis by interfering with HPT axis regulation at multiple levels, potentially contributing to primary, secondary, or tertiary thyroid dysfunction [9].
In moderate to severe COVID-19, an exaggerated systemic inflammatory response may contribute to organ-specific manifestations, including subacute thyroiditis (SAT) and painless thyroiditis [10, 11]. Persistent inflammatory activation may also promote apoptosis and tissue injury, potentially contributing to prolonged thyroid dysfunction in some patients [12].
Panesar et al [13] and Lee et al [14] provide evidence for the role of SARS-CoV-2–activated inflammatory mechanisms in disrupting HPT axis integrity, leading to functional disturbances reflected by alterations in thyroid-stimulating hormone (TSH) and thyroid hormone levels. Inflammatory activation has been associated with suppressed TSH and impaired thyroxine (T4) synthesis. Moreover, NF-κB–dependent signaling modulates deiodinase activity by upregulating type 2 deiodinase (DIO2) while suppressing type 1 deiodinase (DIO1) expression, thereby reducing peripheral triiodothyronine (T3) availability. This dysregulation contributes to the development of low T3 syndrome (euthyroid sick syndrome, ESS), characterized by decreased free triiodothyronine (FT3), elevated reverse triiodothyronine (rT3), and inappropriately normal or reduced TSH levels. Collectively, these findings indicate that inflammation-driven alterations in both central regulation and peripheral hormone metabolism disrupt thyroid hormone feedback homeostasis [13, 14].
A third potential mechanism involves virus-triggered autoimmune thyroid dysfunction, associated with increased production of antithyroid antibodies, leading to thyroid functional abnormalities ranging from hyperthyroidism (thyrotropin receptor antibodies, TRAb) to hypothyroidism (thyroglobulin antibodies, TgAb; thyroid peroxidase antibodies, TPOAb), with occasional transient thyrotoxic phases [7, 15]. Structural and antigenic homology between the SARS-CoV-2 spike (S) protein and thyroid peroxidase (TPO) may promote immune cross-reactivity, thereby providing a potential mechanistic link between viral infection and TPO-directed thyroid autoimmunity [16]. These observations are further supported by Fallahi et al [17], who suggest that SARS-CoV-2–related immune dysregulation may contribute to autoimmune thyroid disease, particularly in the post-COVID phase.
Further observations of Cuadrado et al [18] and Devaux et al [19] on immune dysregulation in COVID-19 indicate that virus-induced hypoxia and hypoxic stress activate signaling pathways involving transcription factors such as hypoxia-inducible factor-1α (HIF-1α) and nuclear factor erythroid 2-related factor 2 (NFE2L2). These pathways promote metabolic reprogramming of immune cells and oxidative stress responses, which may in turn disturb thyroid hormone metabolism. In this context, HIF-1α-related hypoxic signaling may contribute to suppression of deiodinase activity and reduced T3 formation, while NFE2L2 is more likely to reflect activation of antioxidant defense mechanisms in response to oxidative stress [14, 20]. The result is a decrease in TSH and FT3 during active infection, with potential development of low T3 syndrome (ESS) and transient thyroid hormone suppression [14, 21].
Aim of the study
To investigate the relationship between inflammatory status and thyroid hormone levels in patients with active and post-COVID-19 infection, and to determine whether SARS-CoV-2 induces disturbances in thyroid hormone regulation.
| Materials and Methods | ▴Top |
Study design
An observational study with a cross-sectional analytical approach was conducted at the University Hospital “Dr. Georgi Stranski,” Pleven. Participants were recruited from different clinical departments, including the Clinic of Pulmonology and Phthisiology and the Clinic of Endocrinology and Metabolic Diseases, according to disease stage and clinical presentation. The groups were not age-matched, and patients in the acute COVID-19 group were older than those in the post-COVID and reference groups, consistent with the age distribution of hospitalized patients with acute SARS-CoV-2 infection during the recruitment period.
Study population
The study included 101 participants, allocated into three groups: patients with active COVID-19 infection confirmed by a positive reverse transcription polymerase chain reaction (RT-PCR) test (group 1, n = 33); individuals with thyroid dysfunction following recovery from COVID-19 (group 2, n = 35); and a reference group of patients without prior COVID-19 infection and a negative PCR test (group 3, n = 33). All participants were evaluated for key immune response markers, including interleukin-17A (IL-17A), interleukin-7 (IL-7), interleukin-10 (IL-10), and IFN-γ, as well as for factors related to hypoxia and antioxidant defense, namely HIF-1α and NFE2L2. Thyroid function was assessed by measuring FT3, free thyroxine (FT4), thyroglobulin (Tg), TSH, and TPO.
The following inclusion and exclusion criteria were applied.
Inclusion criteria
For all three groups: Individuals aged ≥ 18 years, unvaccinated against COVID-19. For group 1: patients with confirmed active COVID-19 infection verified by a positive RT-PCR test. For group 2: individuals with a history of COVID-19 (confirmed by RT-PCR), a post-infectious interval of more than 6 months, and newly diagnosed thyroid dysfunction during this period (including subacute or painless thyroiditis, Graves’ disease, Hashimoto’s thyroiditis, or nodular goiter). For group 3: individuals with no history of COVID-19 and a negative RT-PCR test.
Exclusion criteria
For all three groups: age < 18 years, pregnancy, and breastfeeding. For group 2: recent COVID-19 infection (< 6 months), long COVID syndrome, prior COVID-19 vaccination, autoimmune diseases, type 2 diabetes mellitus, severe decompensated systemic diseases, active malignancy, treatment with glucocorticoids, biological agents, immunosuppressants, or cytostatics, and pre-existing thyroid disease prior to COVID-19. For group 3, an additional exclusion criterion was a documented history of prior COVID-19 infection.
Definitions and diagnostic criteria
COVID-19 infection was diagnosed based on a positive SARS-CoV-2 RT-PCR result obtained from a nasopharyngeal/oropharyngeal swab at hospital admission. Disease severity was classified according to WHO criteria [22].
Thyroid function was assessed by measuring TSH, FT3, FT4, and Tg. Patients were classified according to the recommendations of the American Thyroid Association and the Bulgarian Society of Endocrinology [23, 24]. Euthyroidism was defined as TSH 0.20–4.20 mIU/L, FT3 3.10–6.80 pmol/L, FT4 11.90–21.60 pmol/L, and Tg 3.50–77.0 ng/mL, based on the reference ranges of the hospital laboratory.
Thyroid dysfunction was defined as any deviation in TSH, FT3, or FT4 levels and classified as subclinical (isolated TSH abnormality) or overt (abnormal TSH with altered FT3 and/or FT4). Autoimmune thyroid disease was diagnosed according to established clinical guidelines, based on thyroid hormone levels, antibody profiles, and thyroid ultrasound findings.
Clinical and laboratory assessments
Blood samples were collected at the moment of hospital admission prior to the initiation of therapy in patients with active COVID-19 (group 1), and at the enrollment in the study in post-COVID (group 2) and non-infected individuals (group 3). Serum and plasma samples were stored at −80 °C until analysis.
The following immuno-inflammatory markers were assessed: IL-17A, IL-7, IL-10, IFN-γ, HIF-1α, and NFE2L2. Their serum concentrations were measured using enzyme-linked immunosorbent assay (ELISA) with commercial kits (Elabscience Biotechnology Inc., Houston, TX, USA), according to the manufacturer’s instructions.
Thyroid-related parameters (TSH, FT3, FT4, Tg, and TPO) were assessed. While TSH, FT3, FT4, and Tg were measured using ECLIA, TPO concentrations were determined by a quantitative ELISA method using a commercial kit (Human TPO ELISA Kit, Antibodies.com, Cambridge, UK; catalog No. A101827), according to the manufacturer’s instructions.
Ultrasonographic examination of the thyroid gland was performed using an Esaote MyLabSigma ultrasound system. The European Thyroid Association (ETA) classification for ultrasound assessment of thyroid nodules and stratification of the need for fine-needle aspiration biopsy (FNAB) and malignancy risk (EU-TIRADS) was applied [25].
Statistical analysis
Data were processed by IBM SPSS Statistics, version 25. Quantitative variables were presented as mean ± standard deviation (SD) for normally distributed data and as median (Me) with interquartile range (IQR) for asymmetrical distributions. Normality was assessed by Kolmogorov–Smirnov and Shapiro–Wilk tests. Since most variables in at least one group showed non-normal distribution, assessment and comparisons were performed through non-parametric methods. Differences among the three independent groups were assessed by Kruskal–Wallis H test. When significant differences were identified (P ≤ 0.05), post hoc pairwise comparisons were performed by Mann–Whitney U test. Spearman’s rank correlation coefficient was used to assess associations between quantitative immunological, inflammatory, and hormonal parameters, with P < 0.05 considered statistically significant. Receiver operating characteristic (ROC) analysis was performed to evaluate the discriminatory performance of HIF-1α.
Ethical considerations
The study protocol was approved by the Ethics Committee of Scientific Research at the Medical University of Pleven (Protocol No. 72/23.06.23). All procedures were conducted in accordance with the ethical principles of the Helsinki Declaration.
| Results | ▴Top |
Basic demographic and clinical characteristics of the study groups
Age and sex distributions across the study groups are presented in Table 1. Overall, the active COVID-19 group included older participants, consistent with the clinical profile of hospitalized patients, while the post-COVID and reference groups comprised younger individuals. A higher proportion of females was observed in the reference group, whereas groups 1 and 2 showed a more balanced sex distribution.
 Click to view | Table 1. Demographic Characteristics of the Participants by Group |
The severity of COVID-19 infection in group 1 was assessed according to the WHO clinical classification [22]. Among the 33 patients, a mild form of infection was identified in six (18.18%), a moderate form in eight (24.24%), a severe form in 16 (48.48%), and a critical form in three patients (9.09%). The case fatality rate in group 1 was 6.06% (two deaths).
Endocrine markers: thyroid status of the studied patients
Thyroid hormone profile
The analysis of thyroid parameters revealed statistically significant differences between the studied groups only in terms of FT3 and TSH levels (Table 2).
 Click to view | Table 2. Thyroid Parameters in the Three Study Groups |
TSH levels differed between the study groups (Table 2), with the highest values observed in group 3. A significant difference was identified between group 1 and group 3, while values in groups 1 and 2 were lower and relatively similar. A similar pattern was observed for FT3 levels, with the lowest values in group 1 and higher levels in group 2 and group 3. Significant differences were found between group 1 and group 2, as well as between group 1 and group 3.
FT4, Tg, and TPO levels did not differ significantly among the three groups. FT4 level was almost equal in group 1 and group 3, with a higher values in post-COVID patients. Tg levels remained relatively stable across the three groups. TPO level showed no significant between-group differences, although higher values were observed in reference group.
Thyroid status in patients with active COVID-19 infection (group 1)
Among the 33 patients in group 1, heterogeneous alterations in thyroid function were observed. Reduced FT3 levels were identified in 12 (36.4%) patients, representing the most frequent abnormality, while 16 (48.5%) patients remained euthyroid (Fig. 1). Less common findings included subclinical and overt thyroid dysfunction in both directions—hypothyroidism and thyrotoxicosis. Overall, these results indicate a predominance of low FT3 syndrome accompanied by variable thyroid functional patterns during the acute phase of the infection.
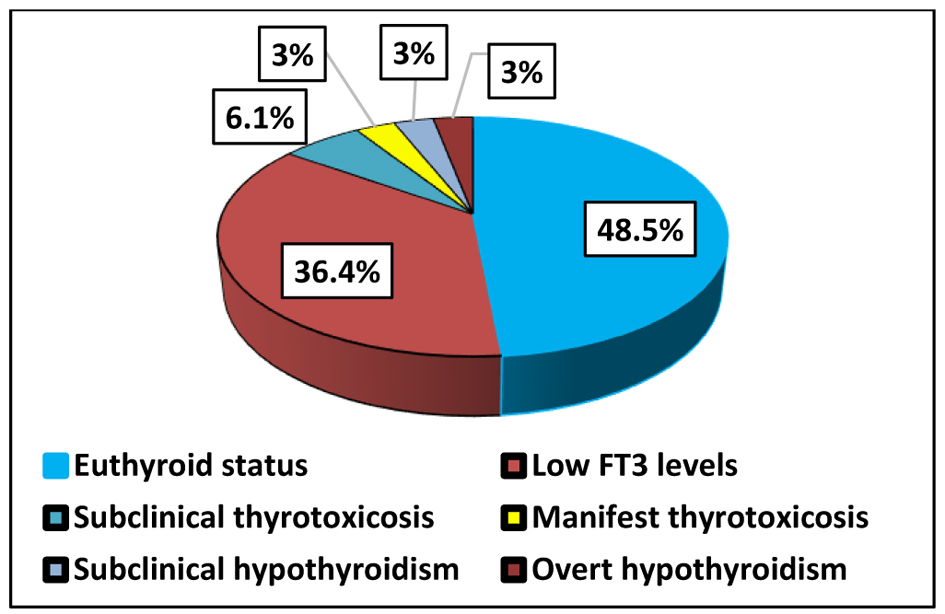 Click for large image | Figure 1. Thyroid status in patients with active COVID-19 infection (group 1). COVID-19: coronavirus disease 2019. |
Thyroid status in the post-COVID group (group 2)
In the post-COVID group (n = 35), endocrine disturbances were frequently observed, predominantly in the form of autoimmune thyroid disorders. Based on TSH, FT3, and FT4 levels, 21 patients were euthyroid (60.0%), followed by subclinical hypothyroidism (20.0%), while overt dysfunction and thyrotoxic states were less common (Fig. 2). In line with these findings, levothyroxine therapy was initiated in 13 patients with clinically significant hypothyroid dysfunction. This distribution indicates a predominance of euthyroid and subclinically hypothyroid status in the post-COVID population, whereas thyrotoxic conditions were less frequently observed.
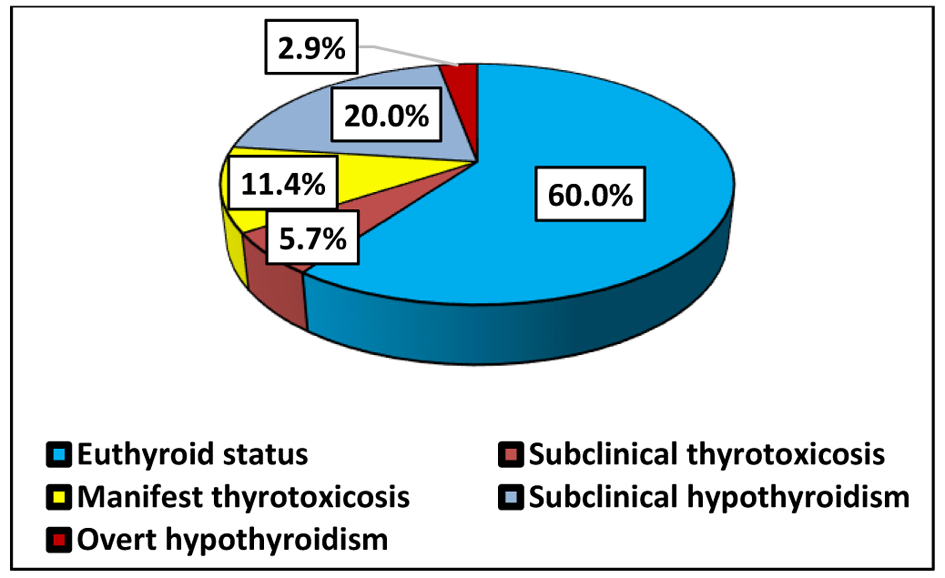 Click for large image | Figure 2. Functional thyroid status in post-COVID patients (group 2). COVID: coronavirus disease. |
The most common diagnosis was Hashimoto’s thyroiditis, observed in 23 patients (65.7%), including one case of thyroid-associated ophthalmopathy (TAO), highlighting the potential for extrathyroidal immunological hyperreactivity in the post-infectious period. Subacute thyroiditis (17.1%) and Graves’ disease (8.6%) were less frequent, reflecting a broad spectrum of post-infectious thyroid involvement (Fig. 3).
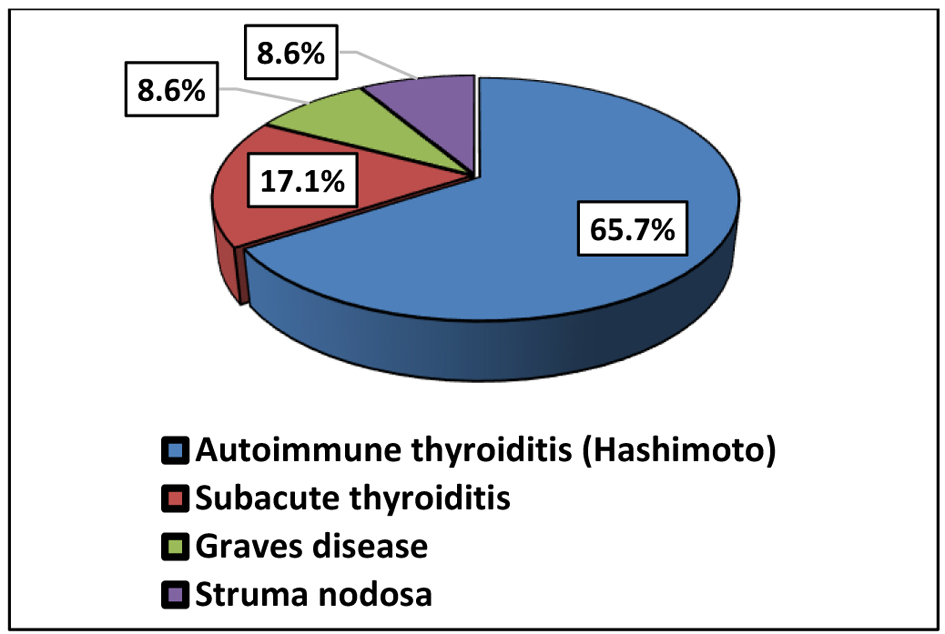 Click for large image | Figure 3. Thyroid diagnosis in post-COVID patients (group 2). COVID: coronavirus disease. |
Ultrasound findings were consistent with inflammatory and autoimmune changes, most commonly demonstrating a heterogeneous and hypoechoic thyroid parenchyma with altered vascularization. In patients with subacute thyroiditis, focal hypoechoic areas with reduced or absent blood flow were observed, corresponding to the destructive inflammatory process, whereas Graves’ disease was characterized by diffusely increased intraparenchymal vascularization on Doppler examination.
Nodular goiter was identified in three patients (8.6%). The nodules were classified as EU-TIRADS 2 in two patients, while one patient had a nodule classified as EU-TIRADS 3.
Key regulators of the immune response
To assess the characteristics of the immune response in COVID-19, we evaluated a targeted panel of pro-inflammatory (IL-17A), anti-inflammatory (IL-10), and dual-function cytokine (IL-7), as well as interferon, which are considered key mediators of immune dysregulation and may influence thyroid functional status (Table 3).
 Click to view | Table 3. Selected Cytokines Associated With Immune Response Differences Between Groups |
IL-17A and IFN-γ levels differed significantly across the study groups (Table 3). The highest medians were observed in the reference group, intermediate in patients with active COVID-19, and the lowest concentrations in the post-COVID group. Significant differences were identified between group 1 and group 2, as well as between group 2 and group 3, whereas no significant difference was found between group 1 and group 3.
IL-10 and IL-7 levels also differed significantly among the study groups (Table 3). IL-10 showed the highest levels in patients with active infection, intermediate values in the reference group, and the lowest concentrations in the post-COVID group, with significant differences between group 1 and group 2 and between group 2 and group 3. In contrast, IL-7 levels were highest in the reference group, intermediate in patients with active COVID-19, and lowest in the post-COVID group, with all pairwise comparisons reaching statistical significance.
Intracellular hypoxia and antioxidant defense
HIF-1α and NFE2L2 levels differed significantly across the study groups (Table 4), with the highest values observed in patients with active COVID-19 infection. HIF-1α levels were significantly higher in group 1 than in both group 2 and group 3, while no significant difference was observed between the latter two groups. Similarly, NFE2L2 levels were highest during the acute phase of COVID-19 and showed a significant reduction in the post-COVID group, reaching values comparable to those in individuals without prior SARS-CoV-2 exposure.
 Click to view | Table 4. Markers of Oxidative and Hypoxic Stress in the Three Study Groups |
Given the substantially increased HIF-1α levels during the active phase of infection, an ROC analysis was conducted to assess its diagnostic performance (Table 5). The table presents ROC-derived diagnostic performance parameters, including area under the curve (AUC), 95% confidence interval (CI), optimal cut-off value, sensitivity, and specificity.
 Click to view | Table 5. ROC Analysis of HIF-1α to Distinguish Patients With Active COVID-19 Infection From Healthy Individuals |
The analysis demonstrated a good diagnostic ability of HIF-1α to discriminate patients with active COVID-19 infection (Fig. 4).
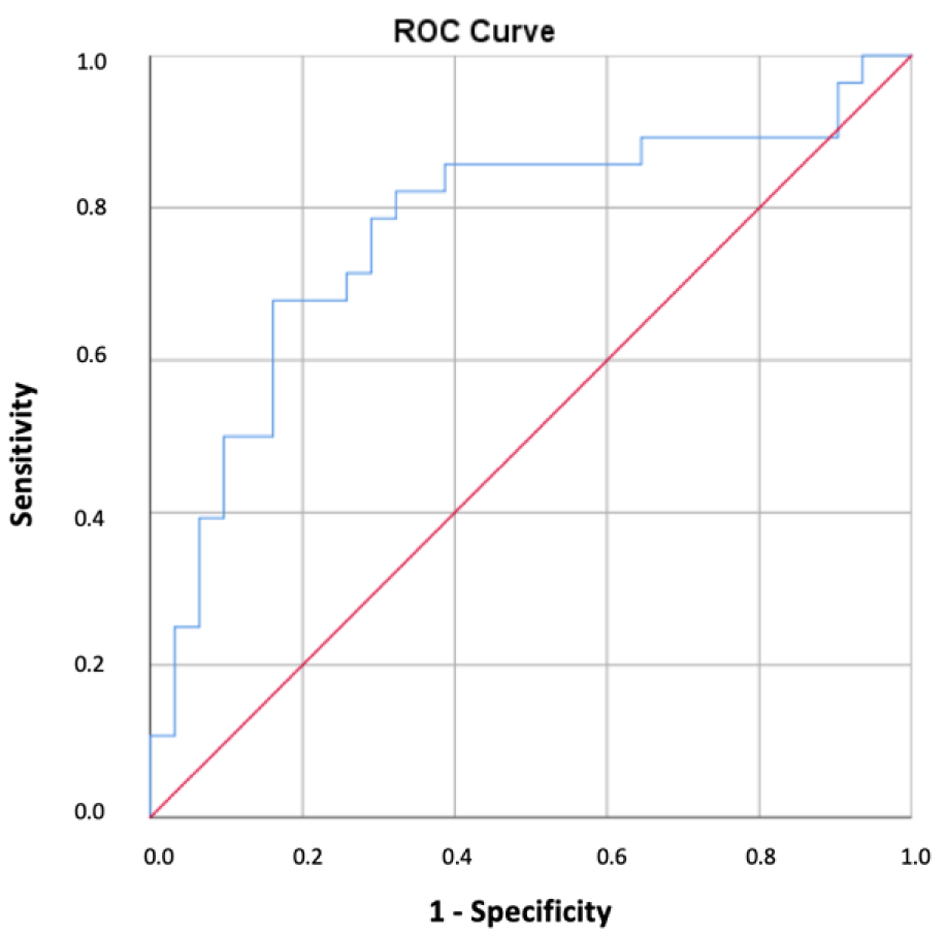 Click for large image | Figure 4. ROC curve analysis for determining the optimal cut-off value of HIF-1α in COVID-19 disease. COVID-19: coronavirus disease 2019; HIF-1α: hypoxia-inducible factor-1 alpha; ROC: receiver operating characteristic. |
Correlations between thyroid and immunological parameters
The correlation analysis between the immunological and thyroid parameters also demonstrated statistically significant associations within the individual groups (Table 6).
 Click to view | Table 6. Correlations Between Immunological and Thyroid Parameters in the Studied Groups |
In patients with active COVID-19, IL-7 showed a negative correlation with FT3 (r = −0.440, P = 0.021) and a positive correlation with TPO (r = 0.390, P = 0.049).
In the post-COVID group, a positive correlation was observed between FT4 and IL-10 (r = 0.438, P = 0.014).
In individuals without prior COVID-19, IL-10 demonstrated negative correlations with both FT3 (r = −0.631, P = 0.001) and FT4 (r = −0.418, P = 0.042).
| Discussion | ▴Top |
Pathophysiological mechanisms of thyroid dysregulation in COVID-19
In our study, low FT3 was the most frequent abnormality in patients with active COVID-19, although the typical pattern of ESS, characterized by low FT3 in combination with low or normal FT4, was not consistently observed. In a substantial proportion of patients, FT4 levels were at or above the upper reference limit, suggesting a more complex pattern of thyroid dysfunction. This observation is consistent with previous studies suggesting that COVID-19-related thyroid alterations may reflect a combination of destructive thyroiditis and cytokine-mediated impairment of peripheral deiodination [26, 27]. Our results are also consistent with Muller et al [28], who reported elevated or normal FT4 concentrations in the presence of low TSH and FT3 values among COVID-19 patients admitted to intensive care units. This pattern, described as “thyroxine thyrotoxicosis” in atypical COVID-19-related thyroiditis, further illustrates the complex interplay between virus-induced inflammation, hormonal regulation, and thyroid hormone metabolism [28].
In addition, hypoxia and oxidative stress appear to play a key role in limiting peripheral T3 production. The elevated HIF-1α levels observed in our study support the involvement of hypoxia-driven mechanisms, including increased type 3 deiodinase (DIO3) activity and enhanced T3 inactivation. HIF-1α, a key transcription factor mediating cellular responses to hypoxia, has been associated with enhanced inflammatory responses and immune dysregulation in COVID-19 [29–31]. Tian et al [32] and da Silva et al [33] further demonstrated that HIF-1α activation correlates with inflammatory burden and disease severity. Hypoxia-driven activation of HIF-1α may directly contribute to thyroid hormone alterations, as shown by Simonides et al [20], who reported that HIF-1α-dependent activation of DIO3 promotes T3 inactivation. These mechanisms link oxygen imbalance to endocrine dysfunction and support the role of hypoxia in COVID-19-related thyroid alterations. Consistent with this interpretation, ROC analysis in our study demonstrated that HIF-1α had good discriminatory performance in distinguishing patients with active COVID-19 from the reference group (AUC = 0.771). This finding may further support its relevance as a potential marker of hypoxia-related pathophysiological activity during acute infection and its association with lower FT3 levels in a substantial proportion of patients in group 1 (36.4%).
Furthermore, impaired antioxidant defense, reflected by altered NFE2L2 expression, may exacerbate oxidative stress and contribute to sustained immune and metabolic dysregulation. NFE2L2, a key regulator of antioxidant responses [34, 35], was increased during active disease, consistent with a compensatory response to oxidative stress [18]. In contrast, lower levels in the post-COVID phase may indicate incomplete recovery of antioxidant capacity and persistent redox imbalance [36, 37], thereby further contributing to thyroid dysfunction.
In parallel, the proinflammatory and dysregulated cytokine responses observed in COVID-19, including alterations in IL-17A, IL-10, IL-7, and IFN-γ, may contribute to reduced deiodinase activity and diminished pituitary sensitivity to thyrotropin-releasing hormone (TRH), thereby suppressing peripheral T4 to T3 conversion, lowering FT3 levels, and promoting functional thyroid dysregulation [38].
Together, these findings suggest that reduced FT3 represents an adaptive response to systemic inflammatory and hypoxic stress rather than primary thyroid gland dysfunction.
IL-17A, a key cytokine produced by Th17 cells, plays an important role in the immunopathogenesis of COVID-19 and autoimmune inflammatory processes [39–41]. In our study, decreased IL-17A levels were observed both during active infection and in the post-COVID phase, suggesting suppression of the Th17 response, likely reflecting SARS-CoV-2–induced immune exhaustion. Similar findings have been reported by De Biasi et al [42]. Reduced IL-17A levels may contribute to thyroid dysfunction by altering proinflammatory signaling pathways, including NF-κB–mediated mechanisms involved in thyroid hormone regulation. In this context, the decreased FT3 levels observed during the acute phase may partly reflect impaired Th17-mediated immune signaling.
IL-10, a key anti-inflammatory cytokine, was elevated during the acute phase of COVID-19, consistent with a compensatory immunoregulatory response to viral infection and activation of Th1- and Th2-mediated pathways [43, 44]. Similar findings have been reported by Lucas et al [45]. In contrast, persistently reduced IL-10 levels in the post-COVID phase suggest impaired regulatory control and ongoing immune imbalance [46], which may contribute to the development of autoimmune thyroid disorders, as supported by recent evidence [46].
Despite these observations, interpretation of IL-10 levels remains challenging due to the lack of universally accepted reference ranges. Compared with the assay-specific reference interval (4.8–9.8 pg/mL), IL-10 levels in our study were reduced across all groups [47], which may reflect diminished anti-inflammatory activity or receptor/post-receptor resistance under hypoxic or stress conditions. IL-10 may exert a predominantly protective effect through suppression of IFN-γ–mediated pathways, as suggested by de la Vega et al [48]. The moderate positive correlation between FT4 and IL-10 observed during the post-COVID phase further supports the role of IL-10 in immune–thyroid interactions, whereas the negative correlation between IL-10 and FT3 in the reference group may reflect physiological immunoendocrine balance.
IL-7, a lymphopoietic cytokine essential for T-cell survival and immune homeostasis [49], showed moderately elevated levels in the early phase of infection, consistent with its compensatory role in response to lymphopenia. However, persistently lower IL-7 levels in both the active and post-COVID groups compared with the reference group suggest incomplete restoration of T-cell homeostasis. Notably, both significant correlations involving IL-7 were observed in the active COVID-19 group: the negative correlation with FT3 supports an interaction between immune activation and thyroid hormone metabolism, possibly reflecting cytokine-mediated suppression of deiodinase activity and impaired peripheral T4 to T3 conversion [13], whereas the moderate positive correlation with TPO may indicate a link between immune activation and thyroid follicular cell function, consistent with findings by Seo et al [50]. Persistent IL-7 reduction in the post-COVID phase may contribute to impaired immune tolerance and the development of autoimmune thyroid disorders, as also suggested by Motylewska et al [51].
IFN-γ, a key Th1 cytokine involved in antiviral immune responses [52, 53], showed elevated levels during the acute phase but decreased significantly in the post-COVID period. This pattern may reflect a transition from early immune activation to post-infectious immune suppression and persistent Th1 dysregulation. Disruption of the Th1/Th2 balance is a key mechanism in autoimmune thyroid diseases, with IFN-γ acting as a central mediator [54]. Chronically reduced IFN-γ levels have also been associated with metabolic and oxidative stress–related alterations that may further promote autoimmune thyroid processes [55].
The FT3/FT4 ratio as an indirect marker of peripheral thyroid sensitivity
In the studies by Le et al [56] and Bian et al [57], the FT3/FT4 ratio was shown to reflect the efficiency of peripheral T4 to T3 conversion and the tissue bioavailability of thyroid hormones, thereby serving as an indirect marker of peripheral thyroid hormone sensitivity. Consistent with these findings, Lang et al [58] also employed the FT3/FT4 ratio as a surrogate marker of peripheral thyroid sensitivity, demonstrating its association with all-cause and cardiovascular mortality, as well as with the risk of cardiovascular disease.
In our study, a tendency toward a relative increase in FT4 was observed during the acute phase of infection, compared to the low FT3 levels, which likely represents a relative phenomenon reflecting a functional imbalance between the two hormones. The calculated FT3/FT4 ratio in the active infection group was reduced (0.14). This confirms the presence of hormonal imbalance and is indicative of suppressed peripheral conversion of T4 to T3—a characteristic finding of the low T3 syndrome. Our results are consistent with data from other authors. Milo et al [59] demonstrated that both serum FT3 levels and the FT3/FT4 ratio were significantly lower in non-survivors and correlated negatively with C-reactive protein (CRP) and the neutrophil-to-lymphocyte ratio (NLR), supporting their prognostic potential in severe COVID-19 infection.
In the post-COVID group, a partial restoration of the hormonal ratio (FT3/FT4 = 0.21) was observed, indicating normalization of deiodinase activity and gradual compensation of the thyroid axis following the resolution of acute inflammatory stress. In individuals without a history of COVID-19, the FT3/FT4 ratio reached 0.24, suggesting more favorable hormonal regulation and more efficient peripheral conversion.
It is important to note that, despite numerous studies demonstrating associations between the FT3/FT4 ratio and various clinical or prognostic outcomes, no internationally established reference ranges currently exist for this parameter. Reported values vary widely, depend on methodology and population characteristics, and should therefore be interpreted within the context of the clinical presentation and the available reference intervals for FT3 and FT4 individually [58].
Serum TPO levels as an indirect marker of oxidative stress and inflammatory dysregulation
The simultaneous assessment of Tg and TPO indicated preserved thyroid hormone synthesis, as no significant deviations in their serum levels were observed across the study groups. However, the elevated HIF-1α and NFE2L2 levels in patients with active COVID-19 suggest the presence of pronounced hypoxic and oxidative stress, which may affect thyroid function at a functional rather than structural level.
Given the sensitivity of TPO–dependent hormonogenesis to oxidative conditions, it is plausible that thyroid hormone synthesis may be affected even in the absence of significant changes in circulating TPO levels. Under such conditions, inflammatory and hypoxia-related mechanisms may interfere with iodine organification and thyroid hormone synthesis at the cellular level [60].
This observation is supported by evidence that proinflammatory cytokines can not only impair thyroid hormone synthesis by reducing deiodinase activity and T3 and Tg production, but also suppress TSH-stimulated TPO expression in thyrocytes [9]. In this setting, altered cytokine patterns involving IL-17A, IL-10, IL-7, and IFN-γ may further sustain thyroid dysfunction by shifting the local balance toward persistent inflammation and insufficient immune regulation within the thyroid microenvironment. Reduced IL-10 and IL-7, together with altered IL-17A and IFN-γ levels, may therefore contribute to delayed thyroid recovery and persistent low-grade inflammation [27].
Notably, the lower TPO levels observed in the post-COVID group could reflect persistent subclinical inflammatory and oxidative imbalance, possibly indicating incomplete recovery of thyroid functional capacity despite apparent normalization of circulating thyroid hormones. Taken together, these findings raise the possibility that TPO may serve as an indirect marker of ongoing oxidative and immune dysregulation in the post-COVID phase, potentially linking persistent redox imbalance with delayed restoration of thyroid homeostasis and a tendency toward autoimmune thyroid dysfunction.
Limitations
The present study should be interpreted in light of several potential limitations. First, the relatively small number of patients in each group (total sample size n = 101) limits the reliability of statistical inferences and the extent to which the findings can be generalized to a broader population. A significant age difference between the study groups may represent a potential confounding factor, particularly given the known influence of age on both immune and endocrine responses. Second, the lack of longitudinal follow-up in both groups of patients with active COVID-19 infection and post-COVID individuals precludes a comprehensive assessment of the temporal evolution of thyroid dysfunction, including recovery kinetics and whether the observed alterations are transient or persistent over time.
The study was conducted as a single-center investigation during a period when various Omicron subvariants were the predominant circulating strains of SARS-CoV-2, which may restrict the external validity of the findings and the ability to directly extrapolate our findings to future variants that may differ in their immunological and clinical characteristics. Despite these limitations, our study provides important insights into the complex interplay between inflammatory, oxidative, and endocrine mechanisms during and after SARS-CoV-2 infection, and outlines key pathophysiological processes associated with post-COVID thyroid dysfunction.
Conclusion
SARS-CoV-2 infection appears to induce transient thyroid hormonal alterations primarily through inflammatory and hypoxia-related mechanisms. These findings support the concept of virus-induced euthyroid sick syndrome during acute infection, with reduced FT3 representing the most consistent abnormality, and highlight the potential for post-infectious thyroid dysfunction.
Acknowledgments
We wish to thank Prof. Milena Atanasova Atanasova BD and Assoc. Prof. Adelaida Lazarova Ruseva MD for their assistance with laboratory analysis of the samples and Ass. Prof. Mircho Vukov for the statistical analysis.
Financial Disclosure
The study was conducted with the support of a research grant from the Medical University - Pleven (Project No. D7/2023): “Relationship between thyroid and adrenal cortical hormones and inflammatory status in COVID-19”.
Conflict of Interest
The authors declare that they have no conflict of interest that could be perceived as prejudicing the impartiality of the research reported. The sponsor had no role in study design, in the collection, analysis and interpretation of data; in the writing of the report, and in the decision to submit the article for publication.
Informed Consent
Written informed consent was obtained from all participants prior to their enrollment in the study.
Author Contributions
Malvina Todorova: conceptualization, methodology, software, formal analysis, investigation, data curation, writing – original draft, visualization. Victoria Tsvetkova: methodology, software, formal analysis, investigation, data curation. Katya Todorova: conceptualization, methodology, software, investigation, supervision, project administration, writing – review and editing. Milena Atanasova Atanasova: methodology, validation, formal analysis. Adelaida Lazarova Ruseva: methodology, validation, formal analysis. All authors read and approved the final manuscript.
Data Availability
The authors declare that data supporting the findings of thisstudy are available within the article.
| References | ▴Top |
- Sarkesh A, Daei Sorkhabi A, Sheykhsaran E, Alinezhad F, Mohammadzadeh N, Hemmat N, Bannazadeh Baghi H. Extrapulmonary clinical manifestations in COVID-19 patients. Am J Trop Med Hyg. 2020;103(5):1783-1796.
doi pubmed - Zheng J, Cui Z, Shi N, Tian S, Chen T, Zhong X, Qiu K, et al. Suppression of the hypothalamic-pituitary-thyroid axis is associated with the severity of prognosis in hospitalized patients with COVID-19. BMC Endocr Disord. 2021;21(1):228.
doi pubmed - Kirikae H, Tezuka Y, Okamoto M, Furuta G, Omata K, Ono Y, Tanaka T, et al. Hypothalamic-pituitary-adrenal dynamics in early-stage COVID-19 observed in a case with arginine vasopressin deficiency. BMC Endocr Disord. 2025;25(1):169.
doi pubmed - D'Onofrio N, Scisciola L, Sardu C, Trotta MC, De Feo M, Maiello C, Mascolo P, et al. Glycated ACE2 receptor in diabetes: open door for SARS-COV-2 entry in cardiomyocyte. Cardiovasc Diabetol. 2021;20(1):99.
doi pubmed - Matarese A, Gambardella J, Sardu C, Santulli G. miR-98 regulates TMPRSS2 expression in human endothelial cells: key implications for COVID-19. Biomedicines. 2020;8(11):462.
doi pubmed - Cabuk SA, Cevher AZ, Kucukardali Y. Thyroid function during and after COVID-19 infection: a review. touchREV Endocrinol. 2022;18(1):58-62.
doi pubmed - Murugan AK, Alzahrani AS. SARS-CoV-2: emerging role in the pathogenesis of various thyroid diseases. J Inflamm Res. 2021;14:6191-6221.
doi pubmed - Zhou Q, Zhang L, Dong Y, Wang Y, Zhang B, Zhou S, Huang Q, et al. The role of SARS-CoV-2-mediated NF-kappaB activation in COVID-19 patients. Hypertens Res. 2024;47(2):375-384.
doi pubmed - Scappaticcio L, Pitoia F, Esposito K, Piccardo A, Trimboli P. Impact of COVID-19 on the thyroid gland: an update. Rev Endocr Metab Disord. 2021;22(4):803-815.
doi pubmed - Attiq A, Afzal S, Wahab HA, Ahmad W, Kandeel M, Almofti YA, Alameen AO, et al. Cytokine storm-induced thyroid dysfunction in COVID-19: insights into pathogenesis and therapeutic approaches. Drug Des Devel Ther. 2024;18:4215-4240.
doi pubmed - Wang W, Su X, Ding Y, Fan W, Zhou W, Su J, Chen Z, et al. Thyroid function abnormalities in COVID-19 patients. Front Endocrinol (Lausanne). 2020;11:623792.
doi pubmed - Li MY, Li L, Zhang Y, Wang XS. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis Poverty. 2020;9(1):45.
doi pubmed - Panesar A, Gharanei P, Khovanova N, Young L, Grammatopoulos D. Thyroid function during COVID-19 and post-COVID complications in adults: a systematic review. Front Endocrinol (Lausanne). 2024;15:1477389.
doi pubmed - Lee S, Farwell AP. Euthyroid sick syndrome. Compr Physiol. 2016;6(2):1071-1080.
doi pubmed - Yanachkova V, Stankova T, Staynova R. Thyroid dysfunction as a long-term post-COVID-19 complication in mild-to-moderate COVID-19. Biotechnol Biotechnol Equip. 2023;37:194-202.
doi - Hou J, Zhang Y, Zhang B, Zhao S, Wu H, Li X, Li M, et al. Emerging evidence from China indicates SARS-CoV-2 may be associated with newly diagnosed Hashimoto’s thyroiditis: a clinical study of 54 patients with COVID-19 complicated by newly diagnosed disease. Front Endocrinol (Lausanne). 2026;17:1749196.
doi - Fallahi P, Elia G, Ragusa F, Paparo SR, Patrizio A, Balestri E, Mazzi V, et al. Thyroid autoimmunity and SARS-CoV-2 infection. J Clin Med. 2023;12(19):6365.
doi pubmed - Cuadrado A, Pajares M, Benito C, Jimenez-Villegas J, Escoll M, Fernandez-Gines R, Garcia Yague AJ, et al. Can activation of NRF2 be a strategy against COVID-19? Trends Pharmacol Sci. 2020;41(9):598-610.
doi pubmed - Devaux CA, Raoult D. The impact of COVID-19 on populations living at high altitude: Role of hypoxia-inducible factors (HIFs) signaling pathway in SARS-CoV-2 infection and replication. Front Physiol. 2022;13:960308.
doi pubmed - Simonides WS, Mulcahey MA, Redout EM, Muller A, Zuidwijk MJ, Visser TJ, Wassen FW, et al. Hypoxia-inducible factor induces local thyroid hormone inactivation during hypoxic-ischemic disease in rats. J Clin Invest. 2008;118(3):975-983.
doi pubmed - Malik J, Malik A, Javaid M, Zahid T, Ishaq U, Shoaib M. Thyroid function analysis in COVID-19: A retrospective study from a single center. PLoS One. 2021;16(3):e0249421.
doi pubmed - Coronavirus disease 2019 (COVID-19) treatment guidelines. Bethesda (MD); 2021.
pubmed - Jonklaas J, Bianco AC, Bauer AJ, Burman KD, Cappola AR, Celi FS, Cooper DS, et al. Guidelines for the treatment of hypothyroidism: prepared by the american thyroid association task force on thyroid hormone replacement. Thyroid. 2014;24(12):1670-1751.
doi pubmed - Bulgarian Society of Endocrinology. Recommendations for good practice in thyroid diseases. Sofia: Bulgarian Society of Endocrinology; 2019. Bulgarian.
- Russ G, Bonnema SJ, Erdogan MF, Durante C, Ngu R, Leenhardt L. European thyroid association guidelines for ultrasound malignancy risk stratification of thyroid nodules in adults: the EU-TIRADS. Eur Thyroid J. 2017;6(5):225-237.
doi pubmed - Lania A, Sandri MT, Cellini M, Mirani M, Lavezzi E, Mazziotti G. Thyrotoxicosis in patients with COVID-19: the THYRCOV study. Eur J Endocrinol. 2020;183(4):381-387.
doi pubmed - Kolpakova EA, Elfimova AR, Nikankina LV, Troshina EA. [The role of systemic immune activation in the development of thyroid dysfunction in COVID-19]. Ter Arkh. 2022;94(10):1136-1142.
doi pubmed - Muller I, Cannavaro D, Dazzi D, Covelli D, Mantovani G, Muscatello A, Ferrante E, et al. SARS-CoV-2-related atypical thyroiditis. Lancet Diabetes Endocrinol. 2020;8(9):739-741.
doi pubmed - Yoon D, Pastore YD, Divoky V, Liu E, Mlodnicka AE, Rainey K, Ponka P, et al. Hypoxia-inducible factor-1 deficiency results in dysregulated erythropoiesis signaling and iron homeostasis in mouse development. J Biol Chem. 2006;281(35):25703-25711.
doi pubmed - Malkov MI, Lee CT, Taylor CT. Regulation of the Hypoxia-Inducible Factor (HIF) by pro-inflammatory cytokines. Cells. 2021;10(9):2340.
doi pubmed - van Uden P, Kenneth NS, Rocha S. Regulation of hypoxia-inducible factor-1alpha by NF-kappaB. Biochem J. 2008;412(3):477-484.
doi pubmed - Tian M, Liu W, Li X, Zhao P, Shereen MA, Zhu C, Huang S, et al. HIF-1alpha promotes SARS-CoV-2 infection and aggravates inflammatory responses to COVID-19. Signal Transduct Target Ther. 2021;6(1):308.
doi pubmed - da Silva FPG, Matte R, Wiedmer DB, da Silva APG, Menin RM, Barbosa FB, Meneguzzi TAM, et al. HIF-1alpha pathway in COVID-19: a scoping review of its modulation and related treatments. Int J Mol Sci. 2025;26(9):4202.
doi pubmed - Baird L, Yamamoto M. The molecular mechanisms regulating the KEAP1-NRF2 pathway. Mol Cell Biol. 2020;40(13):e00099-e00020.
doi pubmed - Ahmed SM, Luo L, Namani A, Wang XJ, Tang X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim Biophys Acta Mol Basis Dis. 2017;1863(2):585-597.
doi pubmed - Sies H, Jones DP. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol. 2020;21(7):363-383.
doi pubmed - Muchtaridi M, Amirah SR, Harmonis JA, Ikram EHK. Role of nuclear factor erythroid 2 (Nrf2) in the recovery of long COVID-19 using natural antioxidants: a systematic review. Antioxidants (Basel). 2022;11(8):1551.
doi pubmed - Lu Y, Xing C, Zhang C, Lv X, Liu G, Chen F, Hou Z, et al. Promotion of IL-17/NF-kappaB signaling in autoimmune thyroid diseases. Exp Ther Med. 2023;25(1):51.
doi pubmed - Gaffen SL, Jain R, Garg AV, Cua DJ. The IL-23-IL-17 immune axis: from mechanisms to therapeutic testing. Nat Rev Immunol. 2014;14(9):585-600.
doi pubmed - Chong WP, Mattapallil MJ, Raychaudhuri K, Bing SJ, Wu S, Zhong Y, Wang W, et al. The cytokine IL-17A limits Th17 pathogenicity via a negative feedback loop driven by autocrine induction of IL-24. Immunity. 2020;53(2):384-397.e385.
doi pubmed - Kuwabara T, Ishikawa F, Kondo M, Kakiuchi T. The role of IL-17 and related cytokines in inflammatory autoimmune diseases. Mediators Inflamm. 2017;2017:3908061.
doi pubmed - De Biasi S, Meschiari M, Gibellini L, Bellinazzi C, Borella R, Fidanza L, Gozzi L, et al. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat Commun. 2020;11(1):3434.
doi pubmed - Couper KN, Blount DG, Riley EM. IL-10: the master regulator of immunity to infection. J Immunol. 2008;180(9):5771-5777.
doi pubmed - Carlini V, Noonan DM, Abdalalem E, Goletti D, Sansone C, Calabrone L, Albini A. The multifaceted nature of IL-10: regulation, role in immunological homeostasis and its relevance to cancer, COVID-19 and post-COVID conditions. Front Immunol. 2023;14:1161067.
doi pubmed - Lucas C, Wong P, Klein J, Castro TBR, Silva J, Sundaram M, Ellingson MK, et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020;584(7821):463-469.
doi pubmed - Di J, Ma X, Wu T, Qiao E, Salouti M, Zhong Y, Xia Q, et al. Association of COVID-19 with thyroid dysfunction and autoimmune thyroid disease: a retrospective cohort study. J Transl Autoimmun. 2024;9:100255.
doi pubmed - Sarris AH, Kliche KO, Pethambaram P, Preti A, Tucker S, Jackow C, Messina O, et al. Interleukin-10 levels are often elevated in serum of adults with Hodgkin's disease and are associated with inferior failure-free survival. Ann Oncol. 1999;10(4):433-440.
doi pubmed - de la Vega JR, Vilaplana JC, Biro A, Hammond L, Bottazzo GF, Mirakian R. IL-10 expression in thyroid glands: protective or harmful role against thyroid autoimmunity? Clin Exp Immunol. 1998;113(1):126-135.
doi pubmed - Chen D, Tang TX, Deng H, Yang XP, Tang ZH. Interleukin-7 Biology and Its Effects on Immune Cells: Mediator of Generation, Differentiation, Survival, and Homeostasis. Front Immunol. 2021;12:747324.
doi pubmed - Seo YB, Im SJ, Namkoong H, Kim SW, Choi YW, Kang MC, Lim HS, et al. Crucial roles of interleukin-7 in the development of T follicular helper cells and in the induction of humoral immunity. J Virol. 2014;88(16):8998-9009.
doi pubmed - Motylewska E, Niec M, Siejka A, Komorowski J, Lawnicka H, Swietoslawski J, Stepien H. Decreased serum level of IL-7 in patients with active Graves' disease. Cytokine. 2015;75(2):373-379.
doi pubmed - Todorovic-Rakovic N, Whitfield JR. Between immunomodulation and immunotolerance: The role of IFNgamma in SARS-CoV-2 disease. Cytokine. 2021;146:155637.
doi pubmed - Schroder K, Hertzog PJ, Ravasi T, Hume DA. Interferon-gamma: an overview of signals, mechanisms and functions. J Leukoc Biol. 2004;75(2):163-189.
doi pubmed - Caturegli P, Hejazi M, Suzuki K, Dohan O, Carrasco N, Kohn LD, Rose NR. Hypothyroidism in transgenic mice expressing IFN-gamma in the thyroid. Proc Natl Acad Sci U S A. 2000;97(4):1719-1724.
doi pubmed - Bae HR, Shin SK, Lee JY, Ko YJ, Kim S, Young HA, Kwon EY. Chronic low-level IFN-gamma expression disrupts mitochondrial complex I activity in renal macrophages: an early mechanistic driver of lupus nephritis pathogenesis. Int J Mol Sci. 2024;26(1):63.
doi pubmed - Le TN, Celi FS, Wickham EP, 3rd. Thyrotropin levels are associated with cardiometabolic risk factors in euthyroid adolescents. Thyroid. 2016;26(10):1441-1449.
doi pubmed - Bian N, Sun X, Zhou B, Zhang L, Wang Q, An Y, Li X, et al. Obese patients with higher TSH levels had an obvious metabolic improvement after bariatric surgery. Endocr Connect. 2021;10(10):1326-1336.
doi pubmed - Lang X, Li Y, Zhang D, Zhang Y, Wu N, Zhang Y. FT3/FT4 ratio is correlated with all-cause mortality, cardiovascular mortality, and cardiovascular disease risk: NHANES 2007-2012. Front Endocrinol (Lausanne). 2022;13:964822.
doi pubmed - Milo M, Di Dalmazi G, Febo F, Coluzzi S, Carrieri F, Terrenzio M, Ferrandu G, et al. Serum FT3 levels and FT3/FT4 ratio as predictors for poor prognosis in hospitalized COVID-19 patients. Endocrine Abstracts. 2022;81:P719.
doi - Gerard AC, Boucquey M, van den Hove MF, Colin IM. Expression of TPO and ThOXs in human thyrocytes is downregulated by IL-1alpha/IFN-gamma, an effect partially mediated by nitric oxide. Am J Physiol Endocrinol Metab. 2006;291(2):E242-253.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.