| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 16, Number 2, April 2026, pages 101-108
Common Bean (Phaseolus vulgaris) Husk Peptides Mitigate Cytokine Dysregulation and Inflammation in Obese Rats
Serhii Shchypanskyia, b , Daryna Krenytskaa, Mariana Kuznietsovaa, Nataliia Rakshaa, Olexii Savchuka
aBiochemistry Department, Educational and Scientific Center “Institute of Biology and Medicine”, Taras Shevchenko National University of Kyiv, 03127 Kyiv, Ukraine
bCorresponding Author: Serhii Shchypanskyi, Biochemistry Department, Educational and Scientific Center “Institute of Biology and Medicine”, Taras Shevchenko National University of Kyiv, 03127 Kyiv, Ukraine
Manuscript submitted December 22, 2025, accepted February 13, 2026, published online April 8, 2026
Short title: Bean Husk Peptides Reduce Inflammation in Obese Rats
doi: https://doi.org/10.14740/jem1627
| Abstract | ▴Top |
Background: Low-grade inflammation is believed to be an integral component and key marker of obesity and metabolic syndrome. Among various plant sources, the common bean (Phaseolus vulgaris) is a promising source of bioactive peptides, which could be effective in mitigating the negative effects of obesity.
Methods: Using an enzyme-linked immunosorbent assay procedure, we evaluated the relative levels of hypoxia inducible factor 1 subunit alpha (HIF-1α), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), interleukin 6 (IL-6), interleukin 8 (IL-8), interleukin 1 beta (IL-1β), tumor necrosis factor alpha (TNF-α), and resistin in the serum, visceral adipose tissue, liver, and kidneys of obese rats that either consumed P. vulgaris husk-derived peptides as a dietary supplement or did not.
Results: In the serum of obese rats, the results revealed increased levels of all assessed cytokines (except IL-8 and TNF-α), mainly resistin, IL-6, and IL-1β. In adipose tissue, the main changes were observed in the levels of resistin, IL-6, and TNF-α. In the liver and kidneys, alterations were primarily noted in the levels of HIF-1α and IL-8.
Conclusions: These cytokine profile patterns are indicative of the development of low-grade inflammation, insulin resistance, metabolic dysfunction-associated steatotic liver disease, and chronic kidney disease, which are inherent in obesity. At the same time, in animals that consumed peptides, cytokine levels were slightly above or at control levels, indicating the effectiveness of common bean husk peptides against obesity-induced low-grade inflammation.
Keywords: Phaseolus vulgaris; Obesity; Peptides; Low-grade inflammation; Cytokine
| Introduction | ▴Top |
According to the World Health Organization (WHO), almost half of the Earth’s population is overweight, and approximately every sixth person has obesity [1]. It poses a significant threat to the global healthcare system, rapidly spreading across all social groups. The expansion of adipose tissue during obesity entails concomitant diseases, such as type 2 diabetes, non-alcoholic fatty liver disease (NAFLD), hypertension, dyslipidemia, renal impairment, and depression [2]. However, there is another neglected, yet possibly a key marker of overweight and obesity—low-grade inflammation (LGI). Adipocyte hypertrophy triggers massive production of adipokines and inflammatory cytokines, laying the basis for LGI. In turn, LGI links adipose tissue distribution to the main cardiometabolic diseases listed above. By targeting LGI, clinicians could build a strategy to treat these metabolic complications and subsequently ease the metabolic burden for obese patients [3].
Modern studies have demonstrated that dietary plants can serve as a source of bioactive peptides. Some bioactive peptides from Chia seeds, walnuts, soybeans, legumes, and other plants have been found to have anti-obesity properties through the inhibition of key metabolic pathways, lipase and amylase inhibition, and antioxidant activity [4]. One of the most promising dietary plants for the production of bioactive peptides with anti-obesity properties is the common bean (Phaseolus vulgaris), aqueous extracts of which are known in folk medicine for their hypoglycemic effects [5]. This plant contains a protein named phaseolin—a natural inhibitor of pancreatic α-amylase, which also has antioxidant and antigenotoxic activities [6], as well as an anti-inflammatory effect in RAW264.7 cells and zebrafish larvae [7]. The protective effects of P. vulgaris against atherosclerosis and cardiovascular disease are linked to several bioactive compounds, particularly their high content of fiber (both soluble and insoluble) and a diverse range of polyphenols. These compounds work through multiple mechanisms to manage key risk factors. Since the production of bioactive peptides from whole P. vulgaris beans is not cost-effective, bean husk, as an alternative raw material, can be used to obtain peptides with similar properties. Recently, Kyznetsova et al showed that long-term administration of aqueous extracts of P. vulgaris bean husk lowers blood glucose levels and free radical production in diabetic rats [8]. Shchypanskyi et al also showed that acidic hydrolysis of P. vulgaris bean husk aqueous extract produces peptides with molecular weights of 192–610 Da, which have antioxidant (2,2-diphenyl-1-picrylhydrazyl (DPPH)-scavenging and ferric reducing antioxidant power (FRAP)) activity [9].
This study aims to expand the existing knowledge of common bean husk’s anti-obesity properties, shedding light on its anti-inflammatory effects in rats with modeled obesity. To achieve this, we have evaluated levels of main inflammatory markers and cytokines in the primary metabolically active organs, including blood, liver, kidneys, and visceral adipose tissue (VAT).
| Materials and Methods | ▴Top |
Bean husk peptide collection
Firstly, P. vulgaris bean husk extracts were prepared with a minor modification, following the technique outlined by Kyznetsova et al. The husks of kidney beans used in this study came from a local field in the Kyiv region, Ukraine. One liter of boiling distilled water was mixed with approximately 132 g of dried, powdered bean husks, and this mixture was then boiled in a covered container for 20 min. Afterwards, it was left to cool overnight. It was then filtered and centrifuged for 10 min at 1,000 × g. The resulting supernatant was lyophilized using a laboratory freeze-dryer, LyoQuest (Telstar, Barcelona, Spain), yielding about 8 g of lyophilized material.
To obtain the peptides from the extract, 8 g of lyophilized bean husk extract and 250 mL of 1 M acetic acid (502000, CARLO ERBA Reagents SAS) were mixed. The mixture was hydrolyzed with continuous stirring for 1 h. Then, the solution was heated to 100 °C, boiled for an hour, and subsequently cooled to room temperature. The resulting suspension was centrifuged at 2,800 × g for 45 min. The supernatant was dialyzed to remove non-protein impurities and then lyophilized.
Maintenance conditions of experimental animals
For the experiment, we used 30 non-linear albino male rats. All animals were maintained in the accredited vivarium at Taras Shevchenko National University of Kyiv. The experiment involving animals was approved by the special bioethics committee at Taras Shevchenko National University of Kyiv (Protocol No. 7 from November 21, 2024). All procedures followed the ethical guidelines and regulations for the use of laboratory animals outlined in the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (ECPE). Furthermore, the procedures followed Ukrainian legislation, specifically the law adopted on February 21, 2006 (No. 3447-IV), titled “On the Protection of Animals from Cruel Treatment”. Conditions for the animals’ care included a natural light/dark cycle, a temperature of 20–24 °C, and a humidity of 30–70%. The rats were randomly divided into three groups of 10 animals each. Five animals from each group were housed in individual cages, and they were all given free access to water and a standard pelleted rodent diet.
Induced obesity model establishment
After 3 days of acclimatization, we started the obesity induction process by feeding the animals a high-calorie diet. The high-calorie chow was made by softening the following ingredients in 2 L of hot water: standard rodent chow pellets (60%), chicken eggs (10%), lard (10%), crushed peanuts (5%), white sugar (9%), dry milk powder (5%), and sunflower oil (1%) [10]. This mixture yielded solid spheres with a diameter of around 4 cm. After drying, these spheres served as high-calorie meals for the experimental groups.
The 12-week obesity modeling trial consisted of two phases. At the beginning of the experiment, all rats were weighed and then randomly assigned to three groups of 10 and housed in cages containing five rats each. Standard rodent chow pellets and filtered water were given to the control group on an as-needed basis. Filtered water and the specially prepared, high-calorie meal mentioned above were provided to the other two groups ad libitum. The sixth week marked the start of the second stage. The feeding schedule for the other animal groups remained the same, but one of the experimental groups that received the high-calorie meal was also given aqueous solutions of P. vulgaris peptides (200 mg/kg of body weight) in place of drinking water, ad libitum. The group with obesity without peptide supplementation was designated DIO (diet-induced obesity), while the peptide-treated obese group was designated DIO + peptides (DIO + P).
Animal euthanasia and organ sample collection
The experimental animals were euthanized after 12 weeks from the start of the trial. To collect an acceptable volume of blood samples while avoiding the potential effect of anesthetics on biochemical parameters in blood and tissues, the decapitation method of euthanasia was used. Blood samples (about 10 mL per animal) were collected immediately following decapitation for serum preparation. To prepare liver, kidney, and VAT homogenates, 1 g of each organ was mixed with 50 mM Tris-HCl buffer (pH 7.4) (C4706, Sigma-Aldrich, Saint Louis, USA) containing 140 mM NaCl (A57006, Thermo Fisher Scientific, Waltham, USA) at a 1:9 (w/v) ratio. The tissues were homogenized at +4 °C with hand-held homogenizers (11799, Reichelt Chemietechnik GmbH + Co., Heidelberg, Germany). The resultant homogenates were centrifuged at 600 × g for 15 min, followed by a second centrifugation at 10,000 × g for 15 min. To prepare rat serum samples, freshly obtained blood was centrifuged at +4 °C and 1,300 × g for 20 min. The homogenates and serum samples were frozen at −20 °C.
Enzyme-linked immunosorbent assay (ELISA)
To determine the relative levels of inflammation-related cytokines, including hypoxia inducible factor 1 subunit alpha (HIF-1α), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), interleukin 6 (IL-6), interleukin 8 (IL-8), interleukin 1 beta (IL-1β), tumor necrosis factor alpha (TNF-α), and resistin, we used a standard ELISA procedure with minor modifications. Serum and homogenate samples (100 µL), diluted in Tris-buffered saline (TBS) buffer (1060.1, Carl Roth GmbH + Co. KG, Karlsruhe, Germany), were added to sterile microplate wells (2205, Thermo Scientific™ Waltham, USA) and left at +4°C overnight. Dilutions of the samples were as follows: serum 1:100, liver and kidneys 1:50, adipose tissue 1:20. Next, the wells were washed three times with Tris-buffered saline Tween (TBST) buffer (1061.1, Carl Roth GmbH + Co. KG, Karlsruhe, Germany), then blocked with a 3% solution of powdered milk (T145, Carl Roth GmbH + Co. KG, Karlsruhe, Germany) and incubated for 1 h at +37 °C. After washing we added 100 µL of diluted solutions (1:3,000 in TBST buffer) of the corresponding antibodies: HIF-1α (PA3-16521, Thermo Fisher Scientific, Waltham, USA), NF-κB (ER0815, Thermo Fisher Scientific, Waltham, USA), IL-6 (700480, Thermo Fisher Scientific, Waltham, USA), IL-8 (701232, Thermo Fisher Scientific, Waltham, USA), IL-1β (PA5-88078, Thermo Fisher Scientific, Waltham, USA), TNF-α (PA1-40281, Thermo Fisher Scientific, Waltham, USA), and resistin (BS-0795R, Bioss Inc., Woburn, USA), and incubated for 1 h at +37 °C. Following incubation, the wells were washed and incubated with horseradish peroxidase (HRP)-conjugated secondary anti-mouse (31430, Thermo Fisher Scientific, Waltham, USA) and anti-rabbit (31460, Thermo Fisher Scientific, Waltham, USA) antibody solutions in TBST buffer (1:7,000) for another hour at 37 °C. The substrate solution consisted of 0.4 mg/mL o-phenylenediamine dihydrochloride (P9029, Sigma-Aldrich, Saint Louis, USA) in 0.05 M citrate-phosphate buffer (P4809, Sigma-Aldrich, Saint Louis, USA) and 0.4 µL/mL of 30% H2O2 (CP26.1, Carl Roth GmbH + Co. KG, Karlsruhe, Germany). After incubation with secondary antibodies, the wells were washed, and 100 µL of substrate solution was added to each well. The wells were then incubated in a dark place for 5 min. The reaction was terminated by adding 100 µL of stop solution (3 M sulfuric acid (1.00713, Merck KGaA, Darmstadt, Germany)). Optical density was immediately measured at 492 nm using a microplate reader (MQX200, BioTek, USA). To compensate for possible background chemiluminescence, we used blank samples containing TBS buffer instead of the homogenate solution. To achieve higher precision, each sample was analyzed in triplicate.
Protein quantification and data analysis
To avoid discrepancies in the results, we took into account the protein concentration in the samples when calculating the relative cytokine content. The concentration of total protein was determined by the standard procedure [11] using commercially available Bradford Reagent (39222.03, SERVA Electrophoresis GmbH, Heidelberg, Germany). Calculation of cytokine content was performed according to the formula:
where λS495 is the optical density of sample at wavelength 495 nm; λB495 is the optical density of blank sample at wavelength 495 nm; KD1 is coefficient of homogenate dilution (KD1 = 100 for serum; KD1 = 50 for liver and kidneys; KD1=20 for VAT); KD2 is coefficient of tissue dilution (KD2 = 10 for all sample types); C is protein concentration in mg/mL, determined by Bradford assay.
Statistical analysis
Normality of distribution was assessed using the Kolmogorov-Smirnov test of normality. The significance of the differences between the groups was analyzed using one-way analysis of variance (ANOVA) and Tukey’s post hoc test at a significance level of P < 0.05. Data in the figures are given as mean ± standard error of mean (M ± m).
| Results | ▴Top |
The results of this study are consistent with the widely accepted fact that LGI is one of the integral markers of metabolic syndrome and obesity. Moreover, LGI is considered a major contributor to the main metabolic dysfunctions, such as insulin resistance, NAFLD, chronic hypertension, etc. [12, 13].
According to Figure 1, serum resistin, IL-6, and IL-1β levels increased significantly in the DIO group during obesity, by approximately 207%, 177%, and 171% of control levels, respectively. Meanwhile, in the DIO + P group, resistin concentration was only 126% of the control value, IL-6 was 133%, and IL-1β levels were similar to the control. NF-κB and HIF-1α levels in the DIO group increased by only 35% and 48%, respectively, while these same parameters in the DIO + P group were nearly the same as in the control group. Other cytokines, such as IL-8 and TNF-α did not show statistically significant differences.
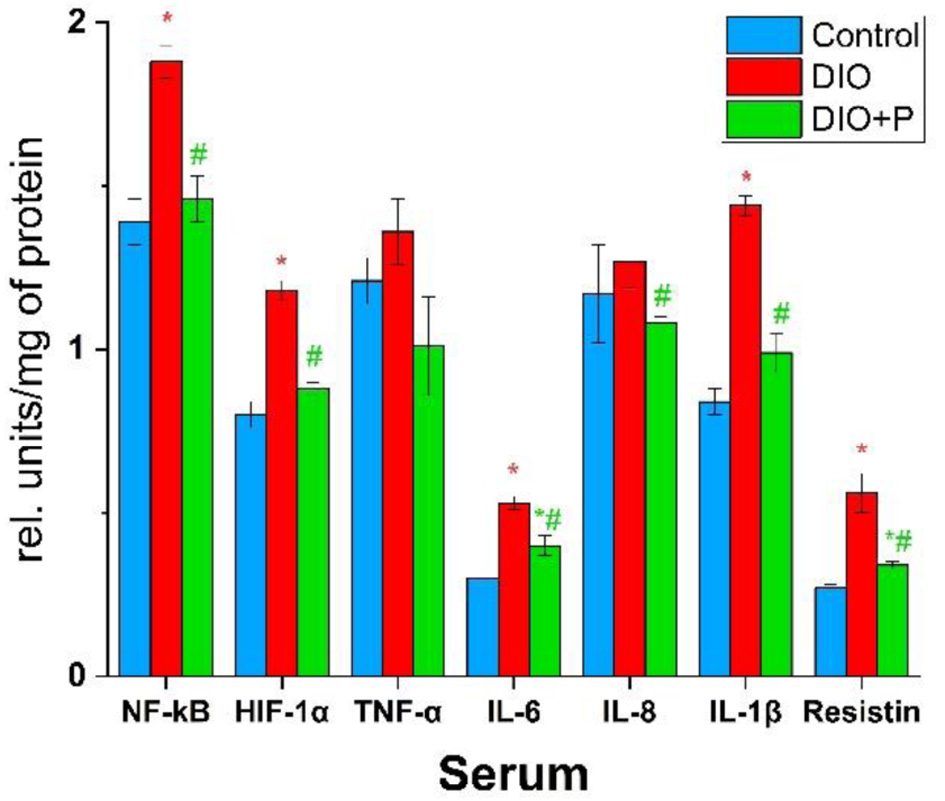 Click for large image | Figure 1. Concentrations of cytokines and pro-inflammatory transcription factors in rats’ sera. *P < 0.05 (compared to control group), #P < 0.05 (compared to DIO group), n = 5. DIO: diet-induced obesity. |
As shown in Figure 2, resistin levels in the VAT of the DIO group increased most markedly, reaching nearly 180% of the control value. In the DIO + P group, this parameter was comparable to the control. A less significant increase was observed for IL-6 and TNF-α, at 153% and 151% of the control levels, respectively. Peptide consumption resulted in IL-6 and TNF-α levels decreasing to 126% and 125% of the control values. Levels of IL-1β, HIF-1α, and IL-8 in the DIO group were approximately 145%, 142%, and 133% of the control levels, respectively. Peptide intake caused a relative decrease in IL-1β to 128%, HIF-1α to 117%, and IL-8 to 114% of the control levels in the DIO + P group. NF-κB concentration in the DIO group increased the least, by only 116% of the control, while in the DIO + P group, this parameter remained at the control level.
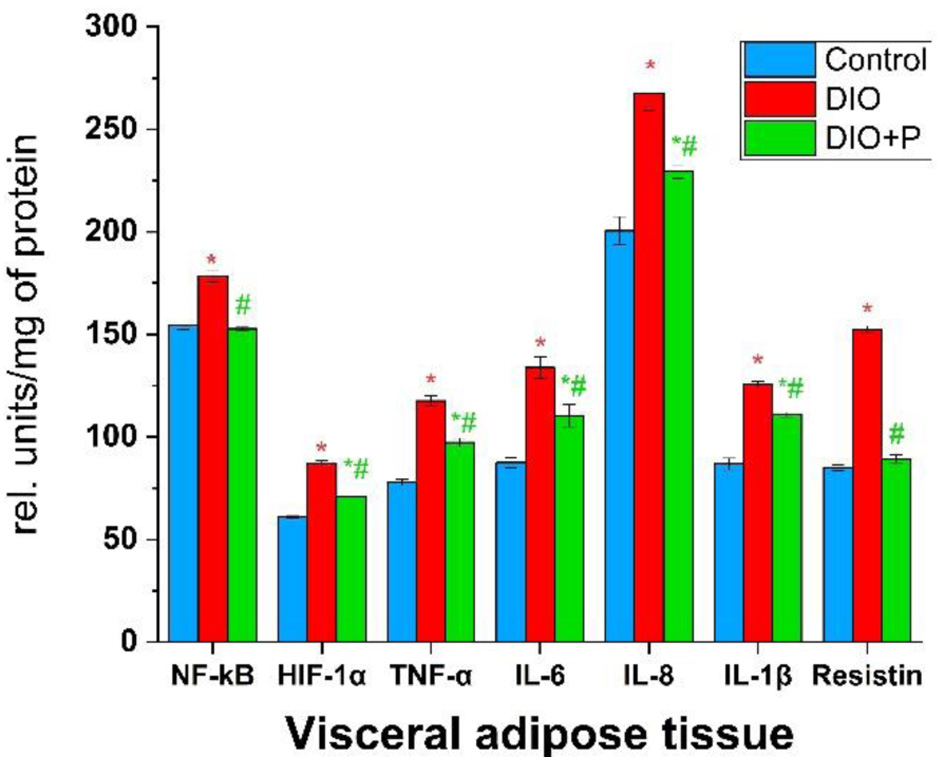 Click for large image | Figure 2. Concentrations of cytokines and pro-inflammatory transcription factors in rats’ visceral adipose tissue. *P < 0.05 (compared to control group), #P < 0.05 (compared to DIO group), n = 5. DIO: diet-induced obesity. |
In our experiment, liver levels of HIF-1α and IL-8 in the DIO group were the highest among the assessed parameters, reaching 144% and 142% of the control values, respectively (Fig. 3). The same parameters in the DIO + P group were comparable to control levels.
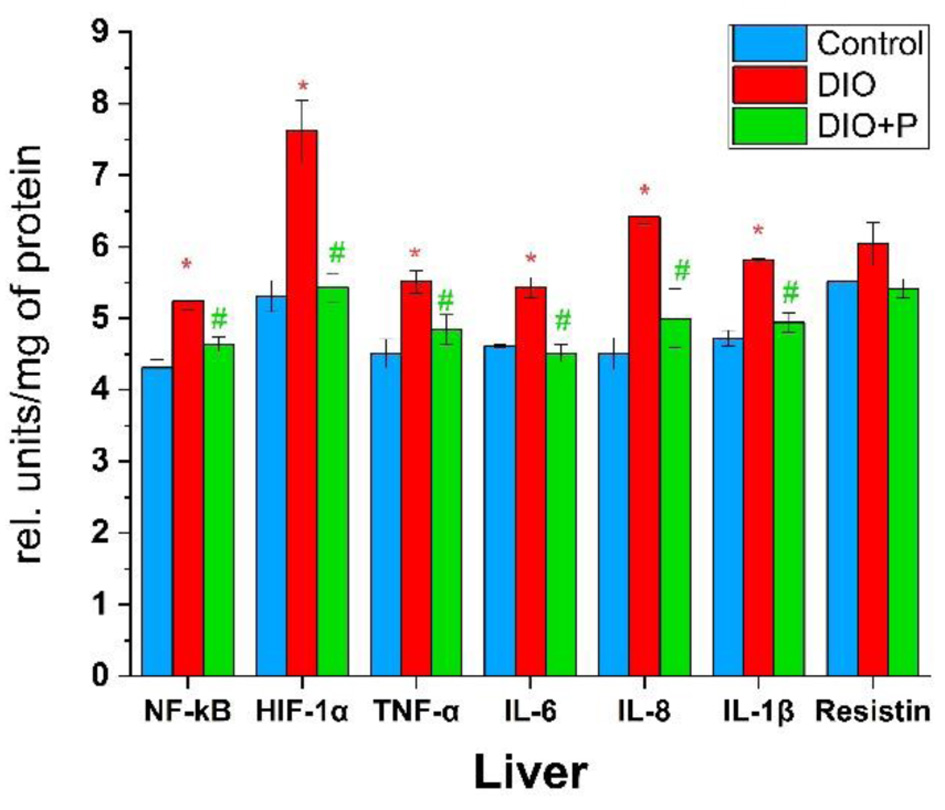 Click for large image | Figure 3. Concentrations of cytokines and pro-inflammatory transcription factors in rats’ liver. *P < 0.05 (compared to control group), #P < 0.05 (compared to DIO group), n = 5. DIO: diet-induced obesity. |
A less pronounced effect of obesity was observed for IL-1β (123% of the control), TNF-α and NF-κB (both 122% of the control), and IL-6 (118% of the control). Levels of all these molecules in the DIO + P group aligned with control values. However, resistin concentration did not show any statistically significant changes in our experiment.
The smallest changes in cytokine profile were observed in kidney samples, yet the pattern of changes is similar to that observed in liver samples. Compared with the control group, the most significant deviation in DIO group was observed for IL-8 (138% of control values) and HIF-1α (135% of control values), yet in the DIO + P group, these values were in line with the control levels (Fig. 4).
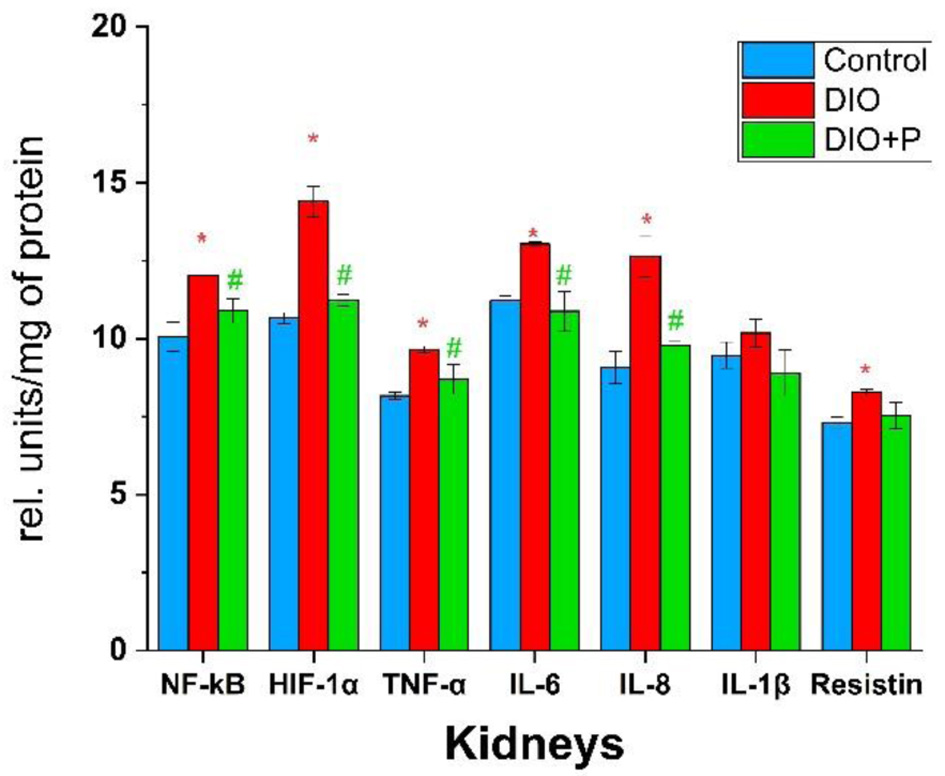 Click for large image | Figure 4. Concentrations of cytokines and pro-inflammatory transcription factors in rats’ kidneys. *P < 0.05 (compared to control group), #P < 0.05 (compared to DIO group), n = 5. DIO: diet-induced obesity. |
Moreover, according to our results, NF-κB concentration in the DIO group was elevated by 20%, TNF-α by 18%, IL-6 by 16%, and resistin by 13% (Fig. 4). In the DIO + P group, these parameters were reduced to the control values. However, the results did not show any statistically significant changes in IL-1β concentration.
| Discussion | ▴Top |
In our experimental conditions, obesity led to an increase in the main pro-inflammatory transcription factors and cytokines in most metabolically active organs, suggesting the development of chronic LGI. We also demonstrated a significant effect of P. vulgaris husk-derived peptides on the cytokine profile in these organs, highlighting their potential therapeutic applications.
Analyzing Figures 1 and 2, we noticed some common cytokine profile features between serum and VAT. In both sample types, major changes were related to resistin, IL-6, and IL-1β, which suggests that VAT may be a main contributor to systemic inflammation in obesity. Expansion of VAT is believed to be the primary origin of LGI. Despite not much attention being paid to this tissue, it is one of the crucial organs in our bodies. It is not only an energy-storing depot, but also an endocrine organ producing adipokines, such as leptin, resistin, adiponectin, adipolin, and pro-inflammatory cytokines, such as TNF-α, IL-8, and IL-6. Expansion of adipose tissue and adipocyte hypertrophy lead to enhanced production of these molecules and their release into the bloodstream [14]. Furthermore, expansion of VAT creates physical pressure inside the abdomen, increasing hypoxia and, therefore, levels of HIF-1α [15]. This process can be compared to tumor formation, because in both conditions, LGI progresses in a similar manner. Chronic hypoxia induces the activity of the mitogen-activated protein kinase (MAPK) kinase pathway, which leads to activation of HIF-1α and NF-κB, transcription factors of hypoxia-induced genes and multiple pro-inflammatory genes [16]. The release of pro-inflammatory cytokines, especially IL-8, which acts as a chemoattractant for immune cells, causes the migration of M1-type macrophages to the primary inflammation site, specifically adipose tissue. They, in turn, release pro-inflammatory molecules themselves, such as IL-6, IL-8, IL-1β, and TNF-α [17, 18].
Results depicted in Figure 1 are consistent with the results obtained by Monk et al, whose study showed a decline in serum IL-17A, IFN-γ, TNF-α, IL-1β, and IL-6 levels of C57BL/6 mice with colonic inflammation [19]. In obese patients, adipose tissue, apart from cytokines, produces high amounts of adipokine resistin, which is particularly notable because, according to some studies, it is an antagonist of insulin [20]. Moreover, resistin may have a direct impact on inflammation progression through activation of Toll-like receptor 4 or adenylyl cyclase-associated protein 1 [21]. The concentration of this adipokine in blood correlates with body mass index in obese rats. Despite the resistin-mediated link between obesity and diabetes not being fully understood, in recent studies, it caused glucose uptake inhibition in L6 muscle cells and 3T3-L1a adipocytes [22]. IL-6 is considered the main inflammatory mediator in obesity. This cytokine can induce lipolysis, adipokine synthesis, oxidation of lipoproteins, and, most importantly, facilitate insulin resistance [23]. IL-6 activates the Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway, which in turn inhibits insulin transduction signals through insulin receptor substrate 1 (IRS-1) and phosphatidylinositol 3-kinase (PI3K) [24]. IL-1β is another pro-inflammatory cytokine, which not only promotes the spread of inflammation but also provokes insulin resistance [25]. TNF-α, levels of which were increased in the VAT of obese rats, is also worth mentioning. This cytokine is also a well-established marker of inflammation involved in LGI development. Previous studies showed a positive correlation between body weight and TNF-α mRNA levels in obese patients [26]. TNF-α and IL-6 can stimulate impaired phosphorylation of IRS-1, therefore inhibiting the signal transduction in the insulin signaling pathway [27]. Given the above, it is highly likely that LGI-mediated insulin resistance developed in our DIO group.
Liver, as one of the most metabolically active organs, probably suffers from the consequences of obesity the most. Obesity serves as the primary driver for a multisystem inflammatory cascade, where dysfunctional adipose tissue triggers a state of chronic LGI that bridges NAFLD and systemic atherosclerosis. NAFLD is not merely a localized liver condition but a potent independent risk factor for cardiovascular disease. This relationship is often quantified via carotid intima-media thickness (cIMT), which serves as a validated surrogate marker for subclinical atherosclerosis. In obese populations, the “crosstalk” between an inflamed liver and the vascular wall—mediated by pro-inflammatory cytokines and altered lipid metabolism—accelerates the thickening of the carotid arterial walls [28]. Metabolic-dysfunction associated steatotic liver disease (MASLD), modern naming of NAFLD, is a common obesity-related disease, which is defined as a state where fat comprises more than 5% of hepatic mass [29, 30]. According to statistics, it is the most widespread liver disease, being diagnosed in every third patient [30]. Importantly, LGI and altered immune response are considered as central factors in MASLD development [31]. Oxidative stress is another trigger to MASLD progression. An excessive amount of free fatty acids in the liver leads to the generation of reactive oxygen species (ROS), which in turn may lead to mitochondria impairment and sudden apoptosis of hepatocytes [27]. In this study we have shown that levels of HIF-1α increase the most during obesity, supporting the findings of previous studies. HIF-1α, as a pro-inflammatory transcription factor, is a mediator between hypoxia, LGI, and MASLD. Mesarwi et al discovered that hepatocyte-specific HIF-1α-knockout mice with induced obesity had lower weight gain rates, improved glucose levels, and better metabolic profiles [32]. Moreover, HIF-1α was also found to be associated with hepatic fibrosis [33]. IL-8 is known as a chemoattractive molecule, which promotes neutrophil migration to inflamed tissues [34]. In a recent study by Auguet et al, IL-8 was the only cytokine whose levels were increased in women with morbid obesity. Authors also state that IL-8, being highly associated with steatosis degree and lobular inflammation, can be used as a marker for nonalcoholic steatohepatitis (NASH), which is a severe case of MASLD [35]. Elevated levels of IL-1β, TNF-α, and IL-6, which were moderately increased in our obese rats are also direct evidence of MASLD, since their presence is indicative of the pathogenesis of fatty liver diseases [36].
Chronic kidney disease (CKD) is one of the most common renal diseases in obesity, with approximately every fifth case of obesity resulting in CKD [37]. Diabetic nephropathy (DN) is another obesity-related pathology, characterized by renal hypoxia, increased oxidative stress, and elevated levels of HIF-1α [38]. As it was mentioned above, cytokine pattern changes in kidneys were similar to those observed in liver samples with HIF-1α and IL-8 being elevated the most. In a study by Tuncay et al, IL-8 was the only cytokine whose levels were elevated in the early stage of CKD in children [39]. Taking into account the above-mentioned, we suppose that CKD could also have been developed in our experimental conditions.
Legumes are already well-known as a source rich in various bioactive peptides and proteins with antioxidant, angiotensin-converting enzyme (ACE)-inhibitory, and anti-inflammatory activities [40]. P. vulgaris beans are very promising in this context due to the presence of peptides with anti-diabetic, antihypertensive, anti-cancer, and anti-inflammatory activities. Indrati reported that more than 20 peptides, obtained by Alcalase, pepsin, and pancreatin treatment of P. vulgaris beans, exerted strong anti-inflammatory properties. Notably, most of them consist merely of 3–5 amino acid residues [41]. According to a study by Monk et al, even cooked common beans can improve gut microbiota, production of short-chain fatty acids, and adipose tissue inflammation [42]. In another study, boiled kidney beans inhibited the expression of TNF-α and IL-6 in the murine peritoneal macrophage cell line through the lectin-mediated mechanism [43]. P. vulgaris husk extracts were also found effective against inflammation processes in a murine second-degree alkali burn esophagus model [44].
The cytokine profile shifts we observed in the DIO group clearly indicate LGI inflammation in obese animals on a broader level. From the perspective of adipose tissue, obesity leads to further intensification of inflammation through a positive feedback mechanism and the development of insulin resistance. From the liver’s perspective, obesity causes local hypoxia, MASLD, and possibly hepatic fibrosis. The kidneys are also affected, with a risk of CKD development. Consumption of P. vulgaris husk peptides led to a significant decrease in pro-inflammatory cytokine levels or even their mitigation to the control levels in most cases, implying a positive effect on the rats’ immune system. Therefore, peptides from kidney bean husk have shown results comparable to those obtained from bean extracts or protein isolates. From an economic perspective, bean husks are a more affordable type of raw material, being practically a byproduct of the bean harvesting industry.
One of the potential mechanisms underlying the anti-inflammatory properties of P. vulgaris peptides may be related to their antibacterial effects. Obesity causes a microbiome shift to the prevalence of Gram-negative bacteria, which in turn causes loss of gut barrier integrity and release of lipopolysaccharide (LPS) into the bloodstream. LPS, as a powerful immunogen, can be one of the LGI contributors [45]. Thus, bioactive peptides from kidney bean and its husks can potentially act on Gram-negative bacteria, shifting the microbiome balance to Gram-positive microorganisms, reducing LPS release into the bloodstream, and as a result, reducing LGI. Another potential mechanism is the inhibition of the NF-κB pathway by decreasing the p65 subunit translocation into the nucleus [46]. In another study, P. vulgaris extract mitigated levels of cyclooxygenase-2 and inducible nitric oxide synthase in the liver of obese mice mediated by peroxisome proliferator-activated receptor (PPAR)-α and γ [47]. The exact mechanism behind the anti-inflammatory effect of kidney bean husk peptides is unknown, yet it may be related to the mechanisms described above, or it may be a combination of them. Further studies are required to elucidate these pathways in detail.
Conclusions
To conclude, we have, for the first time, investigated cytokine profile changes in metabolically active organs of rats during obesity, both with and without P. vulgaris husk-derived peptide diet-supplementation, emphasizing the potential of these peptides in combating the negative effects of obesity. The results revealed an elevation of pro-inflammatory cytokines and transcription factors in all assessed tissues of obese rats, indicating the development of low-grade inflammation. In serum and adipose tissue samples, the most pronounced changes were observed in resistin, IL-6, and IL-1β concentrations, suggesting the possible development of insulin resistance mediated by these cytokines. In liver and kidney samples, we observed elevation of HIF-1α and IL-8 levels, pointing to increased hypoxia and the development of MASLD and CKD in these organs, respectively. In the animal group that consumed peptides, the levels of most of the assessed cytokines were significantly lower, in some cases approaching the control values. These results demonstrate the effectiveness of husk-derived peptides, which are cheaper and easier to obtain, compared to methods involving P. vulgaris bean extracts.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Not applicable.
Author Contributions
Serhii Shchypanskyi: experimental procedures and manuscript preparation; Daryna Krenytska: experimental procedures and data analysis; Mariana Kuznietsova: concept and experimental design; Nataliia Raksha: manuscript editing and review; Olexii Savchuk: concept and experimental design, supervision, and final approval of manuscript.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Islam A, Sultana H, Nazmul Hassan Refat M, Farhana Z, Abdulbasah Kamil A, Meshbahur Rahman M. The global burden of overweight-obesity and its association with economic status, benefiting from STEPs survey of WHO member states: a meta-analysis. Prev Med Rep. 2024;46:102882.
doi pubmed - Vajravelu ME, Tas E, Arslanian S. Pediatric obesity: complications and current day management. Life (Basel). 2023;13(7):1591.
doi pubmed - Bluher M. An overview of obesity-related complications: The epidemiological evidence linking body weight and other markers of obesity to adverse health outcomes. Diabetes Obes Metab. 2025;27(Suppl 2):3-19.
doi pubmed - Suryaningtyas IT, Je J-Y. Bioactive peptides from food proteins as potential anti-obesity agents: Mechanisms of action and future perspectives. Trends Food Sci Technol. 2023;138:141-152.
doi - Bhide YS, Nehete JY, Bhambar RS. Botanical, chemical and pharmacological review of phaseolus vulgaris L. (common bean). Int J Health Sci (Qassim). 2022;6(S5):11527-11543.
doi - Garcia-Cordero JM, Martinez-Palma NY, Madrigal-Bujaidar E, Jimenez-Martinez C, Madrigal-Santillan E, Morales-Gonzalez JA, Paniagua-Perez R, et al. Phaseolin, a protein from the seed of phaseolus vulgaris, has antioxidant, antigenotoxic, and chemopreventive properties. Nutrients. 2021;13(6):1750.
doi pubmed - Hwang SJ, Song YS, Lee HJ. Phaseolin attenuates lipopolysaccharide-induced inflammation in RAW 264.7 cells and zebrafish. Biomedicines. 2021;9(4):420.
doi pubmed - Kyznetsova M, Makieieva O, Lavrovska D, Tymoshenko M, Sheverova D, Halenova T, et al. Effect of aqueous extract from phaseolus vulgaris pods on lipid peroxidation and antioxidant enzymes activity in the liver and kidney of diabetic rats. J Appl Pharm Sci. 2015;5(05):1-6.
doi - Shchypanskyi S, Raksha N, Vovk T, Halenova T, Savchuk O. Antioxidant properties of common bean (<i>Phaseolus vulgaris</i>) husk-derived peptides. Journal of Applied and Natural Science. 2025;17(1):200-204.
doi - Yurchenko A, Krenytska D, Kalmukova O, Raksha N, Halenova T, Vovk T, et al. The kidney beans (Phaseolus vulgaris) pods extract affects the central and peripheral serotonergic systems in rats with high-calorie diet-induced obesity. J Endocrinol Metab. 2021;11(5):123-133.
doi - Reinmuth-Selzle K, Tchipilov T, Backes AT, Tscheuschner G, Tang K, Ziegler K, Lucas K, et al. Determination of the protein content of complex samples by aromatic amino acid analysis, liquid chromatography-UV absorbance, and colorimetry. Anal Bioanal Chem. 2022;414(15):4457-4470.
doi pubmed - Parra-Peralbo E, Talamillo A, Barrio R. Origin and development of the adipose tissue, a key organ in physiology and disease. Front Cell Dev Biol. 2021;9:786129.
doi pubmed - van de Vyver M. Immunology of chronic low-grade inflammation: relationship with metabolic function. J Endocrinol. 2023;257(1):e220271.
doi pubmed - Korbecki J, Siminska D, Gassowska-Dobrowolska M, Listos J, Gutowska I, Chlubek D, Baranowska-Bosiacka I. Chronic and cycling hypoxia: drivers of cancer chronic inflammation through HIF-1 and NF-kappaB activation: a review of the molecular mechanisms. Int J Mol Sci. 2021;22(19):10701.
doi pubmed - Varra FN, Varras M, Varra VK, Theodosis-Nobelos P. Molecular and pathophysiological relationship between obesity and chronic inflammation in the manifestation of metabolic dysfunctions and their inflammation-mediating treatment options (Review). Mol Med Rep. 2024;29(6):95.
doi pubmed - Kim CS, Park HS, Kawada T, Kim JH, Lim D, Hubbard NE, Kwon BS, et al. Circulating levels of MCP-1 and IL-8 are elevated in human obese subjects and associated with obesity-related parameters. Int J Obes (Lond). 2006;30(9):1347-1355.
doi pubmed - Castro AM, Macedo-de la Concha LE, Pantoja-Melendez CA. Low-grade inflammation and its relation to obesity and chronic degenerative diseases. Revista Medica Del Hospital General de Mexico. 2017;80(2):101-105.
doi - Cavaliere G, Cimmino F, Trinchese G, Catapano A, Petrella L, D'Angelo M, Lucchin L, et al. From obesity-induced low-grade inflammation to lipotoxicity and mitochondrial dysfunction: altered multi-crosstalk between adipose tissue and metabolically active organs. Antioxidants (Basel). 2023;12(6):1172.
doi pubmed - Monk JM, Zhang CP, Wu W, Zarepoor L, Lu JT, Liu R, Pauls KP, et al. White and dark kidney beans reduce colonic mucosal damage and inflammation in response to dextran sodium sulfate. J Nutr Biochem. 2015;26(7):752-760.
doi pubmed - Khanna D, Khanna S, Khanna P, Kahar P, Patel BM. Obesity: a chronic low-grade inflammation and its markers. Cureus. 2022;14(2):e22711.
doi pubmed - Vique-Sanchez JL. Resistin, a multipotential therapeutic target. Biointerface Res Appl Chem. 2022;13:384.
doi - Banya RJ, Dallatu MK, Nnamah NK, Tambuwal FM. Serum resistin and glucose among obese and non-obese diabetic rats. Sokoto J Med Lab Sci. 2025;9(4):202-214. https://doi.org/10.4314/sokjmls.v9i4.22
- Eder K, Baffy N, Falus A, Fulop AK. The major inflammatory mediator interleukin-6 and obesity. Inflamm Res. 2009;58(11):727-736.
doi pubmed - Abdullah MY, AlQwaidi SD, Alshehri AM, Alamry FA, Alamri RS, Alsolbi ZK, et al. Obesity-induced inflammation and its role in the development of insulin resistance. Journal of Healthcare Sciences. 2024;4(10):448-454.
doi - Gao D, Madi M, Ding C, Fok M, Steele T, Ford C, Hunter L, et al. Interleukin-1beta mediates macrophage-induced impairment of insulin signaling in human primary adipocytes. Am J Physiol Endocrinol Metab. 2014;307(3):E289-304.
doi pubmed - Wurfel M, Bluher M, Stumvoll M, Ebert T, Kovacs P, Tonjes A, Breitfeld J. Adipokines as clinically relevant therapeutic targets in obesity. Biomedicines. 2023;11(5):1427.
doi pubmed - Accacha S, Barillas-Cerritos J, Srivastava A, Ross F, Drewes W, Gulkarov S, De Leon J, et al. From childhood obesity to metabolic dysfunction-associated steatotic liver disease (MASLD) and hyperlipidemia through oxidative stress during childhood. Metabolites. 2025;15(5):287.
doi pubmed - Tarantino G, Costantini S, Finelli C, Capone F, Guerriero E, La Sala N, Gioia S, et al. Carotid intima-media thickness is predicted by combined eotaxin levels and severity of hepatic steatosis at ultrasonography in obese patients with Nonalcoholic Fatty Liver Disease. PLoS One. 2014;9(9):e105610.
doi pubmed - Dobbie LJ, Burgess J, Hamid A, Nevitt SJ, Hydes TJ, Alam U, Cuthbertson DJ. Effect of a low-calorie dietary intervention on liver health and body weight in adults with metabolic-dysfunction associated steatotic liver disease (MASLD) and overweight/obesity: a systematic review and meta-analysis. Nutrients. 2024;16(7):1030.
doi pubmed - Beyoglu D, Popov YV, Idle JR. Metabolomic hallmarks of obesity and metabolic dysfunction-associated steatotic liver disease. Int J Mol Sci. 2024;25(23):12809.
doi pubmed - Zhou Z, Li L, Wang C, Li S, Chen P, Huang J, Peng M. The association between sedentary behavior and MASLD in overweight and obese adults: investigating the role of inflammatory markers using NHANES data (2017-March 2020). Front Nutr. 2025;12:1579453.
doi pubmed - Mesarwi OA, Moya EA, Zhen X, Gautane M, Zhao H, Wegbrans Giro P, Alshebli M, et al. Hepatocyte HIF-1 and intermittent hypoxia independently impact liver fibrosis in murine nonalcoholic fatty liver disease. Am J Respir Cell Mol Biol. 2021;65(4):390-402.
doi pubmed - Chu Q, Gu X, Zheng Q, Zhu H. Regulatory mechanism of HIF-1alpha and its role in liver diseases: a narrative review. Ann Transl Med. 2022;10(2):109.
doi pubmed - Asiri A, Hazeldine J, Moiemen N, Harrison P. IL-8 induces neutrophil extracellular trap formation in severe thermal injury. Int J Mol Sci. 2024;25(13):7216.
doi pubmed - Auguet T, Bertran L, Binetti J, Aguilar C, Martinez S, Sabench F, Lopez-Dupla JM, et al. Relationship between IL-8 circulating levels and TLR2 hepatic expression in women with morbid obesity and nonalcoholic steatohepatitis. Int J Mol Sci. 2020;21(11):4189.
doi pubmed - Meyer M, Schwarzler J, Jukic A, Tilg H. Innate Immunity and MASLD. Biomolecules. 2024;14(4):476.
doi pubmed - Arabi T, Shafqat A, Sabbah BN, Ashraf N, Shah H, Abdulkader H, Razak A, et al. Obesity-related kidney disease: Beyond hypertension and insulin-resistance. Front Endocrinol (Lausanne). 2022;13:1095211.
doi pubmed - Stanigut AM, Pana C, Enciu M, Deacu M, Cimpineanu B, Tuta LA. Hypoxia-inducible factors and diabetic kidney disease - How deep can we go? Int J Mol Sci. 2022;23(18):10413.
doi pubmed - Tuncay SC, Dogan E, Hakverdi G, Tutar ZU, Mir S. Interleukin-8 is increased in chronic kidney disease in children, but not related to cardiovascular disease. J Bras Nefrol. 2021;43(3):359-364.
doi pubmed - Tawalbeh D, Al-U'datt M H, Wan Ahmad WAN, Ahmad F, Sarbon NM. Recent advances in in vitro and in vivo studies of antioxidant, ACE-inhibitory and anti-inflammatory peptides from legume protein hydrolysates. Molecules. 2023;28(6):2423.
doi pubmed - Indrati R. Bioactive peptides from legumes and their bioavailability. In: Jimenez-Lopez JC, Clemente (eds). Legumes Research - Volume 2. IntechOpen; 2022.
doi - Monk JM, Wu W, Lepp D, Pauls KP, Robinson LE, Power KA. Navy bean supplementation in established high-fat diet-induced obesity attenuates the severity of the obese inflammatory phenotype. Nutrients. 2021;13(3):757.
doi pubmed - Une S, Nakata R, Nonaka K, Akiyama J. Antiproliferative and anti-inflammatory effects of fractionated crude lectins from boiled kidney beans (Phaseolus vulgaris). J Food Sci. 2024;89(1):671-683.
doi pubmed - Raetska Y, Dmytryk V, Chornenka N, Halenova T, Raksha N, Vovk T, et al. The effect of Phaseolus vulgaris pods extract on cytokines profile in the condition of alkali burn esophagus 2 degree. Biomedical Research and Therapy. 2019;6(9):3352-3358.
doi - Cheng Z, Zhang L, Yang L, Chu H. The critical role of gut microbiota in obesity. Front Endocrinol (Lausanne). 2022;13:1025706.
doi pubmed - Oseguera-Toledo ME, de Mejia EG, Dia VP, Amaya-Llano SL. Common bean (Phaseolus vulgaris L.) hydrolysates inhibit inflammation in LPS-induced macrophages through suppression of NF-kappaB pathways. Food Chem. 2011;127(3):1175-1185.
doi pubmed - Avagliano C, De Caro C, Cuozzo M, Liguori FM, La Rana G, Micheli L, Di Cesare Mannelli L, et al. Phaseolus vulgaris extract ameliorates high-fat diet-induced colonic barrier dysfunction and inflammation in mice by regulating peroxisome proliferator-activated receptor expression and butyrate levels. Front Pharmacol. 2022;13:930832.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.