| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 000, Number 000, May 2026, pages 000-000
The Influence of Age on the Efficacy of a Novel Antidiabetic Drug Imeglimin in Patients With Poorly Controlled Type 2 Diabetes
Masataka Kusunokia, h, Fumiya Hisanob, Shin-ichi Matsudac, Kohei Kunitsud, Daijiro Miyanagae, Masaaki Kusunokif, Kazuhiko Tsutsumia, Tetsuro Miyatag
aApollon Nagoya Exercise Therapy Center for Diabetes, Naka-ku, Nagoya City, Aichi, Japan
bDepartment of Integrated Health Sciences, Graduate School of Medicine, Nagoya University, Higashi-ku, Nagoya City, Aichi, Japan
cDepartment of Data Science, Faculty of Science and Technology, Nanzan University, Showa-ku, Nagoya City, Aichi 466-8673, Japan
dAkishima Clinic, Naka-ku, Nagoya City, Aichi, Japan
eNagoya City University, Nagoya City, Aichi, Japan
fAichi Medical University, Nagoya City, Aichi, Japan
gOffice of Medical Education, School of Medicine, International University of Health and Welfare, Narita City, Chiba, Japan
hCorresponding Author: Masataka Kusunoki, Apollon Nagoya Exercise Therapy Center for Diabetes, Naka-ku, Nagoya City, Aichi, Japan
Manuscript submitted February 19, 2026, accepted March 28, 2026, published online May 11, 2026
Short title: Imeglimin and Aging
doi: https://doi.org/10.14740/jem1637
| Abstract | ▴Top |
Background: Imeglimin is a novel drug shown to improve mitochondrial function in patients with diabetes. This study was undertaken to evaluate the efficacy and safety of imeglimin in patients with poorly controlled type 2 diabetes. The effect of aging on the efficacy and safety of this drug was also evaluated.
Methods: Imeglimin (2,000 mg/kg) was administered for 3 months to 52 patients. Hemoglobin A1c (HbA1c) and other blood biochemical parameters were measured before and after the 3-month treatment period. Regression analysis was performed to determine the influence of age on the effects of the drug on these parameters.
Results: Significant reduction of HbA1c and serum lipids were noted after the treatment. Regression analysis to determine the influence of age on relevant biochemical parameters in patients receiving imeglimin revealed that the biochemical effects of this drug on the serum levels of the hepatic function parameters (aspartate aminotransferase (AST), alanine aminotransferase (ALT), and γ-glutamyl transferase (γ-GTP)) and the serum uric acid level were influenced by interactions between the patient’s age and the pre-treatment baseline levels of the parameters. Older patients with higher baseline levels of these parameters demonstrated greater reduction in hepatic biomarkers following treatment with imeglimin.
Conclusions: This study confirmed imeglimin as a useful drug for individuals with type 2 diabetes who exhibit poor glycemia control. The effects of imeglimin in reducing the serum levels of hepatic function parameters were reinforced by aging, indicating that imeglimin is a particularly useful drug for patients with type 2 diabetes who are elderly and/or have hepatopathy.
Keywords: Imeglimin; Type 2 diabetes; Antidiabetic drug; Japan; Aging; HbA1c; Glycemic control; Hepatic function
| Introduction | ▴Top |
Imeglimin is a novel antidiabetic drug that has been shown to improve mitochondrial functions [1]. To date, this drug is approved for therapeutic use only in Japan. Imeglimin exerts two important effects through improving mitochondrial functions by promoting insulin secretion [1, 2] and improving hepatic and skeletal muscle glucose production and utilization [3, 4].
In the treatment of patients with diabetes, drug therapy is typically added when exercise and dietary therapy alone prove insufficient. This study investigated the efficacy and safety of administering imeglimin in patients who were unable to adhere adequately to exercise and dietary therapy and therefore had poor glycemic control.
Previous studies have demonstrated the efficacy of imeglimin in improving the glucose metabolism in patients with type 2 diabetes. Prior to the present study, however, there have been no reports of studies that investigated the influence of the patient’s age on the efficacy and safety of imeglimin. Therefore, the present study was designed to analyze the differences in the efficacy of imeglimin depending on the patients’ age.
| Materials and Methods | ▴Top |
Subjects
This study was conducted in compliance with the ethical standards of the responsible institution for studies in human subjects as well as with Declaration of Helsinki.
The study was conducted in Japanese patients with type 2 diabetes. All the participants provided consent for participation in the study after receiving an explanation about the methods and objectives of the study from their attending physicians. The study protocol was approved in advance by the Ethics Committee of the Medical Corporation Odakai (Institutional Review Board (IRB) Approval No. 2021-1-9). We officially registered the study with the UMIN as an open label study (ID: UMIN000053539).
The subjects consisted of 52 outpatients with type 2 diabetes, including 35 men and 17 women, in whom therapeutic lifestyle modifications failed to yield adequate glycemia control.
Dosing method
Each patient received oral imeglimin (Sumitomo Pharma Co., Ltd., Tokyo, Japan) at 1,000 mg/dose twice daily for 3 months.
Measurement of the blood hemoglobin A1c (HbA1c) level, blood lipid profile, hepatic and renal function parameters, and other blood biochemical parameters
Blood samples from the study subjects were collected before the start and at the end of the 3-month treatment period with imeglimin (each at the appropriate time of the day). Serum was separated from the samples and stored frozen at –80 °C until the measurements. The serum levels of lipids, aspartate aminotransferase (AST), alanine aminotransferase (ALT), γ-glutamyl transferase (γ-GTP), creatinine (Cre), blood urea nitrogen (BUN), amylase, and uric acid were measured at the Handa Medical Association Health Center (Aichi, Japan) in an Auto Analyzer (JCA-BM8000 series, JAOL, Tokyo, Japan). HbA1c was measured at the same center by an automated high performance liquid chromatography (HPLC) method (HLC-723GX, Tosoh Corporation, Tokyo, Japan).
Statistical analysis
Data are expressed as means ± standard deviation (SD). A paired t-test was used for testing the significance of differences in the pre- and post-treatment levels of the blood biochemical parameters. The significance level was set at 0.05 (P < 0.05). A regression analysis was performed, involving interactions, with the data measured at the end of the 3-month imeglimin treatment course as the dependent variables, and the pre-treatment levels and patients’ age as the independent variables.
The regression model is as follows: y = a + bx + cz + dxz, where x is the pre-treatment level, y is the level at the end of 3-month imeglimin treatment, and z is the patient’s age.
An optimal model was determined using independent variables based on the Akaike information criterion [5]. If the original data were expected to improve normality, the data were also analyzed after logarithmic transformation.
We checked if the conclusion from such an analysis would differ from that derived from analysis of the original data without logarithmic transformation.
| Results | ▴Top |
Effects of imeglimin treatment on the body weight, body mass index (BMI), and blood levels of HbA1c, lipids, hepatic function parameters, renal function parameters, uric acid, and amylase
The age distribution and duration of illness are shown in Table 1. We did not investigate whether the duration of illness affects the efficacy of Imeglimin. The mean (with SD) values of each parameter before and after imeglimin treatment and 95% confidence intervals (CIs) of the difference are shown in Table 2.
 Click to view | Table 1. Patient Characteristics at Baseline |
 Click to view | Table 2. Responses to Imeglimin in Japanese Patients With Type 2 Diabetes Mellitus (N = 52 (Male: 35; Female: 17)) |
Body weight and BMI
Significant decreases of the body weight and BMI were observed after imeglimin treatment for 3 months as compared with the pre-treatment levels.
HbA1c
The serum HbA1c decreased significantly following imeglimin treatment as compared with the pre-treatment level.
Lipid profile
The serum levels of total cholesterol (TC), low-density lipoprotein-cholesterol (LDL-C) and non-high-density lipoprotein-cholesterol (non-HDL-C) decreased significantly after 3-month imeglimin treatment as compared with the pre-treatment levels. The serum levels of HDL-cholesterol (HDL-C) and triglycerides remained unaffected by imeglimin treatment. The atherosclerosis index (AI index, non-HDL-C/HDL-C) and the L/H ratio (LDL-C/HDL-C) decreased significantly following imeglimin treatment.
Hepatic function parameters
The serum levels of AST and ALT decreased significantly, while the serum γ-GTP remained unchanged, as compared with the pre-treatment levels, following imeglimin treatment.
Renal dysfunction parameters
No changes in the Cre, estimated glomerular filtration rate (eGFR) or BUN were observed following imeglimin treatment.
Uric acid and amylase
Imeglimin treatment had no effect on the serum levels of uric acid or amylase.
Effects of the patient’s age on the efficacy of imeglimin
Regression analysis was carried out to analyze the influence of the patient’s age on the baseline levels (before the start of imeglimin treatment) and on levels after the 3-month imeglimin treatment period. This analysis revealed that the effect of imeglimin in reducing the serum levels of the hepatic function parameters AST, ALT, and γ-GTP, and the serum uric acid level was influenced by interactions between the patient’s age and baseline levels of these parameters. The higher the patient’s age, the greater was the efficacy of imeglimin in reducing the levels of these parameters (Fig. 1). In Figure 1, we make lines for age 30, 45, 60, and 75, which are obtained by substituting each age into the regression models.
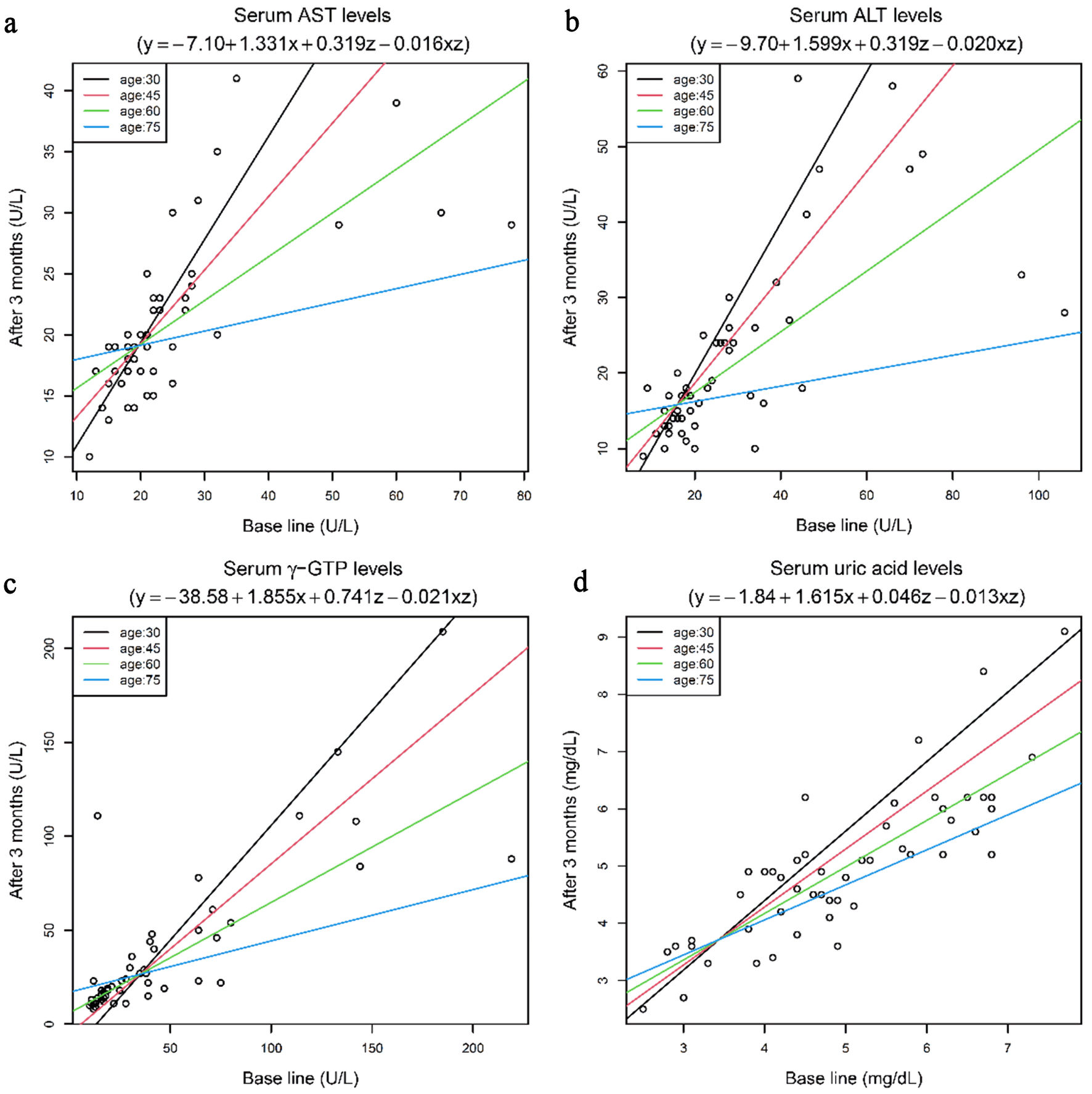 Click for large image | Figure 1. Scatter plot of the serum AST, ALT, γ-GTP, and uric acid levels before and after 3 months of imeglimin treatment, with regression line for each age group. (a) Serum AST levels. (b) Serum ALT levels. (c) Serum γ-GTP levels. (d) Serum uric acid levels. In the equations, x represents baseline values, y represents values after 3 months, and z represents age. N = 52. Regression analysis was performed. AST: aspartate aminotransferase; ALT: alanine aminotransferase; γ-GTP: γ-glutamyl transferase. |
In addition, we then attempted conducting the same analysis after logarithmic transformation of the three liver function parameters (AST, ALT and γ-GTP) from the viewpoint of improving the data normality, and obtained similar results. The blood level of HbA1c, which is the major target of imeglimin treatment, was not affected by the patient’s age.
| Discussion | ▴Top |
Imeglimin, a novel antidiabetic drug, is currently used only in Japan. Imeglimin has been reported to significantly reduce HbA1c levels in type 2 diabetes patients and is considered a drug with a favorable safety profile [6]. Imeglimin exerts a unique effect of improving the mitochondrial functions [1, 2]. The drug stimulates insulin secretion [2] and improves glucose metabolism in the liver and skeletal muscles [3, 4].
In the treatment of diabetes mellitus, it is usual to initially prescribe therapeutic lifestyle interventions, and then to add drug therapy if these prove insufficient for obtaining adequate glycemia control. In practice, there are also patients in whom therapeutic lifestyle interventions are difficult to implement. In such cases, it may not be possible to achieve sufficient blood glucose control by drug therapy.
The present study was undertaken to clarify the efficacy and safety of imeglimin in Japanese patients with type 2 diabetes, in whom it was difficult to apply therapeutic lifestyle interventions. The study was additionally aimed at investigating differences in the drug’s efficacy depending on the patients’ age.
In this study, treatment with imeglimin resulted in a significant reduction of the body weight and BMI. Although the exact reason for the reduction in these parameters remains unclarified, it seems likely that the reduction in body weight was associated with gastrointestinal symptoms, which occur often as adverse reactions to imeglimin. On the other hand, there are also some reports suggesting that imeglimin treatment had no effect on the body weight [7, 8].
The blood level of HbA1c, the primary treatment target, decreased significantly following imeglimin treatment. With regard to imeglimin, recent reports on imeglimin have suggested that prolongation of erythrocyte lifespan may lead to relatively elevated HbA1c levels [9].
In regard to the blood lipid levels, significant decreases of the serum TC, LDL-C and non-HDL-C were observed following imeglimin treatment; as a result, the AI index and L/H ratio also decreased. Patients with type 2 diabetes often show abnormal lipid metabolism, which is a risk factor for atherosclerosis [10]. In the present study, imeglimin improved the lipid profile of patients with type 2 diabetes, thereby reducing the risk of atherosclerosis. Patients with type 2 diabetes tend to be obese, and their body weight often reduces with improved blood glucose control, accompanied by an improvement in the blood lipid profile. The effect of imeglimin in improving the lipid profile observed in the present study could also be explained with this sequence of changes. However, according to one previous study, imeglimin treatment had no effect on the blood lipid levels [8].
Taken together, the results of the present study suggest that imeglimin is a useful drug for improving the glucose metabolism and lipid metabolism in Japanese patients with type 2 diabetes in whom it is difficult to implement therapeutic lifestyle interventions.
In regard to the drug’s safety, imeglimin treatment resulted in a significant reduction of the serum levels of ALT and AST among the three important liver function parameters (ALT, AST and γ-GTP); the serum level of γ-GTP, while it tended to decrease, showed no significant change following imeglimin treatment. Fukunaga et al reported a significant reduction of all of ALT, AST and γ-GTP following imeglimin treatment [7].
Imeglimin treatment had no effect on the parameters of renal function (Cre, eGFR and BUN). The serum amylase and uric acid levels also remained unaffected. These results suggest that imeglimin is an antidiabetic drug with a good safety profile.
Regression analysis was carried out to analyze the influence of the patients’ age on the levels of each of the measured parameters before the start and at the end of 3-month imeglimin treatment period. This analysis revealed that the effect of imeglimin in reducing the serum levels of the hepatic function parameters AST, ALT, and γ-GTP, and the serum uric acid level was influenced by interactions between the patient’s age and baseline levels of these parameters. The higher the patient’s age, the greater was the efficacy of imeglimin in reducing the levels of these parameters (Fig. 1). The significant decrease in the blood level of HbA1c, the major target of imeglimin treatment, was not influenced by the patient’s age, so that imeglimin could be considered as a useful antidiabetic drug for patients over a wide age range, covering juvenile to elderly groups.
Liver function is often compromised in patients with type 2 diabetes, and a high percentage of these patients show evidence of hepatic steatosis [11]. Hepatic steatosis has been reported to be associated with the risk of progression to nonalcoholic steatohepatitis (NASH) [12, 13]. Therefore, the reductions in the hepatic function parameters of AST and ALT by imeglimin treatment in this study may be interpreted as representing advantageous effects of the drug. In addition, it appeared that this effect of imeglimin was reinforced by aging, suggesting that this drug might be particularly suitable for elderly type 2 diabetes patients with hepatic dysfunction. Another advantage of imeglimin in elderly patients is that the drug’s effect of reducing the serum uric acid level (which is known to rise with advancing age) was also reinforced by aging.
This trial did not include a control group. Furthermore, the number of patients was small.
These are the limitations of this study. We plan to conduct a further trial in the future, incorporating a control group, if possible, and increasing the number of patients.
In conclusion, imeglimin treatment was associated with a significant reduction in the serum level of HbA1c (a major treatment target) in Japanese patients with type 2 diabetes in whom therapeutic lifestyle interventions were difficult to implement, irrespective of the patient’s age, and without posing any particular safety risk. These results indicate that imeglimin is a useful antidiabetic drug for patients over a wide age range, covering juvenile to elderly groups. Furthermore, our results suggested that the effect of imeglimin in reducing the serum levels of AST, ALT, γ-GTP and uric acid was reinforced by aging, so that the drug can be used without any safety concerns even in patients with type 2 diabetes who are elderly and/or have hepatopathy.
Acknowledgments
There is no specific acknowledgment that warrants mention.
Financial Disclosure
The research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no financial or non-financial conflict of interest to report.
Informed Consent
Informed consent was obtained.
Author Contributions
MK designed and performed the study. MK, SM, TM and KT drafted the manuscript and did critical editing. SM, KK, FH, DM, MK and KK assisted and supported in data collection and subsequent analysis with statistics.
Data Availability
Any inquiries regarding the availability of the supporting data from this study should be directed to the corresponding author.
Abbreviations
HbA1c: hemoglobin A1c; AST: aspartate aminotransferase; ALT: alanine aminotransferase; γ-GTP: γ-glutamyl transpeptidase; BMI: body mass index; Cre: creatinine; BUN: blood urea nitrogen; HPLC: high performance liquid chromatography; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; L/H: LDL-C/HDL-C; AI index: atherosclerosis index; eGFR: estimated glomerular filtration rate; NASH: nonalcoholic steatohepatitis
| References | ▴Top |
- Pirags V, Lebovitz H, Fouqueray P. Imeglimin, a novel glimin oral antidiabetic, exhibits a good efficacy and safety profile in type 2 diabetic patients. Diabetes Obes Metab. 2012;14(9):852-858.
doi pubmed - Hallakou-Bozec S, Kergoat M, Fouqueray P, Bolze S, Moller DE. Imeglimin amplifies glucose-stimulated insulin release from diabetic islets via a distinct mechanism of action. PLoS One. 2021;16(2):e0241651.
doi pubmed - Vial G, Lamarche F, Cottet-Rousselle C, Hallakou-Bozec S, Borel AL, Fontaine E. The mechanism by which imeglimin inhibits gluconeogenesis in rat liver cells. Endocrinol Diabetes Metab. 2021;4(2):e00211.
doi pubmed - Vial G, Chauvin MA, Bendridi N, Durand A, Meugnier E, Madec AM, Bernoud-Hubac N, et al. Imeglimin normalizes glucose tolerance and insulin sensitivity and improves mitochondrial function in liver of a high-fat, high-sucrose diet mice model. Diabetes. 2015;64(6):2254-2264.
doi pubmed - Akaike H. A new look at the statistical model identification. IEEE Transactions on Automatic Control. 1974;19(6):716-723.
- Dubourg J, Fouqueray P, Quinslot D, Grouin JM, Kaku K. Long-term safety and efficacy of imeglimin as monotherapy or in combination with existing antidiabetic agents in Japanese patients with type 2 diabetes (TIMES 2): A 52-week, open-label, multicentre phase 3 trial. Diabetes Obes Metab. 2022;24(4):609-619.
doi pubmed - Fukunaga K, Morishita A, Imachi H, Oura K, Sato S, Kobayashi T, Saheki T, et al. Efficacy of imeglimin in patients with type 2 diabetes mellitus complicated by metabolic dysfunction-associated steatotic liver disease: A multicentre study. Diabetes Obes Metab. 2025;27(3):1498-1506.
doi pubmed - Oyanagi T, Kawanabe S, Tsukiyama H, Nishine A, Nakamura Y, Nakagawa T, Kanou M, et al. The effects of imeglimin on muscle strength in patients with type 2 diabetes: a prospective cohort study. Diabetes Ther. 2024;15(11):2323-2336.
doi pubmed - Osonoi T, Shirabe S, Saito M, Hosoya M, Watahiki N, Shiozawa N, Douguchi S, et al. Imeglimin may affect hemoglobin A1c accuracy via prolongation of erythrocyte lifespan in patients with type 2 diabetes mellitus: insights from the INFINITY clinical trial. Front Endocrinol (Lausanne). 2025;16:1699591.
doi pubmed - Goldstein JL, Hazzard WR, Schrott HG, Bierman EL, Motulsky AG. Hyperlipidemia in coronary heart disease. I. Lipid levels in 500 survivors of myocardial infarction. J Clin Invest. 1973;52(7):1533-1543.
doi pubmed - Bhatt HB, Smith RJ. Fatty liver disease in diabetes mellitus. Hepatobiliary Surg Nutr. 2015;4(2):101-108.
doi pubmed - Firneisz G. Non-alcoholic fatty liver disease and type 2 diabetes mellitus: the liver disease of our age? World J Gastroenterol. 2014;20(27):9072-9089.
doi pubmed - Le TA, Loomba R. Management of non-alcoholic fatty liver disease and steatohepatitis. J Clin Exp Hepatol. 2012;2(2):156-173.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.