| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://jem.elmerpub.com |
Original Article
Volume 15, Number 1, March 2025, pages 1-14
Altered Hormone and Bioactive Lipid Plasma Profile in Rodent Models of Polycystic Ovarian Syndrome Revealed by Targeted Mass Spectrometry
Hannah C. Scotta, b, h, Martin Philpottc, d, Georgina Berridgeb, Zhanru Yua, b, Darragh P. O’Brienb, Christian Beckere, Cecilia Lindgrene, f, Udo Oppermannc, Joerg Muellerg, Martin Fritschg, Adan Pinto-Fernandeza, b, Benedikt M. Kesslera, b, h
aChinese Academy of Medical Sciences Oxford Institute, Nuffield Department of
Medicine, University of Oxford, Oxford, OX3 7BN, UK
bTarget Discovery Institute,
Centre for Medicines Discovery, Nuffield Department of Medicine, University of Oxford, Oxford
OX3 7FZ, UK
cBotnar Research Centre, Nuffield Department of Orthopaedics,
Rheumatology and Musculoskeletal Sciences, University of Oxford, Oxford, OX3 7LD,
UK
dPresent address: Caeruleus Genomics, Bioescalator Innovation Building, Old
Road Campus, Roosevelt Drive, Oxford, OX3 7FZ, UK
eNuffield Department of
Women’s and Reproductive Health, Women’s Centre, John Radcliffe Hospital, Oxford,
OX3 9DU, UK
fBig Data Institute, Li Ka Shing Centre for Health Information and
Discovery, Nuffield Department of Population Health and the Nuffield Department of Medicine,
University of Oxford, Oxford, OX3 7LF, UK
gBayer AG, Research and Development,
Mullerstrabe 178, 13353 Berlin, Germany
hCorresponding Author: Hannah C. Scott
and Benedikt M. Kessler, Chinese Academy of Medical Sciences Oxford Institute, Nuffield
Department of Medicine, University of Oxford, Oxford, OX3 7BN, UKand
Manuscript submitted October 15, 2024, accepted February 18, 2025, published online March 14,
2025
Short title: Altered Hormone and Lipid Plasma in PCOS Rats
doi:
https://doi.org/10.14740/jem1042
| Abstract | ▴Top |
Background: Polycystic ovarian syndrome (PCOS) symptoms include excessive body or facial hair, irregular periods, reduced fertility, and reoccurring pregnancy loss. Hyperandrogenism and chronic inflammation are hallmarks of PCOS, which is diagnosed by analyzing steroid hormones in the blood. Studies suggest that bioactive lipids are contributing to chronic inflammation.
Methods: To research PCOS, animal models, such as letrozole- and dihydrotestosterone-treated rats, are used. They display similar ovarian and metabolic characteristics, although plasma lipid profiles have not been determined. Therefore, in order to validate the use of these models for PCOS, we have optimized a mass spectrometry-based targeted lipidomics workflow, which increases the sensitivity of measuring these lipids in rat plasma.
Results: Our analysis shows that letrozole caused a significant elevation of 5α-androstene-3,17-dione and testosterone. Dihydrotestosterone treatment resulted in increased dehydroepiandrosterone-sulphate and allopregnanolone but a reduction in testosterone, progesterone, pregnenolone, and D-sphingosine. In both models, 25-hydroxycholesterol and leukotriene C4 were significantly diminished, and 4-cholesten-3-one was significantly increased, and these particular metabolites are not known to be changed in human PCOS.
Conclusion: These results suggest that the plasma lipids of these rodent models exert altered profiles of sterols, leukotrienes and steroid hormones akin to human PCOS but with notable differences.
Keywords: Steroid hormones; Lipids; Eicosanoids; Sphingolipids; Lipidomics; Letrozole; Hyperandrogenism; Inflammation
| Introduction | ▴Top |
Polycystic ovarian syndrome (PCOS) consists of a multifactorial cluster of diseases; the dysregulation of hormone homeostasis underlies reproductive, metabolomic and cardiovascular complications. PCOS is diagnosed based on the presentation of physiological traits, including disturbances in ovulation including oligoovulation or anovulation [1], hyperandrogenism [2], hyperinsulinemia [3], insulin resistance [4], hirsutism [5], acne [6], androgenic alopecia [7], fertility problems, chronic inflammation [8] and polycystic ovaries. PCOS is the most common cause of infrequent or absent periods in women. Preliminary diagnosis of the condition requires steroid hormone analysis to confirm hyperandrogenism [9]. Historically, immunoassays have been used, although more recently, mass spectrometry (MS)-based methods have proven to be more reliable in the identification of PCOS patients with hyperandrogenism [10].
MS methods for steroid and steroid hormone analysis have been developed [11, 12], with targeted approaches such as dynamic multiple reaction monitoring (dMRM) being able to improve accurate measurement of endogenous species [13]. In particular, MS-based profiling has been performed across many different sample types including serum, plasma, ovarian venous blood, follicular fluid, and various tissues derived from pre-/post-menopausal women, plus endometriosis and PCOS patients [14-16]. Measurements indicate that PCOS patients have elevated levels of specific steroid hormones [17] including testosterone [18], 4-androstene-3,17-dione (A4) [19], dehydroepiandrosterone-sulphate (DHEA-S) [20], 5α-androstane-3,17-dione (Aα5) [19], aldosterone [21], cortisone [22], 11-deoxycortisol [23], corticosterone [24], 17α-hydroxyprogesterone [25], 17α-hydroxypregnenolone [26], estriol [27], estrone [28], pregnenolone [24] and dehydroepiandrosterone (DHEA) [29]. In addition, PCOS patients have lower amounts of progesterone [30]. Changes in serum bioactive lipids, in particular omega-fatty acids (Ω-FAs) upstream of arachidonic acid (AA), eicosanoids downstream of AA [31] and sphingolipids [32], have also been implicated to be aberrant in PCOS patients [33].
Letrozole (LET) is a non-steroidal aromatase inhibitor that interferes with the action of the enzyme CYP19A [34]. CYP19A catalyzes the metabolism of testosterone and A4, plus their metabolites to estradiol and estrone, respectively. LET is used as an infertility treatment in PCOS patients by inducing ovulation [35]. However, LET is also able to induce PCOS in rats, mimicking human PCOS physiology [36], therefore is used as a model for human PCOS. 5α-dihydrotestosterone (DHT)-treated rats also display human traits of PCOS pathophysiology and can be used as a suitable alternative model of this disease [37].
Here, we have optimized and applied a targeted analysis for the identification of a selected panel of steroid, steroid hormones and bioactive lipids in the plasma of rats, whereby PCOS is induced by either LET or DHT treatment. Ultra-pressure liquid chromatography coupled to triple quadrupole mass spectrometry (UPLC-MS) using dMRM was optimized for ultra-sensitivity of detection and for minimizing matrix interference (Fig. 1). We highlight the challenges of profiling these lipids in complex matrices such as plasma, compare the lipid profiles of these two rat models of PCOS, discuss the differences and similarities to the lipid profiles observed in human PCOS patients and outline potential molecular mechanisms that contribute to the pathophysiology of this syndrome.
| Materials and Methods | ▴Top |
Reagents
Female Wistar Han IGS rats were ordered from Charles River (strain code 273). Rodent diet with 45% fat (D12451) was purchased from Ssniff. Lipid standards 17α-hydroxypregnenolone (H-105-1ML), DHEA (D-063-1ML), pregnenolone (P-104-1ML), 17α-hydroxyprogesterone (H-105-1ML) and 11-deoxycortisol (D-061-1ML) were purchased from Sigma-Aldrich. Liquid chromatography mass spectrometry (LC-MS) grade water (115333) and methanol (106035), aldosterone (A9477), progesterone (P0130), Aα5 (A7755-100MG), cortisone (C2755-5G), corticosterone (27840), 5α-DHT (D-073-1ML), testosterone (86500-5G), DHEA-S (700086P-10MG), D-sphingosine (D-Sph) (d18:1) (860490P-10MG), leukotriene C4 (LTC4) (700342E-100UG) and estrone (E9750-5G) were available from Merck. Allopregnanolone (CAY16930-1 mg) was acquired from Cambridge Bioscience Limited. Ammonium acetate (10365260) and isopropanol (15686670) were from Fisher Scientific. Methyl tert-butyl ether (MTBE) was acquired from Acros Organics (3787 20010) and acetonitrile was from Honeywell Riedel-de Haen (348512.5L).
Estradiol, estriol, 25-hydroxycholesterol, dihydrocholesterol (diOHchol) and 4-cholesten-3-one were kindly gifted by Professor Udo Oppermann at the Botnar Research Centre (University of Oxford).
Preparation of steroid and lipid standard solutions
The standard mixture was composed of LTC4, estrone, estradiol, estriol, aldosterone, 25-hydroxycholesterol, Aα5, cortisone, testosterone, DHEA, DHEA-S, 17α-hydroxyprogesterone, 11-deoxycortisol, progesterone, diOHchol, 17α-hydroxypregnenolone, pregnenolone, D-Sph, γ-linolenic acid (γ-LA), 5α-DHT, allopregnenolone and 4-cholesten-3-one.
Individual lipid standards were prepared in 100% MeOH, then pooled and diluted to a final concentration of 50 nmol/L.
In vivo models
Animal experiments with rats were performed as described previously [37, 38]. In brief, female rats were purchased at an age of 2 weeks, with their foster mothers, from Charles River Laboratories Inc., Sulzbach, Germany. Rats were raised until 21 days of age and then used for experiments. Rats were randomly divided into three experimental groups: control (n = 6), DHT (n = 3), and LET (n = 3) and implanted subcutaneously with continuous-release implants (Bayer AG, Berlin, Germany) containing either poly-lactic-glycolic acid (PLGA) matrix only (control), 18 mg LET in PLGA at a daily dose of 200 µg, or 11 mg DHT in PLGA at a daily dose of 80 µg. Rats were kept on a high fat diet from Sniff until the study was concluded. When the rats were 49 days of age, blood samples were taken to isolate plasma, which was prepared by lithium heparin protocol. Plasma was kept at -80 °C until lipid extraction and MS analysis. Protocols of accepted standards of animal care were approved by the Landesamt fur Gesundheit und Soziales (LaGeSo, Berlin, Germany) under the animal allowance number A 0077/17.
Lipid extraction
A small amount of coagulation (a clot) was seen in plasma samples: LET-C biological replicates 1, 2 and 3 and LET biological replicates 1, 2 and 3. Plasma (25 µL) material was pipetted to avoid the coagulation clot and processed further for MS analysis.
Collected plasma material was thawed on ice (4 °C), lipids were then extracted with the addition of 100 µL of 50% methanol in water, followed by the same volume of MTBE, vortexed and then mixed at 4 °C for 20 min with gentle rotation. The sample was then centrifuged at 17 × g at 4 °C for 5 min. The upper organic layer was removed and dried by vacuum centrifugation (speedvac). Samples were stored at -20 °C until LC-MS analysis. Before analysis, the lipid extracts were reconstituted into 40 µL of methanol.
All samples were pooled by combining a small portion of each sample to create a “matrix sample” for determining the matrix effects. Two “matrix” samples were extracted as described above by extracting from 20 µL of the pooled plasma. To one of the “matrix” samples, the methanol in the extraction protocol was substituted for the standard mixture (section “Preparation of steroid and lipid standard solutions”), to create a “matrix plus standards” sample.
LC-MS method
Levels of LTC4, estrone, estradiol, estriol, aldosterone, 25-hydroxycholesterol, Aα5, cortisone, testosterone, DHEA, DHEA-S, 17α-hydroxyprogesterone, 11-deoxycortisol, progesterone, diOHchol, 17α-hydroxypregnenolone, pregnenolone, D-Sph, γ-LA, 5α-DHT, allopregnenolone and 4-cholesten-3-one were analyzed. Metabolites were quantified by LC-MS using an optimized dMRM method on a triple quadrupole mass spectrometer with a JetStream ESI source (Agilent 6495) coupled to a 1290 Agilent LC system.
Lipids were separated on an ACQUITY UPLC BEH C18 column (1.7 µm, 100 × 2.1 mm i.d., Waters). Mobile phase A was 40% acetonitrile with 5 mM ammonium acetate and mobile phase B was 90% isopropanol, 10% acetonitrile with 5 mM ammonium acetate. The flow rate was set to 0.2 mL/min, and the sample injection volume was 10 µL. The following gradient (% mobile phase B) was used: 0 - 2.5 min at 2% B, 2.5 - 25 min 100% B. A wash with 100% mobile phase B was performed to clean the column before re-equilibration to starting conditions. The autosampler was maintained at 4 °C.
The following ESI source parameters were used: gas temp at 280 °C, gas flow 14 L/min, nebulizer at 20 psi, sheath gas temp at 250 °C, sheath gas flow at 11 L/min, capillary voltage 4,000 V, nozzle voltage 1,000 V, high-pressure RF at 150 V and low-pressure RF at 90 V. The MS was operated in fast switch mode, changing polarity from positive to negative, depending on the dMRM transitions. The dMRM fragmentor voltage (V) was set at 380 and the cell accelerator voltage at 4. The LC and MS parameters used are shown in Supplementary Table 1 (jem.elmerpub.com). The transitions used in the dMRM analysis are shown in Supplementary Table 2 (jem.elmerpub.com).
Data analysis
Initial data processing was performed using Agilent MassHunter Quantitative Analysis software (v. 10). Post-processing was performed in Excel and GraphPad Prism, in which dMRM responses were corrected with a blank subtraction by removing any response in a methanol only injection. Responses were corrected for drift in instrument performance by adjusting to a quality control sample and then modified by the matrix response factor (matrix RF). To determine the matrix response, and to correct for any matrix effects, we used our previously published protocol for data analysis [39]. Matrix RF-corrected response can subsequently be used to calculate analyte concentration from a standard curve trendline equation (y = Mx + C). Standard curves are generated by the injection of increasing concentrations of the lipid standard solution, calculating the response at each concentration, and plotting the values against the corresponding concentration.
Statistical analysis of the two-sample or unpaired t-test was used to validate difference between no treatment (control) and treated with LET or DHT. A P-value less than 0.05 (*) indicates that the results have highly significant differences. If there is no significance difference, it is unlabeled.
Figures
The illustrations shown in the graphical abstract (Fig. 1) and Figures were created using BioRender.com. Chemical structures shown in Supplementary Figures were made with ChemDraw version 22.2.0.3300, PerkinElmer Informatics, Inc.
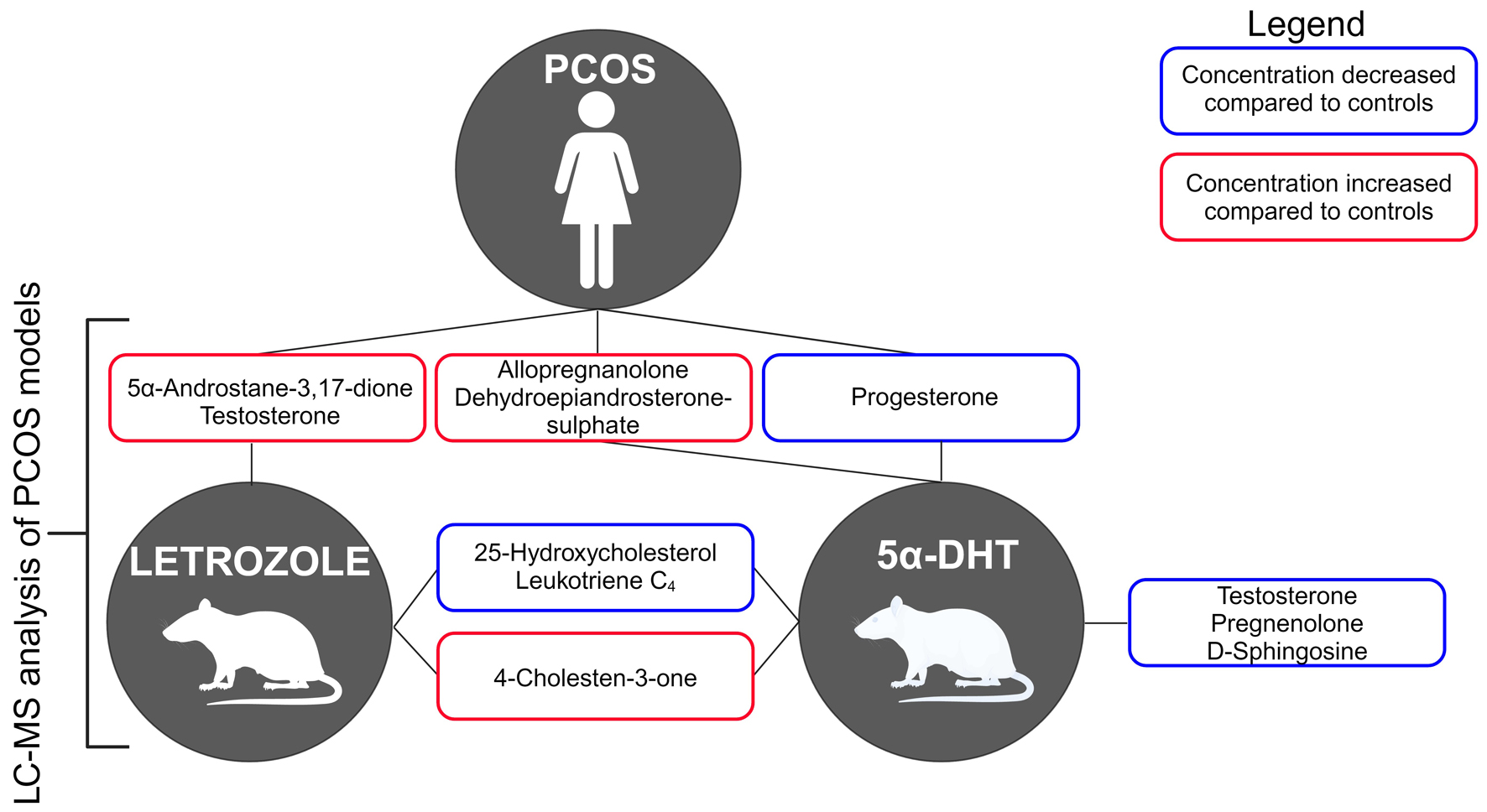 Click for large image |
Figure 1. Graphical abstract. Plasma lipid and hormone profiling in a polycystic ovarian syndrome (PCOS) rat model. PCOS symptoms include excessive body or facial hair, irregular periods, reduced fertility, and reoccurring pregnancy loss. To validate the use of rat models for PCOS, we have optimized a mass spectrometry-based targeted lipidomics workflow. Our work provides new insights into altered lipid and hormone profiles (red boxes - increased, blue boxes - decreased abundance) associated with human disease, recapitulated partially in PCOS rat models. |
Method validation
The lower limit of detection (LLOD) was calculated as a signal-to-noise (S/N) ratio of > 3; the lower limit of quantification (LLOQ) was a S/N ratio of > 10. Intraday precision was calculated using three replicates of two concentrations over the course of 1 day and the results are reported as coefficient of variation (CV in %) between replicates of one concentration. To monitor instrument performance over time and check that analysis that spanned interday were consistent, a quality control sample, which contained 3.5 fmol/L on-column (o.c.) of testosterone was routinely injected. If the total sample analysis time was over multiple days, the samples were briefly vortexed at the start of each day to avoid precipitation. Variations in biological and technical replicates were investigated and reported in the Supplementary Materials section.
| Results | ▴Top |
Lipid standards were selected based on availability and to cover metabolites representing steroids, steroid hormone biosynthesis and degradation, eicosanoid metabolism and sphingolipid metabolism (Fig. 2a). Our panel included lipids from the cholesterol, progestin, mineralocorticoid, glucocorticoid, androgen, estrogen, fatty acid, eicosanoid and sphingolipid families (22 in total, Supplementary Fig. 1, jem.elmerpub.com). We developed a lipidomics workflow for analysis of these lipids at low concentrations and for minimal matrix interference in rat plasma (Fig. 2b). Chromatographic separation of each of the lipid standards was optimized and their detection levels maximized in ESI ionization MS. Tandem MS (MS2) spectra were generated to determine diagnostic fragment ions, as exemplified for testosterone (Supplementary Fig. 2A, jem.elmerpub.com). The protonated mass of testosterone was determined at mass-to-charge (m/z) 289, the sodium adduct is at m/z 311 and a protonated dimer at m/z 577.1, respectively. Endogenous sodium contribution may be from the plasma, although this is removed by the lipid extraction process as the lower aqueous fraction is discarded. We used this information to cross reference our spectra with those found in public databases, such as Human Metabolome Database and LipidMaps, to corroborate identification of the correct lipid. We established the retention time of the lipids by MS2 to determine a retention time window for the application of specific dMRM settings (Supplementary Fig. 2B, C, jem.elmerpub.com). MS-based quantitation of the lipid standard mixture (see section Materials and Methods: Preparation of steroid and lipid standard solutions) was established by a calibration curve of the quantification ion, demonstrating linearity over about 2 orders of magnitude, reaching 1 fmol/L o.c. (Supplementary Fig. 2D, jem.elmerpub.com). Representative dMRM chromatograms for both quantification and qualification ions for all the lipids studied were obtained by optimizing parameters for each individually (Fig. 2a, Supplementary Table 1, jem.elmerpub.com). This resulted in a compendium of dMRM settings applied for quantification of the panel of lipids profiled in this study (Supplementary Table 2, jem.elmerpub.com). The response curve equations and correlation coefficients varied from 0.917 for 5α-dihydrotestosterone to 0.999 for 11-deoxycortisol, Aα5, 17α-hydroxyprogesterone, progesterone, estrone, and testosterone, reflecting accurate quantitation over the defined concentration range (Supplementary Table 3, jem.elmerpub.com). Once optimal MS parameters were determined, we subsequently validated the reproducibility and robustness of our methodology by performing an intraday variation analysis (Supplementary Fig. 3, jem.elmerpub.com). Lipid standards were combined and analyzed twice at 100 fmol o.c. The variation, in percentage, from the first analysis was calculated for the second independent analysis. The observed variations for all the lipids were lower than 5% for most lipids, highlighting the reproducibility of the method. To detect the potential presence of isomers, we performed a transition ion ratio (TIR) analysis (Supplementary Table 4, jem.elmerpub.com). Testosterone, Aα5 and DHEA are isomers having the same molecular mass, therefore chromatographic separation is essential to provide accurate quantitative results. Testosterone and DHEA are chromatographically separated, removing the need for a TIR analysis in this case, but testosterone and Aα5 are only slightly separated. Therefore, the instability of the ratio observed in the TIR of testosterone could be due to Aα5 interference. However, this issue is mitigated by using different quantitation ion transitions for testosterone and Aα5.
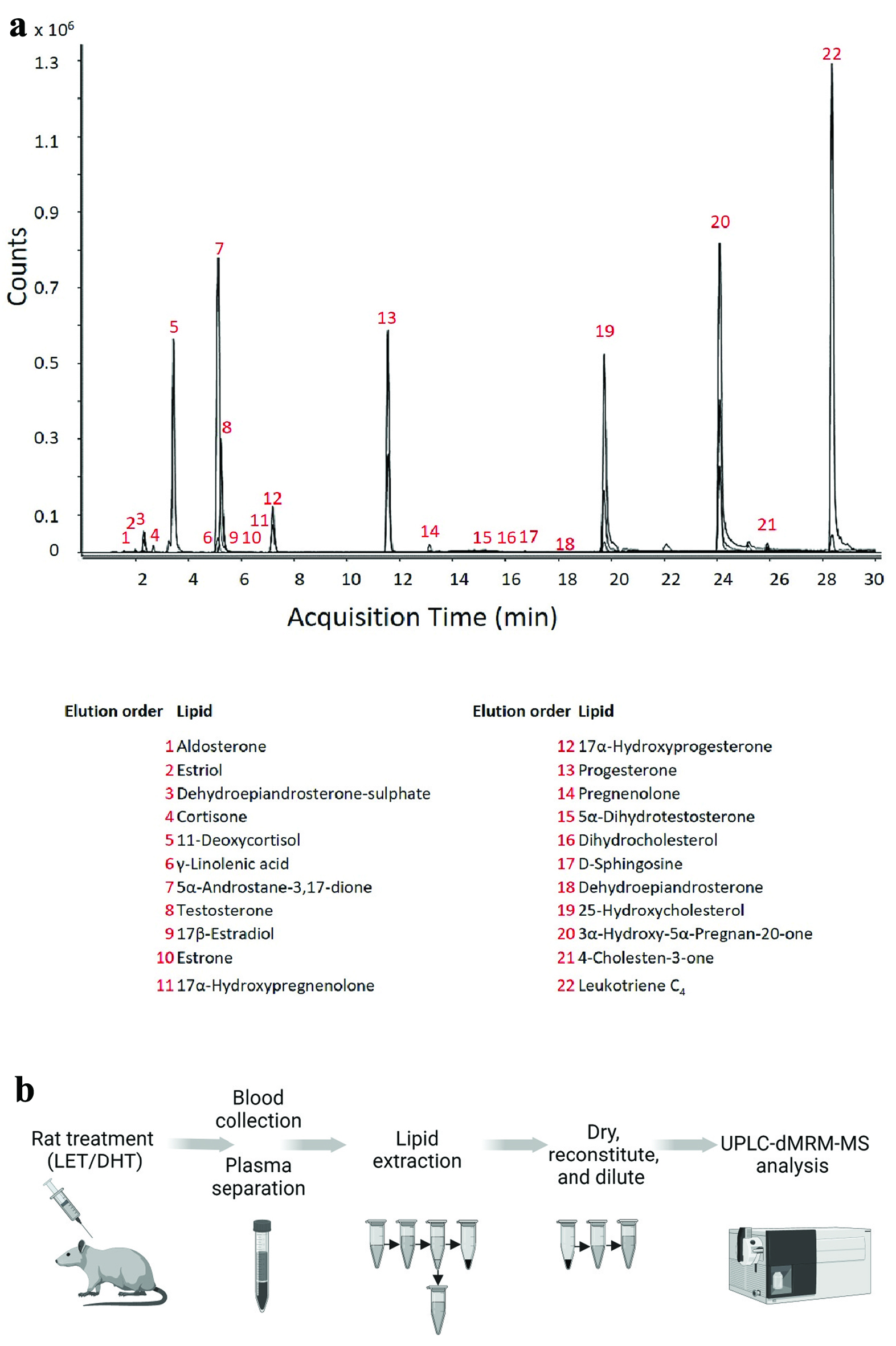 Click for large image |
Figure 2. Optimized lipid chromatography and experimental workflow for the analysis of steroids, steroid hormones and bioactive lipids in rat plasma. (a) The dynamic multiple reaction monitoring (dMRM) chromatograms for both quantitation and qualification ions for all the lipids analyzed with this methodology. The lipids are analyzed at the same on-column concentration of 300 fmol/L, the difference in counts for each lipid highlights the differences in ionization efficiencies of the lipids (molecular structures of analyzed lipids are in Supplementary Fig. 1, jem.elmerpub.com). (b) Analyses were conducted using an ultra-high-performance liquid chromatography-dMRM-mass spectrometry (UPLC-dMRM-MS) analysis. Rats were treated with either letrozole (LET) or dihydrotestosterone (DHT) to induce polycystic ovarian syndrome (PCOS)-like physiology. At age 49 days, blood was taken from rats, and plasma was obtained by lithium-heparin separation. Lipid extraction from plasma was performed with methanol/methyl-tert-butyl-ether (50:50, v:v). Lipid extracts were subsequently dried and reconstituted into methanol prior to LC-MS analysis. |
Having optimized analytical parameters, we set out to discover potential alterations of lipid profiles observed between individual rats for each treatment condition; LET, control for LET (LET-C), DHT and control for DHT (DHT-C) (Supplementary Fig. 4, jem.elmerpub.com). The CV for biological variation for the profiled lipids ranged from 1.06% for allopregnanolone (3α-hydroxy-5α-pregnan-20-one, AP) measurement in LET-C-treated rats, to 141.42% for Aα5 measurement in DHT-C rats (Supplementary Table 5, jem.elmerpub.com). Trace amounts of Aα5 in DHT-C were responsible for the high variation. It is worthwhile to note that LET-C and LET rat plasma had coagulation, and this may contribute to the biological variation seen (Materials and Methods section: Lipid extraction).
Technical triplicate analysis of each biological replicate revealed tighter consistency (Supplementary Fig. 5, jem.elmerpub.com). The CV for technical variation had a range of 0.02% for progesterone analysis in DHT-C biological replicate 1 to 90.23% for Aα5 analysis in LET-C biological replicate 3 (Supplementary Table 6, jem.elmerpub.com). Trace amounts of Aα5 were responsible for the high variation in LET-C. Aα5 was not present in DHT biological replicates 1 - 3 or in DHT-C biological replicates 1 and 2, therefore no technical variation analysis could be performed. The biological condition LET showed an average technical replicate CV of 7.34%, LET-C had 10.44%, DHT had 5.95% and DHT-C showed 7.30%. In conclusion, having established the variation of lipid measurements in rat plasma, we could then assess quantitative changes and their significance upon treatment conditions.
LET treatment of rats resulted in marked changes to the lipids profiled in rat plasma. We observed significant increases in the concentration of Aα5 (+135.75 fmol/L, P ≤ 0.0001), testosterone (+150.71 fmol/L, P ≤ 0.0001) and 4-cholesten-3-one (+72.53 pmol/L, P = 0.0063). DHEA-S, 11-deoxycortisol, pregnenolone, D-Sph and 3α-AP were also increased in LET-treated rat plasma, although these metabolites were not significantly elevated (Figs. 3a, c, 4, Supplementary Table 7, jem.elmerpub.com). A significant decrease in the plasma levels of 25-hydroxycholesterol (-4.21 pmol/L, P ≤ 0.0001) and LTC4 (-2.34 pmol/L, P = 0.0027) was observed. Progesterone and γ-LA were also reduced in LET-treated rat plasma, although these metabolites were not significantly decreased (Fig. 3a, Supplementary Table 7, jem.elmerpub.com).
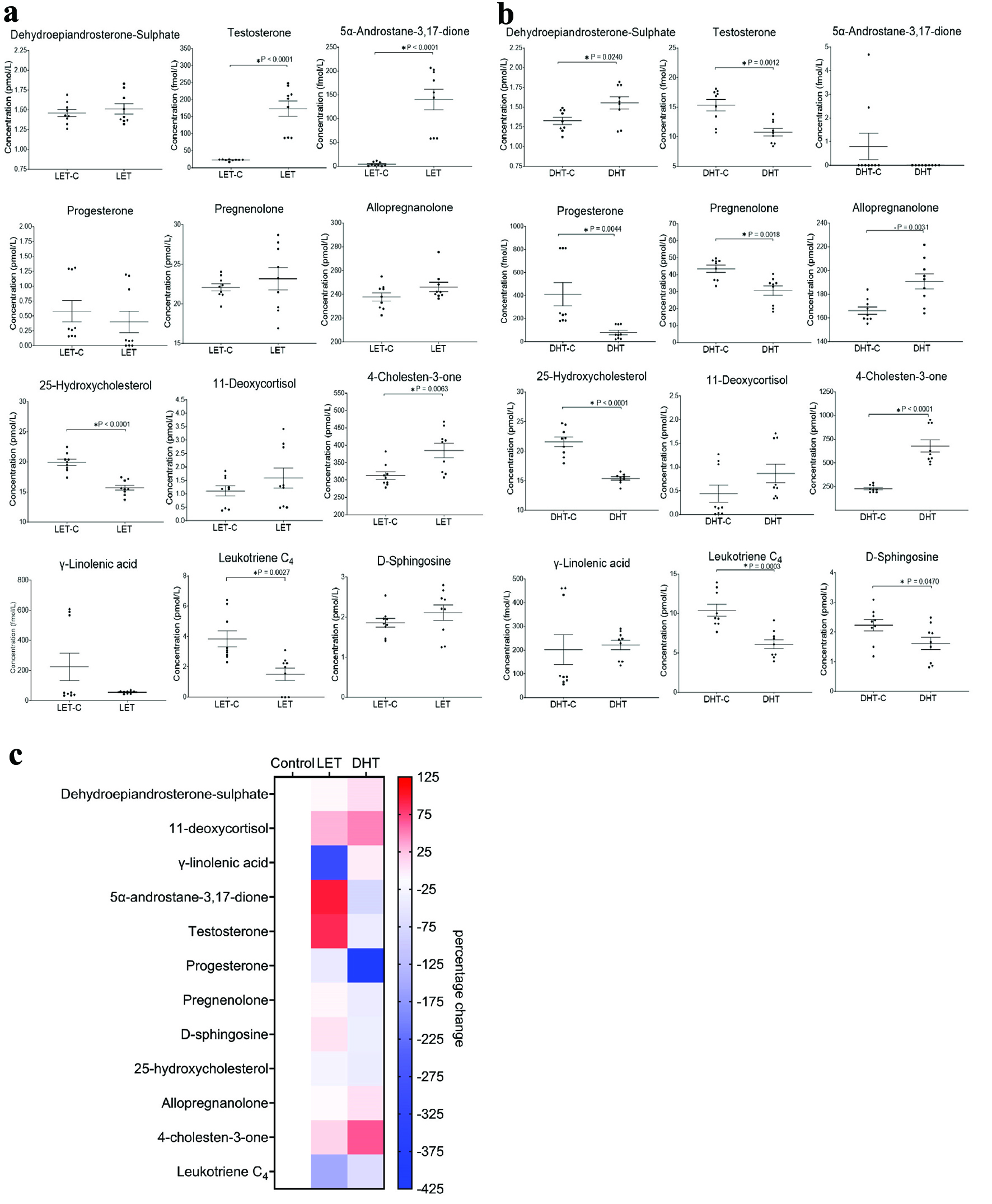 Click for large image |
Figure 3. Rat plasma profiles modulated by letrozole (LET) and dihydrotestosterone (DHT). (a) Analysis of lipids in rat plasma after LET treatment. LET induced polycystic ovarian syndrome (PCOS) in rats modulates steroid hormone biosynthesis, steroid hormone degradation, eicosanoid and sphingolipid pathways. The letrozole control (LET-C) is on the left and the LET treated is on the right. Black dots are each data point (technical triplicate of biological triplicates, n = 9). The statistical analysis is the two-sample (or unpaired) t-test. A P-value less than 0.05 (*) suggests that the results have highly significant differences. If there is no significant difference, it is not labelled. Values for mean, standard deviation (SD) and population number (n) are described in the Supplementary Materials section. (b) Analysis of lipids in rat plasma after DHT treatment. DHT induced PCOS in rats modulates steroid hormone biosynthesis, steroid hormone degradation, eicosanoid and sphingolipid pathways. Graphs represent the mean (line) with the standard error of the mean (error bars). The DHT control is on the left and the DHT treated is on the right. Black dots are each data point (technical triplicate of biological triplicates, n = 9). The statistical analysis is the two-sample (or unpaired) t-test. A P-value less than 0.05 (*) suggests that the results have highly significant differences. If there is no significance difference, it is not labelled. Values for mean, standard deviation (SD) and population number (n) are described in the Supplementary Materials section. (c) Analysis of lipids in rat plasma after LET or DHT treatment. Mean percentage change of lipids in rat plasma, not-treated control (control) and treated with LET or DHT. Heat maps display normalized lipid abundances relative to the mean concentration of the respective non-treated control samples (control), which is represented by the white boxes. For LET or DHT treatment, blue results are for a decrease in concentration, in percentage, white is for no change and red is for an increase in concentration in percentage. |
DHT treatment of rats also resulted in significant changes to the lipids profiled. For instance, we observed significant increases in the concentration of DHEA-S (+225 fmol/L, P = 0.024), allopregnanolone (+24.67 pmol/L, P = 0.0031) and 4-cholesten-3-one (+450.52 pmol/L, P < 0.0001) (Figs. 3b, c, 5, Supplementary Table 7, jem.elmerpub.com). 11-deoxycortisol and γ-LA were also increased in DHT-treated rat plasma, although these metabolites were not significantly elevated. Notably, a significant decrease in the plasma levels of testosterone (-4.55 fmol/L, P = 0.0012), progesterone (-332.34 fmol/L, P = 0.0044), pregnenolone (-12.90 pmol/L, P = 0.0018), D-Sph (-617 fmol/L, P = 0.047), 25-hydroxycholesterol (-6.20 pmol/L, P ≤ 0.0001), and LTC4 (-4.32 pmol/L, P = 0.0003) was observed.
Aα5 was also decreased in DHT-treated rat plasma, although this androgen was not significantly reduced. Aside from two data points (two technical replicates of one biological replicate), it is all but absent in both DHT and DHT-C samples (Fig. 3b, Supplementary Table 7, jem.elmerpub.com), contributing to the high variation seen in the analysis of this androgen.
Taken together, our optimized targeted MS method reported considerable changes in rat plasma steroid hormone and bioactive lipids upon LET and DHT treatment.
| Discussion | ▴Top |
Here, we investigated steroid hormones and pro-inflammatory lipid mediators in PCOS rat models to understand its underlying pathophysiology. The lipids analyzed in the rat models are representative of lipids involved in steroid hormones biosynthesis and inflammation. We choose bioactive lipids with known involvement in inflammation, and immunity to study this phenomenon in these two models.
Lipidomics remains challenging and continues to benefit from improved methodologies in detection, identification and sample preparation using applicable matrices [39]. Alternatively, a surrogate matrix for steroid hormone analysis may be used, e.g. charcoal stripped samples [40]. However, pre-analytical influences may change lipid concentrations or matrices, which is why we prefer to use extensive matrix-dependent normalizations for each analyte within a “true” matrix and not use a surrogate matrix. We demonstrate its application tailored to specific endogenous lipid detection in research relevant biological samples matrices as well as plasma in the present study.
Stable isotope labelling [41] or derivatization methods [42] may provide increased sensitivity for quantitative analysis of steroid hormones. Isomeric elucidation, such as determining the identification of, and causing the separation of Aα5 and 5β-Androstane-3,17-dione would be possible by using an MS equipped with ion mobility separation [43]. Alternatively, for determining the presence of isomers in biological matrices, the use of a TIR analysis is a possibility. However, TIR is based upon fragmentation of standards, and qualification ion fragments for standards are not always present in biological samples. This can be due to falling below the LLOD or LLOQ, matrix effects and other analytical singularities including concentration dependence [44]. Our preference is to use qualification ions solely when the identity of the lipid is ambiguous; however, our use of standards for confirmation of identification prevents this. In our analysis, not all qualification ions for every lipid profiled are present in all the samples (Supplementary Table 6, jem.elmerpub.com). Therefore, we believe that the application of TIR may not provide further advantages, at least for the study we describe here. TIR analysis of progesterone suggests the presence of isomers; however, the potential isomer of progesterone is currently unknown. Our method chromatographically separates the isomer species aldosterone and cortisone (C21H28O5), plus testosterone with DHEA and Aα5 (C19H28O2), allowing for accurate identification. However, for the isomer pairs androstenedione with 5α-DHT (C19H30O2), 11-deoxycorticosterone with 17α-hydroxyprogesterone (C21H30O3, 330.5 g/mol), and pregnenolone with 5α-dihydroprogesterone (316.49 g/mol), the situation is less clear. We did not include the standards for androstenedione, 11-deoxycorticosterone or 5α-dihydroprogesterone within our standard mixture, so are currently unable to know if these lipids are chromatographically resolved. We were also unable to detect 5α-DHT or 17α-hydroxyprogesterone in these samples. We are therefore unable to determine if the responses of 5α-DHT, 17α-hydroxyprogesterone or pregnenolone within our samples are contributed solely by these lipids or also include a contribution from their isomers. Measurement of 17α-hydroxyprogesterone, 17α-hydroxypregnenolone, corticosterone, diOHchol, DHEA, aldosterone, cortisone, 5α-DHT, estradiol, estrone and estriol was not detected in these samples, indicating either their absence, that they fell below the LLOD, and/or the matrix caused considerable ion suppression. Certain metabolites, such as LTC4, may benefit from a more tailored analysis that increases sensitivity and quantitation [39].
Despite natural biological heterogeneity, our results show clear differences in the concentration of Aα5 in LET-treated rats as well as differences in DHEA-S, progesterone, pregnenolone, allopregnanolone and D-Sph quantities in the plasma of DHT-treated rats as compared to controls. Both LET and DHT rats display considerable changes in testosterone, 4-cholesten-3-one, 25-hydroxycholesterol and LTC4. For both, LET and DHT treatment in the case of increased metabolite concentrations, this could suggest an increase in the synthesis of these lipids, a reduction in their metabolism or conversion, their cellular excretion maybe reduced.
Allopregnanolone (3α-hydroxy-5α-pregnane-20-one, AP) is a pregnane neurosteroid, which has been implicated in reproductive-related psychiatric disorders, such as post-partum depression [45] and premenstrual disorders [46, 47]. Women with PCOS experience increased rates of depression and anxiety [48]. Neuroactive steroids synthesized in the brain and/or crossing the blood-brain barrier (e.g., allopregnanolone and DHEA-S) may affect the serotonergic and glutamatergic systems, and act as allosteric agonists of the GABAA receptor, affecting sleep, mood and appetite [49]. Both allopregnanolone and DHEA-S have been implicated in the mental health complications and overeating caused by or associated with PCOS [50, 51]. However, human and animal models of PCOS have mixed and sometimes conflicting results on the contribution of allopregnanolone in PCOS development and pathology [52, 53]. In our analysis, progesterone is decreased in LET- (Fig. 4) and DHT-treated rats (Fig. 5), and allopregnanolone levels are elevated. Progesterone levels may be reduced due to conversion to 5α-dihydroprogesterone, and, subsequently, to allopregnanolone. DHEA-S is increased in both, LET- and DHT-treated rats as compared to controls. High allopregnanolone and DHEA-S amounts may be correlated with weight gain, psychiatric complications and associated metabolic syndromes in these animal models.
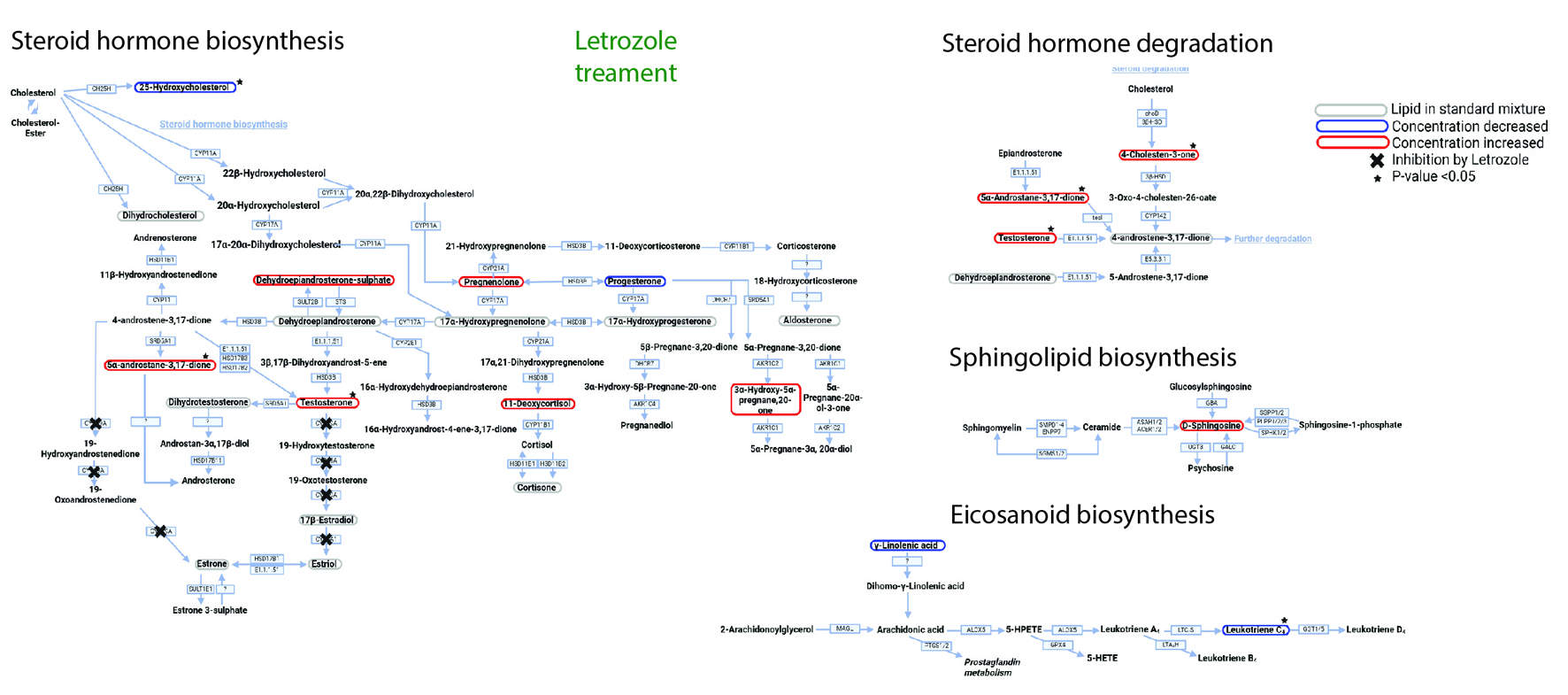 Click for large image |
Figure 4. Letrozole (LET) affects metabolic networks. LET treatment pathway analysis. Lipid profile after treatment with LET. Converting enzymes are in boxes and the lipids in this study are circled. Lipids circled in grey have either no change compared to non-treated control rat plasma or were not detected with this methodology; lipids circled in blue have a decreased concentration compared to non-treated control rats; and lipids circled in red have an increased concentration compared to non-treated control rats. Lipids with a significant difference between non-treated control and LET treated (a P-value of less than 0.05) are indicated by a black star and inhibition of CYP19A activity by LET with a black cross. |
 Click for large image |
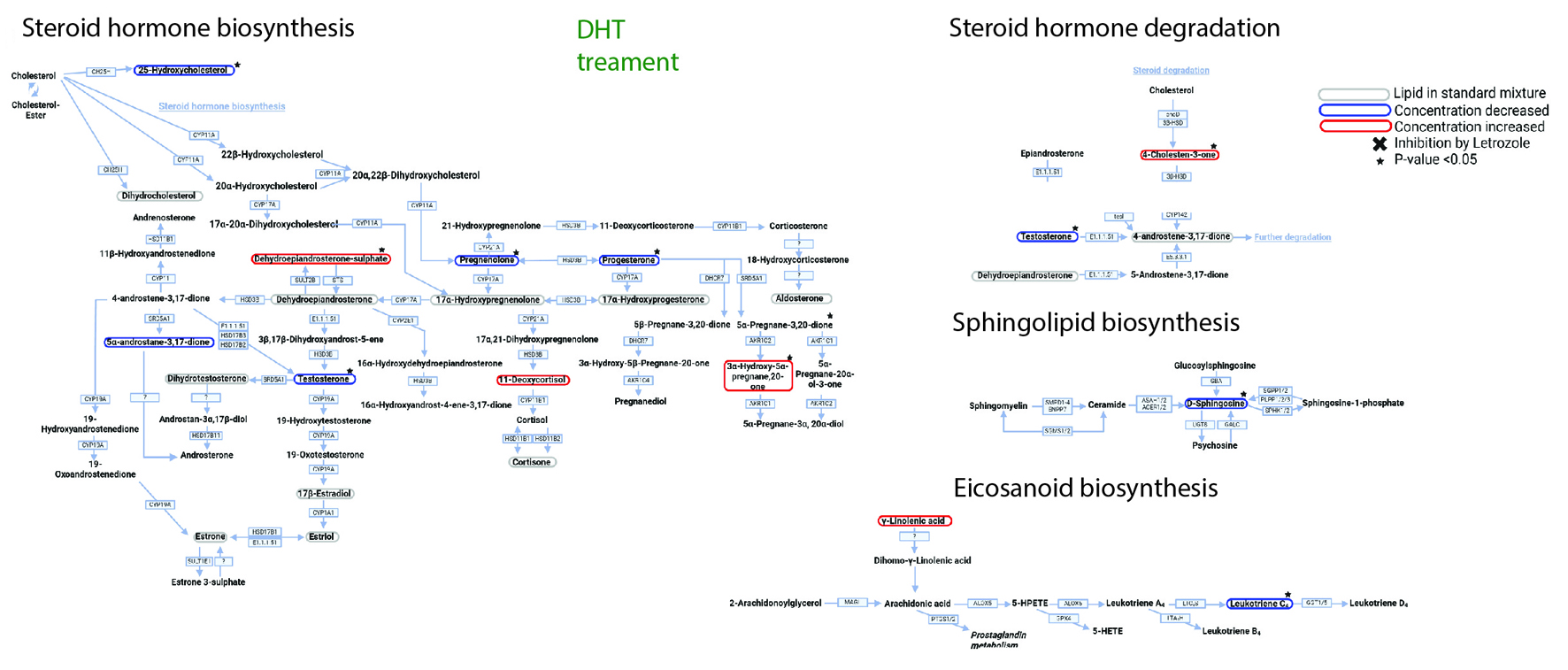
Figure 5. Dihydrotestosterone (DHT) affects metabolic networks. DHT treatment pathway analysis. Lipid profile after treatment with DHT. Converting enzymes are in boxes and the lipids in this study are circled. Lipids circled in grey have either no change compared to non-treated control rat plasma or was not detected with this methodology; lipids circled in blue have a decreased concentration compared to non-treated control rats; and lipids circled in red have an increased concentration compared to non-treated control rats. Lipids with a significant difference between non-treated control and DHT treated (a P-value of less than 0.05) are indicated by a black star. |
4-cholesten-3-one is a cholesterol metabolite involved in the steroid degradation pathway. It can reduce breast cancer cell viability, lipogenesis and enhance the expression of liver X receptor-dependent cholesterol transporters, including ABCA1 and ABCG1 [54]. 25-hydroxycholesterol, a primary bile acid, can be transported into cells by ABCA1/G1 [55]. Both rat models have high plasma levels of 4-cholesten-3-one and low amounts of 25-hydroxycholesterol, which may suggest the reduction of 25-hydroxycholesterol in the plasma is due to an increased expression of ABCA1/G1 by high 4-cholesten-3-one which has caused the cellular uptake of 25-hydroxycholesterol by ABCA1/G1.
CYP19A inhibition by LET treatment caused an increased concentration of testosterone, as it cannot be converted into downstream metabolites. Conversely, DHT treatment reversed the effect, with a significant decrease in testosterone. This could be because DHT and testosterone are within the same metabolic pathway, and high levels of DHT may cause a negative feedback loop, leading to a decreased production of testosterone, to control DHT levels. The decrease in testosterone observed in DHT-treated rats may also be the result of testosterone glucuronidation, by uridine diphosphoglucuronosyltransferase enzymes, which lowers plasma testosterone levels through secretion of testosterone-glucuronidation products [56].
In addition to LET inhibition of CYP19, Aα5 is also capable of inhibiting CYP19 activity. Reduction in CYP19 catalyzation reduces estrogen production from androgens. Aα5 has been shown to be elevated in PCOS patients [19], and therefore may contribute to reduced estrogen levels in humans with PCOS. Estrogens are not identified in our analysis of PCOS rodent models, potentially by Aα5 contributing to further inhibition of aromatase or by glucuronidation of androgens, or a combination of both. In our analysis, Aα5 is increased in LET-treated rats, but completely absent in DHT-treated rats. Aα5 was absent in two biological replicates for the DHT-control rat group and detected at very low levels in the LET-control rats (Fig. 3c). This may be due to LET-mediated inhibition of CYP19A mediating metabolic adaptations that are not mimicked by DHT administration [57].
Androgens are involved in the expression of the enzyme cyclooxygenase-2 [58], which metabolizes AA to prostaglandins. Abnormal cyclooxygenase-2 levels are associated with infertility, failure of ovulation and disorders of oocyte implantation [59]. Androgens, γ-LA, an Ω-FA capable of producing AA, and AA itself are increased in certain PCOS subtypes [60]. In one rat model of PCOS, induced by alendronate, prostaglandins are increased in ovarian tissue [61]. These associations suggest a strong link between steroid hormones and mediators of inflammation in PCOS.
Leukotriene A4 (LTA4), produced from AA, can be metabolized to either leukotriene B4 (LTB4) or LTC4. LTB4 is increased in PCOS patients [31]. In both, LET- and DHT-treated rats, LTC4 is significantly decreased. Reduced LTC4 levels may be due to the production of LTB4 from LTA4 instead of LTC4.
We have discovered a significant decrease in the concentration of D-Sph after DHT treatment, which indicates that sphingolipid metabolism is affected. However, we have not yet researched other lipids in this metabolism pathway, in particular ceramides, which may be involved in this syndrome [32].
In conclusion, our method was able to simultaneously measure 22 lipids spanning the metabolic pathways of steroids, steroid hormones, bile acids, FA-based eicosanoid precursors, eicosanoids, and sphingolipids. We have quantified 12 of these metabolites, with significant changes in the amounts of LTC4, 25-hydroxycholesterol and 4-cholesten-3-one in the plasma of rodent models of PCOS. These lipids may provide alternative biomarkers for the diagnosis of this syndrome. However, the models show markedly different lipid profiles to each other. Therefore, this study proposes that caution is required when assessing the results from different models due to distinct lipid differences, as well as the presence of novel changes that are yet to be validated in humans.
| Supplementary Material | ▴Top |
Suppl Fig 1. Chemical structures of the analyzed lipids. Lipids analyzed with this method include those within the groups of cholesterols, progestins, glucocorticoids and mineralocorticoids, estrogens and bioactive lipids. Their molecular mass is given in g/mol.
Suppl Fig 2. Analytical characteristics of testosterone (T). (A) The MS2 ion spectra of testosterone. Testosterone has a molar mass of 288.431. In positive electrospray ionization the protonated mass was identified at 289 m/z, the sodium adduct at 311 m/z and a protonated dimer at 577.1 m/z. There is a fragment ion caused by in-source fragmentation at 271.2 m/z. We used this information to cross reference our spectra with those found in databases (such as HMDB and LipidMaps) to corroborate identification of the correct lipid. We were also able to establish the retention time of the lipid in MS2 mode, to use in the setting of a retention time window for the dMRM transition. (B) dMRM chromatogram of testosterone. The protonated molecule at m/z 289.2 is exposed to a collisional energy 28 eV to obtain the fragment (product ion) of m/z 97.0, and collisional energy 32 eV for a product ion of 109.0 m/z. The product ion m/z 97.0 is the most abundant and is therefore used as the quantifier (quant) ion for determining concentration. (C) dMRM spectra of testosterone. The protonated ion (precursor ion) is indicated by the red star at 289.2 m/z. The fragments (product ions) are at m/z 97.0 and 109.0. The product ion 97.0 m/z is the most abundant and is therefore used as the quantifier (quant) ion for determining concentration. The use of ammonium acetate (CH3COONH4) as a mobile phase modifier can create [M+NH4]+ adducts, as seen in the analysis of 17α-hydroxypregnenolone (17αOHpreg) and 25-hydroxycholesterol (25OHchol), whereby some dMRM precursor ions are [M+NH4]+ adducts (a mass increase of 18). (D) Response curve of testosterone. Calibration curves (response curves) are generated using the response of the product ion in the mass spectrometer. On the X axis is the concentration, on the Y axis is the response (the counts), also known as the area under the curve (AUC). The trendline equation (Y = MX + C) is presented and accuracy of the trendline is shown by the R2.
Suppl Fig 3. Method reproducibility (intraday analysis). Lipid standards were combined and analyzed twice in technical triplicate at 100 fmol o.c. The mean percentage change from the first analysis was calculated (as %) for each standard. Most lipids had an intraday variation of less than ±5%.
Suppl Fig 4. Biological replicate analysis. Samples are in biological triplicate (n = 3). The coefficient of variation in percentage (CV %) is the standard deviation divided by the mean, multiplied by 100. The mean of the three biological replicates has been calculated for each lipid. LET-C represents non-treated control for letrozole, LET is for letrozole treated, DHT-C is a non-treated control for DHT, and DHT is dihydrotestosterone treated. A CV of less than 30% represents a good biological reproducibility, less than 20% is very good reproducibility and less than 10% is excellent reproducibility.
Suppl Fig 5. Technical replicate analysis. Technical replicates (× 3) analysis of the samples that are in biological triplicate (× 3), n = 9. The coefficient of variation in percentage (CV %) is the standard deviation divided by the mean, multiplied by 100. The mean of the three technical replicates, the mean of the means (black line) and standard error of the means (SEM, error bars) has been calculated for each lipid. LET-C represents non-treated control for letrozole, LET is for letrozole treated, DHT-C is a non-treated control for DHT, and DHT is dihydrotestosterone treated. A CV of less than 30% represents a good technical reproducibility, less than 20% is very good reproducibility and less than 10% is excellent reproducibility.
Suppl Table 1. Chromatography and mass spectrometry parameters. This table represents the instrumentation settings used in the analysis method and parameters applied. Settings are selected for the most favorable separation and ionization of steroids, steroid hormones, eicosanoids and sphingolipids.
Suppl Table 2. Optimized transitions for LC-dMRM-MS. Lipid names, dMRM parameters and lower limits of quantification (LLOQ) are listed for all lipid standards analyzed. The collisional energy used for the precursor-to-product ion transitions are indicated in electron volts (eV). The quantifier ion is indicated with bold text.
Suppl Table 3. Response (calibration) curve equations and accuracy. Increasing concentrations of the standard lipid mix were injected and the response of the quantitation (quant) ion was monitored by observing the area under the curve (AUC). The MRM AUCs were plotted against the on-column concentration to give response curves (calibration curves). Each response curve was plotted with a linear trendline. The values for the slope and intercept are given in the table above. R2: the accuracy of the fit of the linear trendline.
Suppl Table 4. Transition ion ratio (TIR) analysis of lipids identified in samples to identify potential isomers. TIR analysis requires the presence of both quantifier (qual) and qualifier (qual) ion transitions. A high average ratio of quant to qual ions indicates the presence of isomeric species. The reproducibility of the TIR average ratio is represented by the standard deviation (SD) and the coefficient of variation (CE). When the TIR analysis is not relevant or cannot be applied then it is represented by NA (not applicable).
Suppl Table 5. Biological variation analysis. Coefficient of variation of biological replicates: samples are in biological triplicate. The mean and standard deviation (std. dev/SD) has been calculated for biological replicates, and the coefficient of variation in percentage (CV %), given, is the standard deviation divided by the mean, multiplied by 100. LET-Control represents non-treated control for letrozole, LET is for letrozole treated, DHT-Control is a non-treated control for DHT. DHT: dihydrotestosterone treated.
Suppl Table 6. Technical variation analysis. Coefficient of variation (CV) in technical replicates: samples are in biological triplicate. The mean and standard deviation (std. dev) has been calculated for biological replicates, the coefficient of variation in percentage (CV %), given, is the standard deviation divided by the mean, multiplied by 100. LET-Control represents non-treated control for letrozole, LET is for letrozole treated, DHT-Control is a non-treated control for DHT. DHT: dihydrotestosterone treated.
Suppl Table 7. LET and DHT treatment of rats resulted in marked changes to the lipids profiled in rat plasma. The concentration (mean) of all analyses inclusive of technical triplicate of biological triplicates, the standard deviation (SD) and the number of analyses (N). The concentration units are given in each mean cell.
Acknowledgments
Many thanks to Danielle Wellington (Chinese Academy of Medical Sciences) for contributing to the editing of the abstract and to James Dunford (Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences) for providing the standards from the Oppermann lab.
Financial Disclosure
HCS, MF, JM, DO’B, GB, CB, CL, MP, UO, and BMK were funded by Bayer AG, Research and Development, Berlin, Germany. HCS, APF, ZY and BMK were additionally supported by the Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Science (CIFMS), China (grant no. 2018-I2M-2-002) and Pfizer.
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent
Not applicable.
Author Contributions
Conceptualization and writing-original draft preparation: HCS, MF, JM, and BMK; methodology: HCS, MF, JM, and GB; validation, formal analysis and visualization: HCS, MF, and JM; software and data curation: HCS and ZY; investigation: HCS, MF, JM, DO’B, CB, and CL; resources, supervision, project administration and funding acquisition: BMK, MF, MP, UO, and APF; writing-review and editing, HCS, DO’B, APF, and BMK. All authors have read and agreed to the published version of the manuscript.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
γ-LA: γ-linolenic acid; 3αAP/AP: 3α-hydroxy-5α-pregnane-20-one; 3β-HSD: 3β-hydroxysteroid dehydrogenase; 5-HPETE: 5-hydroperoxyeicosatetraenoic acid; A4: 4-androstene-3,17-dione; Aα5: 5α-androstane-3,17-dione; AA: arachidonic acid; AKR1C1/2/4: 3α-hydroxysteroid dehydrogenase class 1/2/4; ALOX5: arachidonate 5-lipoxygenase; ASAH1/2/ACER1/2: neutral ceramidase; AUC: area under the curve; CE: collisional energy; CH25H: cholesterol 25-hydroxylase b; choD: cholesterol oxidase; COX-2: cyclooxygenase 2; CV: coefficient of variation; CYP11: steroid 11β-monooxygenase; CYP11A: cytochrome P450 family 11 subfamily A; CYP11B1: steroid 11β-monooxygenase; CYP142: cholest-4-en-3-one 26-monooxygenase; CYP17A: steroid 17α-monooxygenase; CYP19A1: cytochrome P450 aromatase; CYP21A: steroid 21-monooxygenase; CYP2E1: cytochrome P450 family 2 subfamily E1; DHCR7: 7-dehydrocholesterol reductase; DHEA: dehydroepiandrosterone; DHEA-S: dehydroepiandrosterone-sulphate; DHT/5α-DHT: 5α-dihydrotestosterone; DHT-C: dihydrotestosterone control; diOHchol: dihydrocholesterol; D-Sph/Sph: D-sphingosine; dMRM: dynamic multiple reaction monitoring; E1: estrone; E1.1.1.51: 3 (or 17) β-hydroxysteroid dehydrogenase; E2: β-estradiol; E3: estriol; E5.3.3.1: steroid Δ-isomerase; GABAA: γ-aminobutyric acid type A; GALC: galactosylceramidase; GBA: non-lysosomal glucosylceramidase; GGT1/5: leukotriene-C4 hydrolase; GPX4: phospholipid-hydroperoxide glutathione peroxidase; H2O: water; HSD11B1/2: hydroxysteroid 11-β-dehydrogenase isozyme 1/2; HSD17B1/2: 17β-estradiol 17-dehydrogenase 1/2; HSD17B3: testosterone 17β-dehydrogenase (NADP+); HSD17B11: hydroxysteroid 17β-dehydrogenase 11; IM: ion mobility; IPA: isopropanol; LC-dMRM-MS: liquid chromatography-dynamic multiple reaction monitoring-mass spectrometry; LET: letrozole; LET-C: letrozole control; LLOD: lower limit of detection; LLOQ: lower limit of quantification; LTA4: leukotriene A4; LTA4H: leukotriene A4 hydrolase; LTB4: leukotriene B4; LTC4: leukotriene C4; LTC4S: leukotriene C4 synthase; MAGL: monoacylglycerol lipase; MeOH: methanol; MRM: multiple reaction monitoring; MTBE: methyl tert-butyl ether; o.c.: on-column; PCOS: polycystic ovarian syndrome; PLGA: poly-lactic-glycolic acid; PLPP1/2/3: phosphatidate phosphatase 1/2/3; Preg: pregnenolone; Prog: progesterone; PTGR1/2: prostaglandin reductase 1/2; PTGS1/2: prostaglandin-endoperoxide synthase 1; QC: quality control; Qual: qualifier ion; Quant: quantifier ion; RF: response factor; S/N: signal-to-noise ratio; SGMS1/2: sphingomyelin synthase; SGPP1/2: sphingosine-1-phosphate phosphatase 1/2; SMPD1-4/ENPP7: ectonucleotide pyrophosphatase/phosphodiesterase family member 7; SPHK1/2: sphingosine kinase 1/2; SRD5A1: 3-oxo-5α-steroid 4-dehydrogenase 1; STD: standard; STS: stearyl-sulfatase; SULT1E1: estrone sulfotransferase; SULT2B: sulfotransferase family 2B member 1; TesI: 3-oxo-5α-steroid 4-dehydrogenase; TIR: transition ion ratios; UGT: uridine diphosphoglucuronosyltransferase; UGT8: ceramide galactosyltransferase; ZADH2: zinc binding alcohol dehydrogenase domain containing 2
| References | ▴Top |
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop
group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to
polycystic ovary syndrome (PCOS). Hum Reprod. 2004;19(1):41-47.
doi pubmed - Falsetti L, Dordoni D, Omodei U, Ghielmi S, Gastaldi C. A
new method for free testosterone assay: clinical evaluation. Acta Eur Fertil.
1985;16(5):343-346.
pubmed - Cotrozzi G, Matteini M, Relli P, Lazzari T. Hyperinsulinism
and insulin resistance in polycystic ovarian syndrome: a verification using oral glucose, I.V.
Glucose and tolbutamide. Acta Diabetol Lat. 1983;20(2):135-142.
doi pubmed - Ehrmann DA. Polycystic ovary syndrome.
N Engl J Med. 2005;352(12):1223-1236.
doi pubmed - Wajchenberg BL, Achando SS, Okada H, Czeresnia CE, Peixoto
S, Lima SS, Goldman J. Determination of the source(s) of androgen overproduction in hirsutism
associated with polycystic ovary syndrome by simultaneous adrenal and ovarian venous
catheterization. Comparison with the dexamethasone suppression test. J Clin Endocrinol
Metab. 1986;63(5):1204-1210.
doi pubmed - Timpatanapong P, Rojanasakul A. Hormonal profiles and
prevalence of polycystic ovary syndrome in women with acne. J Dermatol.
1997;24(4):223-229.
doi pubmed - Cela E, Robertson C, Rush K, Kousta E, White DM, Wilson H,
Lyons G, et al. Prevalence of polycystic ovaries in women with androgenic alopecia.
Eur J Endocrinol. 2003;149(5):439-442.
doi pubmed - Repaci A, Gambineri A, Pasquali R. The role of low-grade
inflammation in the polycystic ovary syndrome. Mol Cell Endocrinol. 2011;335(1):30-41.
doi pubmed - Martin KA, Anderson RR, Chang RJ, Ehrmann DA, Lobo RA, Murad
MH, Pugeat MM, et al. Evaluation and treatment of hirsutism in premenopausal women: an endocrine
society clinical practice guideline. J Clin Endocrinol Metab.
2018;103(4):1233-1257.
doi pubmed - Grassi G, Polledri E, Fustinoni S, Chiodini I, Ceriotti F,
D'Agostino S, Filippi F, et al. Hyperandrogenism by liquid chromatography tandem mass
spectrometry in PCOS: focus on testosterone and androstenedione. J Clin Med.
2020;10(1):119.
doi pubmed - Gravitte A, Archibald T, Cobble A, Kennard B, Brown S.
Liquid chromatography-mass spectrometry applications for quantification of endogenous sex
hormones. Biomed Chromatogr. 2021;35(1):e5036.
doi pubmed - Conklin SE, Knezevic CE. Advancements in the gold standard:
Measuring steroid sex hormones by mass spectrometry. Clin Biochem. 2020;82:21-32.
doi pubmed - Guan F, Uboh CE, Soma LR, Luo Y, Rudy J, Tobin T. Detection,
quantification and confirmation of anabolic steroids in equine plasma by liquid chromatography
and tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life
Sci. 2005;829(1-2):56-68.
doi pubmed - Masood MA, Khatoon R, Veenstra TD. Quantitative analysis of
specific androgens in serum and urine samples from male, pre, and postmenopausal subjects using
LC-MRM-MS. Steroids. 2022;185:109060.
doi pubmed - Hakkinen MR, Heinosalo T, Saarinen N, Linnanen T,
Voutilainen R, Lakka T, Jaaskelainen J, et al. Analysis by LC-MS/MS of endogenous steroids from
human serum, plasma, endometrium and endometriotic tissue. J Pharm Biomed Anal.
2018;152:165-172.
doi pubmed - Peti APF, Locachevic GA, Prado MKB, de Moraes LAB, Faccioli
LH. High-resolution multiple reaction monitoring method for quantification of steroidal hormones
in plasma. J Mass Spectrom. 2018;53(5):423-431.
doi pubmed - Rosenfield RL, Ehrmann DA. The Pathogenesis of Polycystic
Ovary Syndrome (PCOS): the hypothesis of PCOS as functional ovarian hyperandrogenism revisited.
Endocr Rev. 2016;37(5):467-520.
doi pubmed - Manni A, Pardridge WM, Cefalu W, Nisula BC, Bardin CW,
Santner SJ, Santen RJ. Bioavailability of albumin-bound testosterone. J Clin Endocrinol
Metab. 1985;61(4):705-710.
doi pubmed - Agarwal SK, Judd HL, Magoffin DA. A mechanism for the
suppression of estrogen production in polycystic ovary syndrome. J Clin Endocrinol Metab.
1996;81(10):3686-3691.
doi pubmed - Yildiz BO, Azziz R. The adrenal and polycystic ovary
syndrome. Rev Endocr Metab Disord. 2007;8(4):331-342.
doi pubmed - Cascella T, Palomba S, Tauchmanova L, Manguso F, Di Biase S,
Labella D, Giallauria F, et al. Serum aldosterone concentration and cardiovascular risk in women
with polycystic ovarian syndrome. J Clin Endocrinol Metab. 2006;91(11):4395-4400.
doi pubmed - Yang Z, Zhou W, Zhou C, Zhou Y, Liu X, Ding G, Hu Y, et al.
Steroid metabolome profiling of follicular fluid in normo- and hyperandrogenic women with
polycystic ovary syndrome. J Steroid Biochem Mol Biol. 2021;206:105806.
doi pubmed - Loughlin T, Cunningham S, Moore A, Culliton M, Smyth PP,
McKenna TJ. Adrenal abnormalities in polycystic ovary syndrome. J Clin Endocrinol Metab.
1986;62(1):142-147.
doi pubmed - Wang Z, Wang H, Peng Y, Chen F, Zhao L, Li X, Qin J, et al.
A liquid chromatography-tandem mass spectrometry (LC-MS/MS)-based assay to profile 20 plasma
steroids in endocrine disorders. Clin Chem Lab Med. 2020;58(9):1477-1487.
doi pubmed - Gennarelli G, Holte J, Stridsberg M, Lundqvist U, Massobrio
M, Backstrom T, Berne C. Response of the pituitary-adrenal axis to hypoglycemic stress in women
with the polycystic ovary syndrome. J Clin Endocrinol Metab. 1999;84(1):76-81.
doi pubmed - Bongrani A, Plotton I, Mellouk N, Rame C, Guerif F, Froment
P, Dupont J. High androgen concentrations in follicular fluid of polycystic ovary syndrome
women. Reprod Biol Endocrinol. 2022;20(1):88.
doi pubmed - Yu L, Liu M, Wang Z, Liu T, Liu S, Wang B, Pan B, et al.
Correlation between steroid levels in follicular fluid and hormone synthesis related substances
in its exosomes and embryo quality in patients with polycystic ovary syndrome. Reprod Biol
Endocrinol. 2021;19(1):74.
doi pubmed - Fritz MA, Speroff L. Clinical gynecologic endocrinology and infertility. 8th ed. Philadelphia: Lippincott Williams & Wilkins. 2011;501-518.
- Lizneva D, Gavrilova-Jordan L, Walker W, Azziz R. Androgen
excess: investigations and management. Best Pract Res Clin Obstet Gynaecol.
2016;37:98-118.
doi pubmed - Kim JY, Song H, Kim H, Kang HJ, Jun JH, Hong SR, Koong MK,
et al. Transcriptional profiling with a pathway-oriented analysis identifies dysregulated
molecular phenotypes in the endometrium of patients with polycystic ovary syndrome.
J Clin Endocrinol Metab. 2009;94(4):1416-1426.
doi pubmed - Regidor PA, de la Rosa X, Muller A, Mayr M, Gonzalez Santos
F, Gracia Banzo R, Rizo JM. PCOS: a chronic disease that fails to produce adequately specialized
pro-resolving lipid mediators (SPMs). Biomedicines. 2022;10(2):456.
doi pubmed - Eliyahu E, Shtraizent N, Shalgi R, Schuchman EH.
Construction of conditional acid ceramidase knockout mice and in vivo effects on oocyte
development and fertility. Cell Physiol Biochem. 2012;30(3):735-748.
doi pubmed - Li S, Chu Q, Ma J, Sun Y, Tao T, Huang R, Liao Y, et al.
Discovery of novel lipid profiles in PCOS: do insulin and androgen oppositely regulate bioactive
lipid production? J Clin Endocrinol Metab. 2017;102(3):810-821.
doi pubmed - Demers LM. Effects of Fadrozole (CGS 16949A) and Letrozole
(CGS 20267) on the inhibition of aromatase activity in breast cancer patients. Breast Cancer Res
Treat. 1994;30(1):95-102.
doi pubmed - Bansal S, Goyal M, Sharma C, Shekhar S. Letrozole versus
clomiphene citrate for ovulation induction in anovulatory women with polycystic ovarian
syndrome: A randomized controlled trial. Int J Gynaecol Obstet.
2021;152(3):345-350.
doi pubmed - Kafali H, Iriadam M, Ozardali I, Demir N. Letrozole-induced
polycystic ovaries in the rat: a new model for cystic ovarian disease. Arch Med Res.
2004;35(2):103-108.
doi pubmed - Manneras L, Cajander S, Holmang A, Seleskovic Z, Lystig T,
Lonn M, Stener-Victorin E. A new rat model exhibiting both ovarian and metabolic characteristics
of polycystic ovary syndrome. Endocrinology. 2007;148(8):3781-3791.
doi pubmed - Lin Q, Mueller J, Fritsch M, Lesche R, Kageyama J, Zollner TM. Towards the molecular understanding of PCOS pathogenesis by RNA-seq analysis of multiple tissues of two rat PCOS models. Reproductive Sciences. 2021;28(SUPPL 1):295A-295A.
- Scott HC, Draganov SD, Yu Z, Kessler BM, Pinto-Fernandez A.
Targeted mass spectrometry reveals interferon-dependent eicosanoid and fatty acid alterations in
chronic myeloid leukaemia. Int J Mol Sci. 2023;24(21):15513.
doi pubmed - Zhang X, Xu H, Feng G, Yang L, Zhao L, Li R, Zhao R.
Sensitive HPLC-DMS/MS/MS method coupled with dispersive magnetic solid phase extraction followed
by in situ derivatization for the simultaneous determination of multiplexing androgens and
17-hydroxyprogesterone in human serum and its application to patients with polycystic ovarian
syndrome. Clin Chim Acta. 2023;538:221-230.
doi pubmed - Guo N, Liu P, Ding J, Zheng SJ, Yuan BF, Feng YQ. Stable
isotope labeling - Liquid chromatography/mass spectrometry for quantitative analysis of
androgenic and progestagenic steroids. Anal Chim Acta. 2016;905:106-114.
doi pubmed - Ke Y, Gonthier R, Labrie F. A sensitive and accurate
LC-MS/MS assay with the derivatization of 1-Amino-4-methylpiperazine applied to serum
allopregnanolone, pregnenolone and androsterone in pre- and postmenopausal women. Steroids.
2017;118:25-31.
doi pubmed - Hernandez-Mesa M, Le Bizec B, Monteau F, Garcia-Campana AM,
Dervilly-Pinel G. Collision Cross Section (CCS) database: an additional measure to characterize
steroids. Anal Chem. 2018;90(7):4616-4625.
doi pubmed - Bunch DR, McShane AJ, Wang S. Investigation of transition
ion ratio variation for liquid chromatography-tandem mass spectrometry: A case study approach.
Clin Chim Acta. 2018;486:205-208.
doi pubmed - Pinna G, Almeida FB, Davis JM. Allopregnanolone in
postpartum depression. Front Glob Womens Health. 2022;3:823616.
doi pubmed - Monteleone P, Luisi S, Tonetti A, Bernardi F, Genazzani AD,
Luisi M, Petraglia F, et al. Allopregnanolone concentrations and premenstrual syndrome.
Eur J Endocrinol. 2000;142(3):269-273.
doi pubmed - Wang M, Seippel L, Purdy RH, Backstrom T. Relationship
between symptom severity and steroid variation in women with premenstrual syndrome: study on
serum pregnenolone, pregnenolone sulfate, 5 alpha-pregnane-3,20-dione and 3 alpha-hydroxy-5
alpha-pregnan-20-one. J Clin Endocrinol Metab. 1996;81(3):1076-1082.
doi pubmed - Cooney LG, Lee I, Sammel MD, Dokras A. High prevalence of
moderate and severe depressive and anxiety symptoms in polycystic ovary syndrome: a systematic
review and meta-analysis. Hum Reprod. 2017;32(5):1075-1091.
doi pubmed - Barbaccia ML, Roscetti G, Trabucchi M, Purdy RH, Mostallino
MC, Concas A, Biggio G. The effects of inhibitors of GABAergic transmission and stress on brain
and plasma allopregnanolone concentrations. Br J Pharmacol.
1997;120(8):1582-1588.
doi pubmed - Strous RD, Maayan R, Weizman A. The relevance of
neurosteroids to clinical psychiatry: from the laboratory to the bedside. Eur
Neuropsychopharmacol. 2006;16(3):155-169.
doi pubmed - Holmberg E, Sjostedt J, Malinina E, Johansson M, Turkmen S,
Ragagnin G, Lundqvist A, et al. Allopregnanolone involvement in feeding regulation, overeating
and obesity. Front Neuroendocrinol. 2018;48:70-77.
doi pubmed - Genazzani AD, Strucchi C, Luisi M, Casarosa E, Lanzoni C,
Baraldi E, Ricchieri F, et al. Metformin administration modulates neurosteroids secretion in
non-obese amenorrhoic patients with polycystic ovary syndrome. Gynecol Endocrinol.
2006;22(1):36-43.
doi pubmed - Saito K, Matsuzaki T, Iwasa T, Miyado M, Saito H, Kubota T,
Irahara M, et al. Blood allopregnanolone levels in women with polycystic ovary syndrome. Clin
Endocrinol (Oxf). 2016;85(1):151-152.
doi pubmed - Elia J, Carbonnelle D, Loge C, Ory L, Huvelin JM, Tannoury
M, Diab-Assaf M, et al. 4-cholesten-3-one decreases breast cancer cell viability and alters
membrane raft-localized EGFR expression by reducing lipogenesis and enhancing LXR-dependent
cholesterol transporters. Lipids Health Dis. 2019;18(1):168.
doi pubmed - Tam SP, Mok L, Chimini G, Vasa M, Deeley RG. ABCA1 mediates
high-affinity uptake of 25-hydroxycholesterol by membrane vesicles and rapid efflux of oxysterol
by intact cells. Am J Physiol Cell Physiol. 2006;291(3):C490-502.
doi pubmed - Luo Y, Nie Y, Tang L, Xu CC, Xu L. The correlation between
UDP-glucuronosyltransferase polymorphisms and environmental endocrine disruptors levels in
polycystic ovary syndrome patients. Medicine (Baltimore). 2020;99(11):e19444.
doi pubmed - Caldwell AS, Middleton LJ, Jimenez M, Desai R, McMahon AC,
Allan CM, Handelsman DJ, et al. Characterization of reproductive, metabolic, and endocrine
features of polycystic ovary syndrome in female hyperandrogenic mouse models. Endocrinology.
2014;155(8):3146-3159.
doi pubmed - Yazawa T, Kawabe S, Kanno M, Mizutani T, Imamichi Y, Ju Y,
Matsumura T, et al. Androgen/androgen receptor pathway regulates expression of the genes for
cyclooxygenase-2 and amphiregulin in periovulatory granulosa cells. Mol Cell Endocrinol.
2013;369(1-2):42-51.
doi pubmed - Sales KJ, Jabbour HN. Cyclooxygenase enzymes and
prostaglandins in pathology of the endometrium. Reproduction. 2003;126(5):559-567.
doi pubmed - Ma Y, Zheng L, Wang Y, Gao Y, Xu Y. Corrigendum: Arachidonic
acid in follicular fluid of PCOS induces oxidative stress in a human ovarian granulosa tumor
cell line (KGN) and upregulates GDF15 expression as a response. Front Endocrinol (Lausanne).
2022;13:988767.
doi pubmed - Huang R, Xue X, Li S, Wang Y, Sun Y, Liu W, Yin H, et al.
Alterations of polyunsaturated fatty acid metabolism in ovarian tissues of polycystic ovary
syndrome rats. J Cell Mol Med. 2018;22(7):3388-3396.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Endocrinology and Metabolism is published by Elmer Press Inc.