Altered Hormone and Bioactive Lipid Plasma Profile in Rodent Models of Polycystic Ovarian Syndrome Revealed by Targeted Mass Spectrometry
DOI:
https://doi.org/10.14740/jem1042Keywords:
Steroid hormones, Lipids, Eicosanoids, Sphingolipids, Lipidomics, Letrozole, Hyperandrogenism, InflammationAbstract
Background: Polycystic ovarian syndrome (PCOS) symptoms include excessive body or facial hair, irregular periods, reduced fertility, and reoccurring pregnancy loss. Hyperandrogenism and chronic inflammation are hallmarks of PCOS, which is diagnosed by analyzing steroid hormones in the blood. Studies suggest that bioactive lipids are contributing to chronic inflammation.
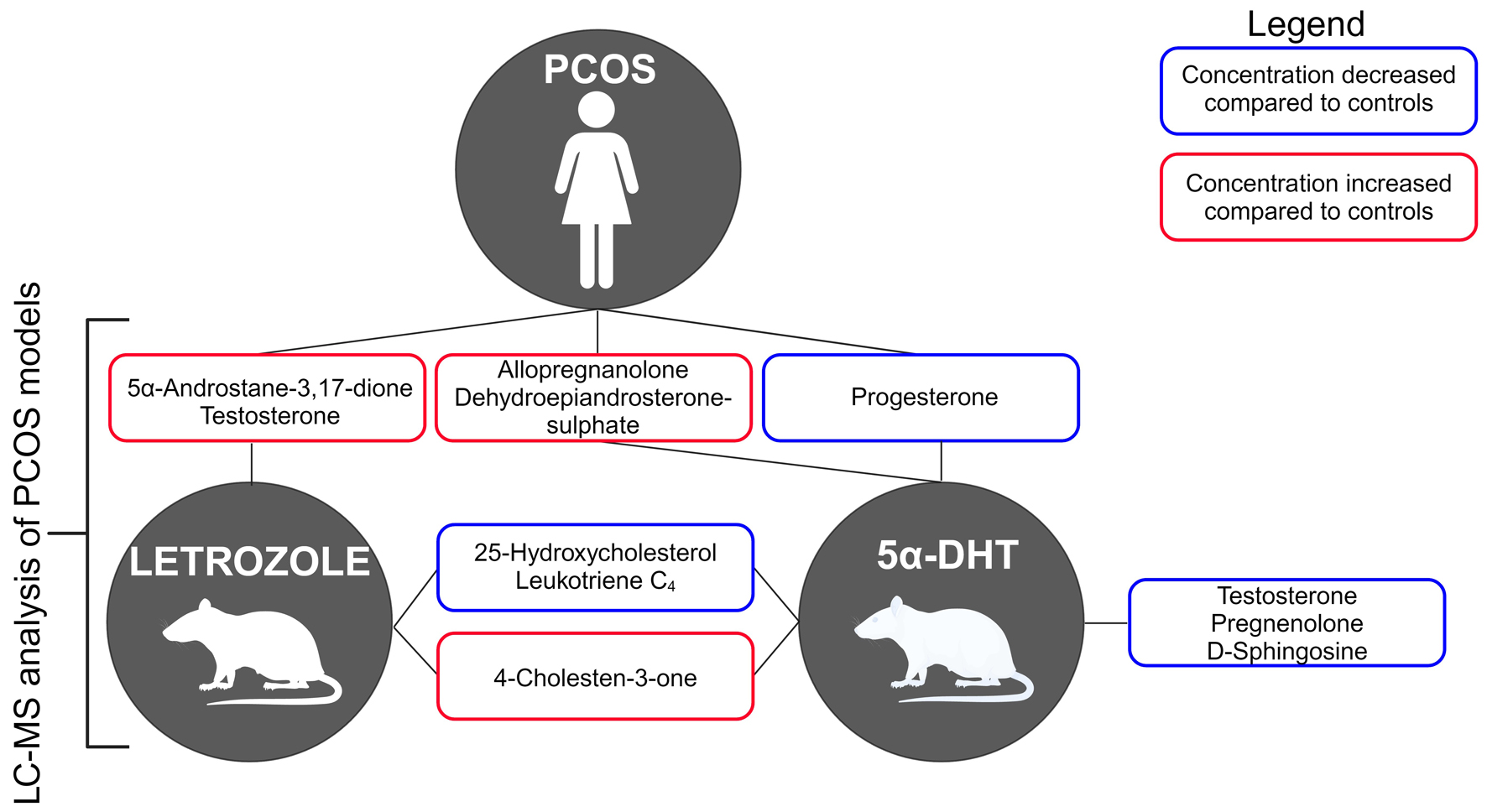
Methods: To research PCOS, animal models, such as letrozole- and dihydrotestosterone-treated rats, are used. They display similar ovarian and metabolic characteristics, although plasma lipid profiles have not been determined. Therefore, in order to validate the use of these models for PCOS, we have optimized a mass spectrometry-based targeted lipidomics workflow, which increases the sensitivity of measuring these lipids in rat plasma.
Results: Our analysis shows that letrozole caused a significant elevation of 5α-androstene-3,17-dione and testosterone. Dihydrotestosterone treatment resulted in increased dehydroepiandrosterone-sulphate and allopregnanolone but a reduction in testosterone, progesterone, pregnenolone, and D-sphingosine. In both models, 25-hydroxycholesterol and leukotriene C4 were significantly diminished, and 4-cholesten-3-one was significantly increased, and these particular metabolites are not known to be changed in human PCOS.
Conclusion: These results suggest that the plasma lipids of these rodent models exert altered profiles of sterols, leukotrienes and steroid hormones akin to human PCOS but with notable differences.
Downloads
Published
Issue
Section
License
Copyright (c) 2025 The authors

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.









