From Inflammation to Endocrine Dysregulation: Thyroid Hormonal Alterations in COVID-19
DOI:
https://doi.org/10.14740/jem1625Keywords:
COVID-19, Thyroid function, FT3, TSH, HIF-1α, Inflammation, Post-COVIDAbstract
Background: Although thyroid dysfunction has been increasingly reported in coronavirus disease 2019 (COVID-19), the mechanisms linking severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection to thyroid hormonal disturbances remain incompletely understood. The study aimed to examine the relationship between inflammatory status and thyroid function in patients with active COVID-19 and in post-COVID individuals, compared with healthy individuals.
Methods: An observational study with a cross-sectional analytical approach was conducted in 101 participants allocated into three groups: patients with active COVID-19 (group 1, n = 33), individuals with thyroid dysfunction following recovery from COVID-19 (group 2, n = 35), and a non-infected reference group (group 3, n = 33). Thyroid parameters, including thyroid-stimulating hormone (TSH), free triiodothyronine (FT3), free thyroxine (FT4), thyroglobulin (Tg), and thyroid peroxidase (TPO); selected cytokines, including interleukin-17A (IL-17A), interleukin-10 (IL-10), interleukin-7 (IL-7), and interferon-gamma (IFN-γ); and markers related to hypoxia and antioxidant defense, including hypoxia-inducible factor 1-alpha (HIF-1α) and nuclear factor erythroid 2-related factor 2 (NFE2L2), were assessed.
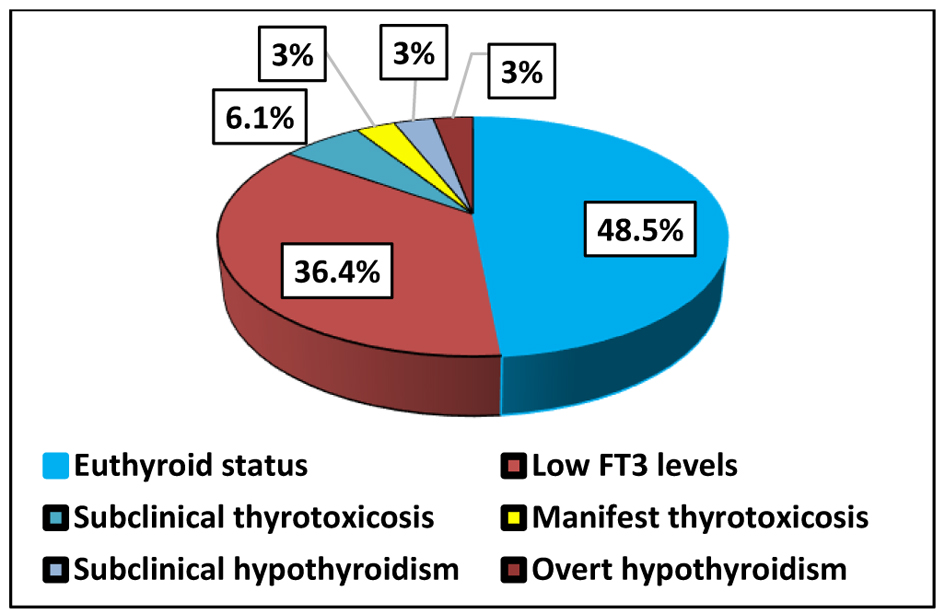
Results: Significant between-group differences were observed for FT3 and TSH, with the lowest FT3 levels detected in the active COVID-19 group. Reduced FT3 was the most frequent abnormality in group 1, identified in 36.4% of patients, supporting a pattern consistent with euthyroid sick syndrome. TSH levels were significantly lower in group 1 than in group 3, while FT3 levels differed significantly between group 1 and both group 2 and group 3. HIF-1α and NFE2L2 levels were highest in active COVID-19, indicating marked intracellular hypoxia together with activation of the antioxidant defense response. Receiver operating characteristic analysis showed good discriminatory performance of HIF-1α for distinguishing active COVID-19 from the reference group (area under the curve = 0.771). Post-COVID patients showed the lowest median levels of IL-17A, IL-10, IL-7, and IFN-γ, together with a high frequency of autoimmune thyroid disorders, most commonly Hashimoto’s thyroiditis, present in 65.7% of patients.
Conclusion: SARS-CoV-2 infection appears to induce transient thyroid hormonal alterations primarily through inflammatory and hypoxia-related mechanisms. These findings support the concept of virus-induced euthyroid sick syndrome during acute infection and highlight the potential for post-infectious thyroid dysfunction.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









