Imeglimin: Discovery, Pharmacology, and Trials
DOI:
https://doi.org/10.14740/jem1605Keywords:
Imeglimin, Discovery, Pharmacology, T2DMAbstract
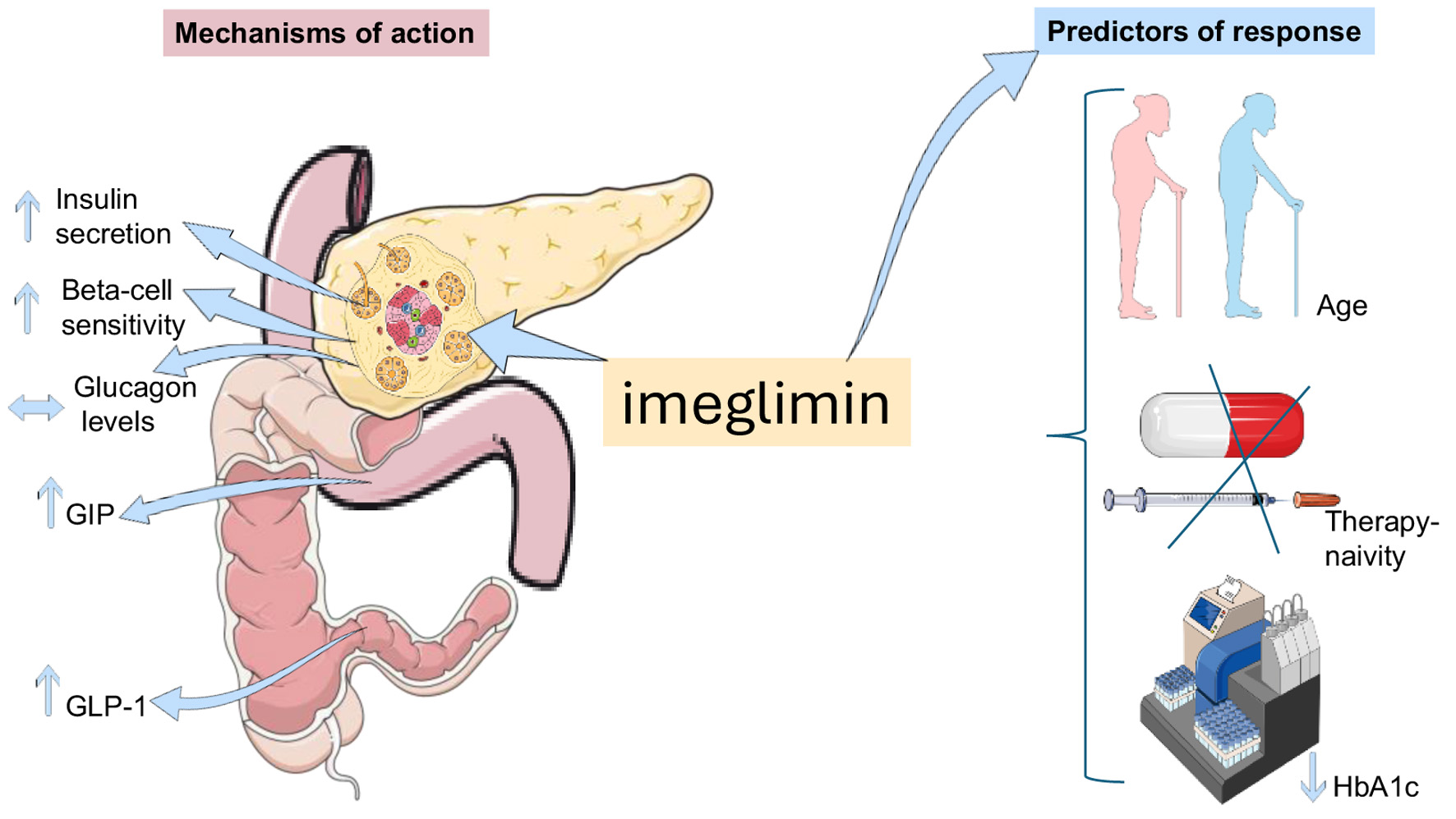
This review briefly summarizes imeglimin’s discovery, development, properties, mechanisms, and core findings on efficacy and safety from major studies. Imeglimin is a first-in-class, orally available tetrahydro-triazine that was designed from the metformin scaffold to address the pathophysiological defects of β-cell dysfunction and insulin resistance in type 2 diabetes mellitus (T2DM). Pre-clinical work shows that imeglimin partially inhibits mitochondrial complex I, corrects complex III deficiency, lowers reactive oxygen species, and boosts NAD+-dependent ATP generation. This enhances glucose-stimulated insulin secretion and preserves β-cell mass. Additionally, the compound augments endogenous glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) release and improves hepatic and skeletal-muscle insulin signaling, resulting in combined insulinotropic and insulin-sensitizing actions. Pharmacokinetic studies reveal rapid absorption, an elimination half-life of approximately 9–12 h, high oral bioavailability, and predominantly renal excretion with no clinically meaningful interactions with metformin or sitagliptin. In phase 2 and 3 trials, the optimized regimen of 1,000 mg twice daily consistently lowers glycated hemoglobin (HbA1c) by approximately 0.8–0.9% as monotherapy and provides additional reductions of 0.4–0.6% when combined with metformin or insulin. It maintains placebo-like tolerability and a minimal risk of hypoglycemia. Long-term data confirm durable glycemic efficacy, a neutral cardiovascular profile with no QT/QTc prolongation, and predominantly mild gastrointestinal adverse events. Real-world evidence supports sustained HbA1c lowering, modest weight loss, and lipid improvements over 12 months, independent of age, sex, body mass index, or renal function. Multivariate and cluster analyses suggest that older age, therapy-naive status, and lower baseline HbA1c predict a more pronounced response, highlighting the importance of patient stratification. Research on imeglimin for T2DM is constrained by small sample sizes, mainly Japanese data, lack of cardiovascular outcome trials, limited comparisons with other antidiabetic drugs, and insufficient long-term safety information. More studies are needed to clarify its efficacy and safety profiles.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









