The Influence of Age on the Efficacy of a Novel Antidiabetic Drug Imeglimin in Patients With Poorly Controlled Type 2 Diabetes
DOI:
https://doi.org/10.14740/jem1637Keywords:
Imeglimin, Type 2 diabetes, Antidiabetic drug, Japan, Aging, HbA1c, Glycemic control, Hepatic functionAbstract
Background: Imeglimin is a novel drug shown to improve mitochondrial function in patients with diabetes. This study was undertaken to evaluate the efficacy and safety of imeglimin in patients with poorly controlled type 2 diabetes. The effect of aging on the efficacy and safety of this drug was also evaluated.
Methods: Imeglimin (2,000 mg/kg) was administered for 3 months to 52 patients. Hemoglobin A1c (HbA1c) and other blood biochemical parameters were measured before and after the 3-month treatment period. Regression analysis was performed to determine the influence of age on the effects of the drug on these parameters.
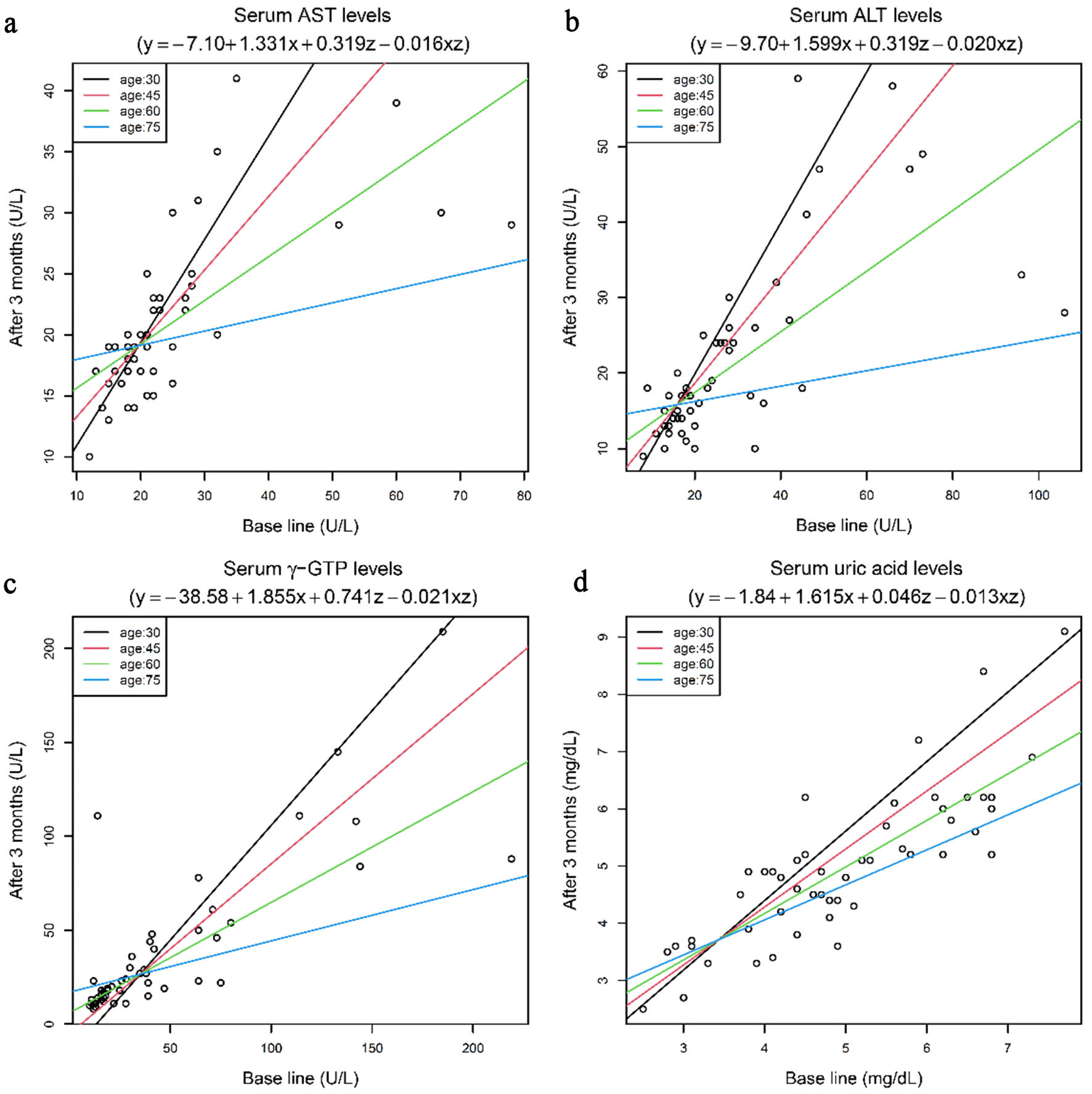
Results: Significant reduction of HbA1c and serum lipids were noted after the treatment. Regression analysis to determine the influence of age on relevant biochemical parameters in patients receiving imeglimin revealed that the biochemical effects of this drug on the serum levels of the hepatic function parameters (aspartate aminotransferase (AST), alanine aminotransferase (ALT), and γ-glutamyl transferase (γ-GTP)) and the serum uric acid level were influenced by interactions between the patient’s age and the pre-treatment baseline levels of the parameters. Older patients with higher baseline levels of these parameters demonstrated greater reduction in hepatic biomarkers following treatment with imeglimin.
Conclusions: This study confirmed imeglimin as a useful drug for individuals with type 2 diabetes who exhibit poor glycemia control. The effects of imeglimin in reducing the serum levels of hepatic function parameters were reinforced by aging, indicating that imeglimin is a particularly useful drug for patients with type 2 diabetes who are elderly and/or have hepatopathy.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.









